-
PDF
- Split View
-
Views
-
Cite
Cite
Arturo Figueroa, Julian A. Trivino, Marcos A. Sanchez-Gonzalez, Florence Vicil, Oral L-Citrulline Supplementation Attenuates Blood Pressure Response to Cold Pressor Test in Young Men, American Journal of Hypertension, Volume 23, Issue 1, January 2010, Pages 12–16, https://doi.org/10.1038/ajh.2009.195
Close - Share Icon Share
Abstract
Oral L-citrulline is efficiently converted to L-arginine, which has been shown to decrease brachial blood pressure (BP) at rest and during the cold pressor test (CPT). However, aortic BP may better reflect cardiovascular risk than brachial BP. The purpose of this study was to test the hypothesis that oral L-citrulline supplementation attenuates brachial BP and aortic hemodynamic responses to CPT.
Brachial BP, aortic BP, stroke volume (SV), and wave reflection at rest and during CPT were evaluated in 17 young (21.6 ± 0.9 years) normotensive men. Subjects were randomly assigned to 4 weeks of oral L-citrulline (6g/day) or placebo in a crossover design. Hemodynamic responses to CPT were reevaluated after each treatment.
During CPT, there were significant (P < 0.05) increases in brachial and aortic BP [systolic (SBP), diastolic (DBP), and pulse pressure (PP)], augmentation index (AIx), SV, and a decrease in transit time of the reflected wave (Tr) from baseline. Compared to placebo, oral L-citrulline treatment decreased (P < 0.05) brachial SBP (−6 ± 11mmHg), aortic SBP (−4 ± 10mmHg), and aortic PP (−3 ± 6mmHg) during CPT but not at rest. There was an inverse correlation (r = −0.40, P < 0.05) between changes in aortic SBP and Tr during CPT after L-citrulline supplementation.
We conclude that oral L-citrulline supplementation attenuates the brachial SBP, aortic SBP, and aortic PP responses to CPT in young normotensive men. Increased wave reflection time contributes to the reduction in aortic SBP response to CPT.
Oral L-arginine, the nitric oxide precursor, can reduce resting blood pressure (BP) via improved nitric oxide production in patients with cardiovascular risk factors.1–3 In healthy individuals, oral L-arginine has been associated with decrease4 or no change5 in resting brachial BP. It has been suggested that an increased arginase activity makes chronic oral L-arginine supplementation somewhat ineffective.6 Conversely, oral L-citrulline provides greater plasma L-arginine levels due to an efficient conversion of L-citrulline to L-arginine.7 Recent data have shown that oral L-citrulline treatment reduced peripheral BP by improving aortic relaxation in rats.8,9
Although elevated brachial pulse pressure (PP) is associated with increased cardiovascular mortality,10 aortic systolic BP (SBP) and PP may be better predictors of cardiovascular risk than brachial BP.11,12 As vasoactive drugs may affect aortic BP more than brachial BP,13,14 aortic BP measurements have been proposed as the treatment target.11,15
Sympathetic-mediated vasoconstriction induced by the cold pressor test (CPT) leads to acute increases in peripheral (brachial and digital) BP, aortic BP, and wave reflection in healthy subjects without increasing the heart rate.16,17 The CPT has been used to evaluate the efficacy of lifestyle3,18 and pharmacological interventions19 on BP and vascular reactivity. In addition, oral L-arginine supplementation was found effective in reducing the brachial BP response to CPT in men with hypercholesterolemia.3 Thus, the CPT may be a suitable tool to evaluate the effect of oral L-citrulline on BP and aortic hemodynamics. Therefore, the purpose of the study was to test the hypothesis that oral L-citrulline supplementation would reduce brachial BP, aortic BP, and wave reflection responses to CPT.
Methods
Subjects. The study population included 17 healthy young (22 ± 1 years) men. Subjects were normotensive (SBP <140mmHg and diastolic BP (DBP) <90mmHg), free of cardiovascular and metabolic/endocrine diseases as evaluated by health history and did not smoke or were taking medications or nutritional supplements. This study was approved by the institutional review board of the Florida State University. All subjects gave written consent before data collection.
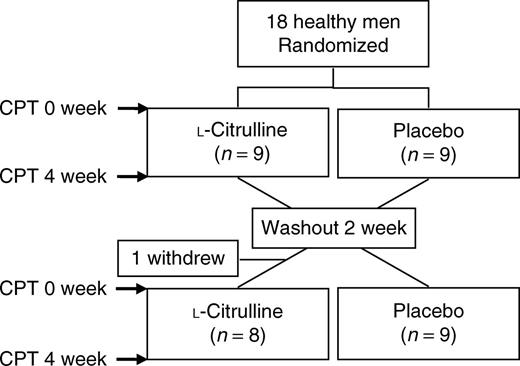
Study design. We used a randomized, double-blind, two-period, crossover design (Figure 1). On the first visit, subjects received a familiarization with the tests after the initial screening process. All tests were performed in the morning with the subjects seated in a quiet temperature-controlled room (23°C) after at least 3-h fast and avoiding caffeinated drinks, alcohol, and intense exercise for at least 24h before testing. After electrocardiogram and BP instrumentation, participants rested for at least 20min before data collection. Brachial BP, electrocardiogram, and aortic hemodynamic measures were collected before (rest) and during the second minute of CPT. The CPT was performed by the immersion of the right foot up to the ankle in cold water (4°C) during 2min. Subjects were encouraged to maintain a constant rate and depth of breathing throughout the experiment, but breathing was not paced.
Study design. CPT, cold pressor test.
After baseline measurements, subjects were randomly (1:1) assigned to receive oral L-citrulline (3-g dose twice a day) or placebo (maltodextrine) for 4 weeks separated by a 2-week washout period. This prescription of oral L-citrulline has shown to be efficacious to increase plasma levels of L-arginine.7 In addition, oral L-arginine at this dose and duration reduces SBP1 at rest and during CPT3 in individuals with high cardiovascular risk. Capsules containing L-citrulline and placebo were provided by NOW Foods (Bloomingdale, IL). The CPT was conducted at the same time of the day (8–12h) for each subject to reduce possible diurnal variations in BP at the beginning and end of each treatment. The last dose of L-citrulline or placebo was consumed the night before the CPT at the end of each treatment.
Anthropometrics. Height was measured using a stadiometer to the nearest 0.5cm and body weight was measured using a seca scale (Sunbeam Products, Boca Raton, FL) to the nearest 0.1kg. Body mass index was calculated as kg/m2.
Hemodynamics. Continuous electrocardiogram and digital BP recordings were obtained from a bipolar lead sampled by a data acquisition system (Biopac, Santa Barbara, CA) and a Finometer device (TNO Biomedical Instrumentation, Amsterdam, the Netherlands), respectively. Heart rate was assessed from the electrocardiogram and stroke volume (SV) from the BP waveform obtained from the Finometer by employing the model flow method.20
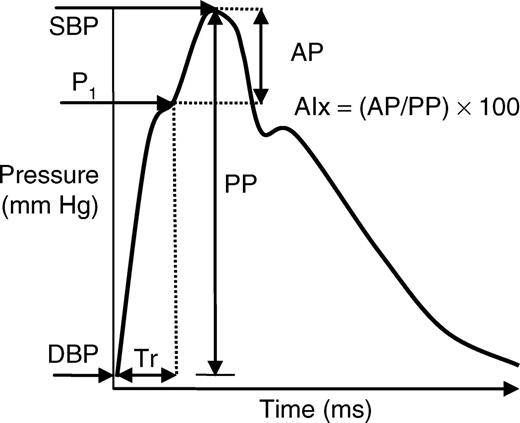
Pulse-wave analysis. After 10min of seated rest, brachial BP was recorded using an automated oscillometric device (HEM-705CP; Omron Healthcare, Vernon Hill, IL). Brachial SBP and DBP were used to calibrate radial waveforms, which were obtained from a 10-s epoch using a high-fidelity tonometer (SPT-301B; Millar Instruments, Houston, TX). PP was the difference between SBP and DBP. Aortic BP waveforms were derived using a generalized validated transfer function (SphygmoCor; AtCor Medical, Sydney, Australia).21,22 The aortic BP wave (Figure 2) is composed of a forward wave, caused by SV ejection, and a reflected wave that returns to the aorta from peripheral sites.23 Augmentation pressure was defined as the difference between the second and first systolic peaks. The augmentation index (AIx) was defined as the augmentation pressure expressed as a percentage of the aortic PP. Transit time of the reflected wave (Tr) indicates the round-trip travel of the forward wave to the peripheral reflecting sites and back to the aorta.23 AIx and Tr have been used as markers of wave reflection and aortic stiffness.24 The average of two measurements of brachial BP and high-quality (operator index ≥80%) aortic hemodynamics was used in the analysis. In our laboratory, the intraclass correlation coefficients for aortic SBP and aortic PP during the CPT, calculated on two separate days, are 0.94 and 0.95, respectively.
Aortic pulse waveform. AIx, augmentation index; AP, augmentation pressure; DBP, diastolic blood pressure; P1, first systolic peak; PP, pulse pressure; SBP, systolic blood pressure; Tr, time delay of reflected wave.
Statistical analysis. The effects of L-citrulline and placebo were evaluated by a 2 × 2 analysis of variance with repeated measures at rest and CPT (treatment (placebo vs. L-citrulline) × time (baseline vs. 4 weeks)). When analysis of variance produced a significant treatment-by-time interaction, t-tests were used for post hoc comparisons. Pearson's correlation coefficient was used to examine associations between changes in variables. Values are shown as means ± s.e.m. Statistical significance was set at P < 0.05. Statistical analyses were performed using SPSS, version 15.0 (SPSS, Chicago, IL).
Results
Height, weight, and body mass index were 1.80 ± 0.01m, 82.8 ± 1.4kg, and 25.7 ± 0.5kg/m2, respectively.
Acute responses to CPT
During CPT, there were significant increases in brachial SBP (P < 0.01), brachial DBP (P < 0.001), brachial PP (P < 0.05), aortic SBP (P < 0.001), aortic DBP (P < 0.001), aortic PP (P < 0.05), AIx (P < 0.01), and SV (P < 0.01) as well as a decrease in Tr (P < 0.001) before and after both treatments (Table 1). The increases in heart rate during CPT were not statistically significant from resting values.
Hemodynamic parameters at rest and during CPT before and after placebo and L-citrulline treatment
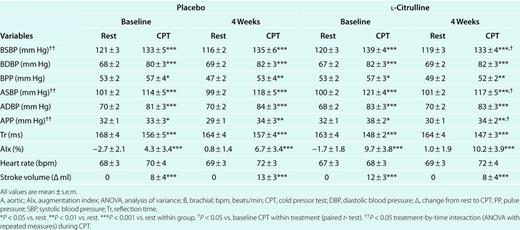
Hemodynamic parameters at rest and during CPT before and after placebo and L-citrulline treatment

Oral L-citrulline supplementation
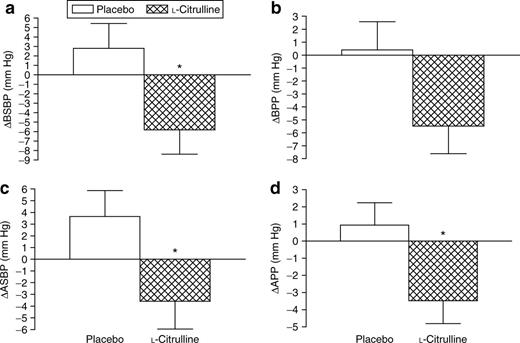
Of the 18 subjects at baseline, 17 completed the two treatments. One subject dropped-out because of lack of interest. There were no significant changes in all resting hemodynamic parameters and in wave reflection indicators during CPT after the treatments (Table 1). There was a significant (P < 0.05) treatment-by-time interaction for brachial SBP, aortic SBP, and aortic PP, but not for brachial PP (Table 1). During CPT, L-citrulline significantly (P < 0.05) decreased brachial SBP (−6 ± 11mmHg), aortic SBP (−4 ± 10mmHg), and aortic PP (−3 ± 6mmHg) from 0 to 4 weeks compared to placebo (brachial SBP (3 ± 10mmHg), aortic SBP (4 ± 9mmHg), and aortic PP (1 ± 5mmHg), P = NS) (Figure 3). There was a significant and inverse correlation (r = −0.40, P < 0.05) between changes in aortic SBP and Tr during the CPT after L-citrulline supplementation.
Changes in (a) brachial systolic blood pressure (BSBP); (b) brachial pulse pressure (BPP); (c) aortic systolic blood pressure (ASBP); (d) aortic pulse pressure (APP) from baseline to 4 weeks during cold pressor test (CPT). *P < 0.05 vs. placebo. Values are means ± s.e.m.
Discussion
Our main finding was that 4 weeks of oral L-citrulline supplementation attenuated the increase in brachial SBP, aortic SBP, and aortic PP during sympathetic stimulation induced by CPT in young men. Consistent with previous studies, CPT resulted in a significant increase in wave reflection, as assessed by increased AIx and reduced Tr. These arterial responses have been shown during cold-induced sympathetic stimulation via CPT17 and whole-body exposure.25 Sympathetic-mediated vasoconstriction induces a faster return of the reflected wave from the periphery to the aorta and its fusion with the forward wave during late systole leads to an increase in aortic SBP and consequently in PP and AIx.23,26 Besides the increase in wave reflection,17 a concomitant increase in SV may have contributed to the SBP and PP response in this study. Moreover, the increase in DBP has been attributed to vasoconstriction of primarily peripheral arteries,27 which leads to a decrease in arterial distensibility.28
As reduced L-arginine availability can contribute to high BP,1–3 we evaluated the effect of the L-arginine-nitric oxide precursor L-citrulline on aortic hemodynamics. It has been shown that most of the L-citrulline absorbed from the intestine is converted to L-arginine,29,30 thereby decreasing peripheral BP in humans1 and rats8,9 with high cardiovascular risk due to improved endothelial function. However, oral L-arginine has no apparent effect on resting BP and vasodilatory capacity in young healthy men.5 Consistent with this observation, L-citrulline supplementation failed to affect resting BP and wave reflection because of a normal arterial function in our population.
The main finding of this study was that oral L-citrulline treatment attenuated the increase in brachial SBP, aortic SBP, and aortic PP during the CPT. In this study, we evaluate the treatment effect on vascular responses to CPT after at least 10h of the last dose of L-citrulline. We have shown that the change in brachial (−5.8mmHg) and aortic SBP (−3.6mmHg) responses to CPT from baseline to 4 weeks was greater with L-citrulline supplementation compared to placebo. West et al.3 reported a significant reduction in brachial SBP (−3.7mmHg) response to CPT that was attributed to reductions in cardiac output. As L-citrulline did not decrease heart rate and SV in this study, the effect on SBP most likely resulted from decreased wave reflection. Despite no apparent effect on AIx and Tr after L-citrulline, the decrease in aortic SBP during CPT was inversely correlated (r = −0.40, P < 0.05) with the increase in wave reflection time but not with AIx. Similarly, during sympathetic overactivity induced by orthostatic stress, the infusion of L-arginine results in decreased aortic SBP due to increased wave reflection time.31 Because a reduction in SV does not occur concurrently with the increase in wave reflection time, the AIx may not change.31 Thus a slower wave reflection from peripheral arteries to the aorta leads to a reduction in aortic SBP.32
In this study, L-citrulline reduced aortic PP, but had no apparent effect on brachial PP response to CPT. This result confirms the notion that the reduction in aortic SBP and PP with some antihypertensive drugs is underestimated or not detected by brachial BP.23,33 Our results showed that brachial and aortic DBP did not change at the end of the study, suggesting that sympathetic-mediated vasoconstriction of peripheral arteries during CPT27 is not modified by L-citrulline. As PP is influenced more by SBP than by DBP, the decrease in the aortic PP response to CPT can be mainly attributed to an attenuated aortic SBP.
Potential limitations of this study include a small sample size and the lack of measurements of plasma L-arginine and endothelial function. It could be that our study was not powered to detect significant changes in wave reflection. We cannot provide a mechanism to explain our results without L-arginine and endothelial function measures. Nevertheless, this study demonstrated that 6g daily dosage of oral L-citrulline reduced aortic SBP and PP responses to CPT—two major predictors of cardiovascular disease.11,12 More importantly, in contrast to previous studies that have evaluated the effect of L-arginine on brachial BP between 1 and 2h after the last dose,3,5 the effect of L-citrulline on brachial and aortic BP was observed several hours after the last dose in this study.
In conclusion, we found that a 4-week oral L-citrulline supplementation attenuated the brachial SBP, aortic SBP, and aortic PP responses to CPT. Increased aortic wave reflection time contributes to the reduced aortic SBP response to CPT, which consequently contributes to the reduction in aortic PP.
Disclosure
The authors declared no conflict of interest.
Acknowledgements
This study was supported by College of Human Sciences Research Award and Bess Ward Honors Thesis Award. We are grateful to NOW foods for providing the supplements.
References
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.