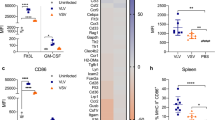
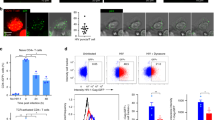
Abstract
Dendritic cells and macrophages can process extracellular antigens for presentation by MHC-I molecules. This exogenous pathway may have a crucial role in the activation of CD8+ cytotoxic T lymphocytes during human viral infections. We show here that HIV-1 epitopes derived from incoming virions are presented through the exogenous MHC-I pathway in primary human dendritic cells, and to a lower extent in macrophages, leading to cytotoxic T-lymphocyte activation in the absence of viral protein synthesis. Exogenous antigen presentation required adequate virus-receptor interactions and fusion of viral and cellular membranes. These results provide new insights into how anti-HIV cytotoxic T lymphocytes can be activated and have implications for anti-HIV vaccine design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rock, K.L. A new foreign policy: MHC class I molecules monitor the outside world. Immunol. Today 17, 129–137 (1996).
Jondal, M., Schirmbeck, R. & Reimann, J. MHC class I-restricted CTL responses to exogenous antigens. Immunity 5, 295–302 (1996).
Yewdell, J.W., Norbury, C.C., Bennink, J.R. Mechanisms of exogenous antigen presentation by MHC class I molecules in vitro and in vivo: Implications for generating CD8+ T cell responses to infectious agents, tumors, transplants, and vaccines. Adv. Immunol. 73, 1–77 (1999).
Lanzavecchia, A. Mechanisms of antigen uptake for presentation. Curr. Op. Immunol. 8, 348–354 (1996).
Watts, C. Capture and processing of exogenous antigens for presentation on MHC molecules. Ann. Rev. Immunol. 15, 821–850 (1997).
Kovacsovics-Bankowski, M. & Rock, K.L. A phagosome to cytosol pathway for exogenous antigens presented on MHC class I molecules. Science 267, 243–246 (1995).
Singh-Jasuja, H. et al. Cross-presentation of glycoprotein 96-associated antigens on major histocompatibility complex class I molecules requires receptor-mediated endocytosis. J. Exp. Med. 191, 1965–1974 (2000).
Castellino, F. et al. Receptor-mediated uptake of antigen/heat shock protein complexes results in major histocompatibility complex class 1 antigen presentation via two distinct processing pathways. J. Exp. Med. 191, 1957–1964 (2000).
Regnault, A. et al. Fcγ receptor-mediated induction of dendritic cell maturation and major histocompatibility complex class I-restricted antigen presentation after immune complex internalization. J. Exp. Med. 189, 371–380 (1999).
Rodriguez, A., Regnault, A., Kleijmeer, M., Ricciardi-Castagnoli, P. & Amigorena, S. Selective transport of internalized antigens to the cytosol for MHC class I presentation in dendritic cells. Nature Cell Biol. 1, 362–368 (1999).
Gromme, M. et al. Recycling MHC class I molecules and endosomal peptide loading. Proc. Nat. Acad. Sci. USA 96, 10326–10331 (1999).
Banchereau, J. & Steinman, R.M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Albert, M.L., Sauter, B. & Bhardwaj, N. Dendritic cells acquire antigen from apoptotic cells and induce class I-restricted CTLs. Nature 392, 86–89 (1998).
Sigal, L.J., Crotty, S., Andino, R. & Rock, K.L. Cytotoxic T-cell immunity to virus-infected non-hematopoietic cells requires presentation of exogenous antigen. Nature 398, 77–80 (1999).
Yewdell, J.W., Bennink, J.R. & Hosaka, Y. Cells process exogenous proteins for recognition by cytotoxic T lymphocytes. Science 239, 637–640 (1988).
Reimann, J. & Schirmbeck, R. Alternative pathways for processing exogenous and endogenous antigens that can generate peptides for MHC class I-restricted presentation. Immunol. Rev. 172, 131–152 (1999).
Cameron, P., Pope, M., Granelli-Piperno, A. & Steinman, R.M. Dendritic cells and the replication of HIV-1. J. Leuk. Biol. 59, 158–171 (1996).
Knight, S.C. & Patterson, S. Bone marrow-derived dendritic cells, infection with human immunodeficiency virus, and immunopathology. Annu. Rev. Immunol. 15, 593–615 (1997).
Granelli-Piperno, A. et al. Efficient interaction of HIV-1 with purified dendritic cells via multiple chemokine coreceptors. J. Exp. Med. 184, 2433–2438 (1996).
Ayehunie, S. et al. Human immunodeficiency virus-1 entry into purified blood dendritic cells through CC and CXC chemokine coreceptors. Blood 90, 1379–1386 (1997).
Klagge, I.M. & Schneider-Schaulies, S. Virus interactions with dendritic cells. J. Gen. Virol. 80, 823–833 (1999).
Granelli-Piperno, A., Finkel, V., Delgado, E. & Steinman, R.M. Virus replication begins in dendritic cells during the transmission of HIV-1 from mature dendritic cells to T cells. Curr. Biol. 9, 21–29 (1999).
Geijtenbeek, T.B. et al. DC-SIGN, a dendritic cell-specific HIV-1–binding protein that enhances trans-infection of T cells. Cell 100, 587–597 (2000).
Lapham, C.K., Zaitseva, M.B., Lee, S., Romanstseva, T. & Golding, H. Fusion of monocytes and macrophages with HIV-1 correlates with biochemical properties of CXCR4 and CCR5. Nature Med. 5, 303–308 (1999).
Aiken, C. Pseudotyping HIV-1 by the glycoprotein of VSV targets HIV-1 entry to an endocytic pathway and suppresses both the requirement for Nef and the sensitivity to cyclosporin A. J. Virol. 71, 5871–5877 (1997).
Yang, O.O. et al. Efficient lysis of Human Immunodeficiency Virus type 1-infected cells by cytotoxic T lymphocytes. J. Virol. 70, 5799–5806 (1996).
Naldini, L. et al. In vivo gene delivery and stable transduction of non dividing cells by a lentiviral vector. Science 272, 263–267 (1996).
Rossio, J.L. et al. Inactivation of human immunodeficiency virus type 1 infectivity with preservation of conformational and functional integrity of virion surface proteins. J. Virol. 72, 7992–8001 (1998).
Arthur, L.O. et al. Chemical inactivation of retroviral infectivity by targeting nucleocapsid protein zinc fingers: a candidate SIV vaccine. AIDS Res. Hum. Retrov. 14, s311–s319 (1998).
Bergeron, L., Sullivan, N. & Sodroski, J. Target cell-specific determinants of membrane fusion within the Human Immunodeficiency virus type 1 gp120 third variable region and gp41 amino terminus. J. Virol. 66, 2389–2397 (1992).
Whitt, M.A., Zagouras, P., Crise, B. & Rose, J.K. A fusion-defective mutant of the Vesicular Stomatitis Virus glycoprotein. J. Virol. 64, 4907–4913 (1990).
Buseyne, F., Février, M., Garcia, S., Gougeon, M.L. & Rivière, Y. Dual function of a human immunodeficiency virus (HIV)-specific cytotoxic T-lymphocyte clone: inhibition of HIV replication by noncytolytic mechanisms and lysis of HIV-infected CD4+ cells. Virology 225, 248–253 (1996).
Piatak, M., Jr . et al. High levels of HIV-1 in plasma during all stages of infection determined by competitive PCR. Science 259, 1749–1754 (1993).
Embreston, J. et al. Massive covert infection of helper T lymphocytes and macrophages by HIV during the incubation period of AIDS. Nature 362, 359–362 (1993).
Schwartz, O., Marechal, V., Friguet, B., Arenzana-Seisdedos, F. & Heard, J.M. Antiviral activity of the proteasome on incoming HIV-1. J. Virol. 72, 3845–3850 (1998).
Schwartz, O., Maréchal, V., Le Gall, S., Lemonnier, F. & Heard, J.M. Endocytosis of MHC-I molecules is induced by HIV-1 Nef. Nature Med. 2, 338–342 (1996).
Collins, K.L., Chen, B.K., Kalams, S.A., Walker, B.D. & Baltimore, D. HIV-1 Nef protein protects infected primary cells against killing by cytotoxic T lymphocytes. Nature 391, 397–401 (1998).
Pinto, L.A. et al. Env-specific cytotoxic T lymphocytes responses in HIV seronegative health care workers occupationally exposed to HIV-contaminated body fluids. J. Clin. Invest. 96, 867–876 (1995).
Rowland-Jones, S. et al. HIV-specific cytotoxic T-cells in HIV-exposed but uninfected Gambian women. Nature Med. 1, 59–64 (1995).
Rowland-Jones, S., Tan, R. & McMichael, A. Role of cellular immunity in protection against HIV infection. Adv. Immunol. 65, 277–346 (1997).
Brander, C. & Walker, B.D. T lymphocyte responses in HIV-1 infection: implications for vaccine development. Curr. Op. Immunol. 11, 451–459 (1999).
Goxe, B., Latour, N., Bartholeyns, J., Romet-Lemonne, J.L. & Chokri, M. Monocyte-derived dendritic cells: development of a cellular processor for clinical applications. Res. Immunol. 149, 643–646 (1998).
Buseyne, F. et al. Cross-clade-specific cytotoxic T lymphocytes in HIV-1-infected children. Virology 250, 316–324 (1998).
Tsomides, T.J. et al. Naturally processed viral peptides recognized by cytotoxic T lymphocytes on cells chronically infected by Human Immunodeficiency Virus type 1. J. Exp. Med. 180, 1283–1293 (1994).
Czerkinsky, C. et al. Reverse ELISPOT assay for clonal analysis of cytokine production. I. Enumeration of gamma-interferon-secreting cells. J. Immunol. Meth. 110, 29–36 (1988).
Acknowledgements
We thank F. Lemonnier and S. Michelson for critical reading of the manuscript; C. Petit and M.L. Michel for discussions; V. Maréchal for preparation of macrophages; F. Mammano, F. Romagné, F. Latron, M. Takigushi, Chiron and Novartis for gifts of reagents; C. Rouzioux and S. Blanche for their support; and J. Bess Jr for preparation of inactivated HIV-1MN. This work was supported by grants from the Agence Nationale de Recherche sur le SIDA (ANRS), SIDACTION, the Pediatric AIDS Foundation and the Pasteur Institute, and in part with Federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. NO1-CO-56000.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buseyne, F., Gall, S., Boccaccio, C. et al. MHC-I–restricted presentation of HIV-1 virion antigens without viral replication. Nat Med 7, 344–349 (2001). https://doi.org/10.1038/85493
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85493
This article is cited by
-
Dying cells actively regulate adaptive immune responses
Nature Reviews Immunology (2017)
-
The HIV-1 integrase-LEDGF allosteric inhibitor MUT-A: resistance profile, impairment of virus maturation and infectivity but without influence on RNA packaging or virus immunoreactivity
Retrovirology (2017)
-
CD8+T cells from HLA-B*57 elite suppressors effectively suppress replication of HIV-1 escape mutants
Retrovirology (2013)
-
Primary CD8+ T cells from elite suppressors effectively eliminate non-productively HIV-1 infected resting and activated CD4+T cells
Retrovirology (2013)
-
Resolution of de novo HIV production and trafficking in immature dendritic cells
Nature Methods (2008)