Abstract
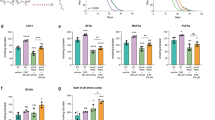
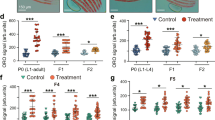
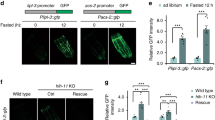
Regulation of body fat storage involves signalling between centres that regulate feeding in the brain and sites of fat storage and use in the body1,2. Here we describe an assay for analysing fat storage and mobilization in living Caenorhabditis elegans. By using RNA-mediated interference (RNAi)3,4 to disrupt the expression of each of the 16,757 worm genes, we have systematically screened the C. elegans genome for genes necessary for normal fat storage. We identify 305 gene inactivations that cause reduced body fat and 112 gene inactivations that cause increased fat storage. Analysis of the fat-reducing gene inactivations in insulin, serotonin and tubby signalling mutants of C. elegans, which have increased body fat, identifies a core set of fat regulatory genes as well as pathway-specific fat regulators. Many of the newly identified worm fat regulatory genes have mammalian homologues, some of which are known to function in fat regulation. Other C. elegans fat regulatory genes that are conserved across animal phylogeny, but have not previously been implicated in fat storage, may point to ancient and universal features of fat storage regulation, and identify targets for treating obesity and its associated diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Schwartz, M. W., Woods, S. C., Porte, D. Jr, Seeley, R. J. & Baskin, D. G. Central nervous system control of food intake. Nature 404, 661–671 (2000)
Friedman, J. M. & Halaas, J. L. Leptin and the regulation of body weight in mammals. Nature 395, 763–770 (1998)
Fire, A. et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391, 806–811 (1998)
Kamath, R. S. et al. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421, 231–237 (2003)
Greenspan, P. & Fowler, S. D. Spectrofluorometric studies of the lipid probe, Nile Red. J. Lipid Res. 26, 781–789 (1985)
Kimura, K. D., Tissenbaum, H. A., Liu, Y. & Ruvkun, G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science 277, 942–946 (1997)
Riddle, D. L. & Albert, P. S. C. elegans II (eds Riddle, D. L., Blumenthal, T., Meyer, B. J. & Priess, J. R.) 739–768 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 1997)
Wadsworth, W. G. & Riddle, D. L. Developmental regulation of energy metabolism in Caenorhabditis elegans. Dev. Biol. 132, 167–173 (1989)
Ogg, S. et al. The Fork head transcription factor DAF-16 transduces insulin-like metabolic and longevity signals in C. elegans. Nature 389, 994–999 (1997)
Lin, K. et al. An HNF-3/forkhead family member that can function to double the life-span of Caenorhabditis elegans. Science 278, 1319–1322 (1997)
Ogg, S. & Ruvkun, G. The C. elegans PTEN homolog, DAF-18, acts in the insulin receptor-like metabolic signaling pathway. Mol. Cell 2, 887–893 (1998)
Tecott, L. H. et al. Eating disorder and epilepsy in mice lacking 5-HT2c serotonin receptors. Nature 374, 542–546 (1995)
Noben-Trauth, K., Naggert, J. K., North, M. A. & Nishina, P. M. A candidate gene for the mouse mutation tubby. Nature 380, 534–538 (1996)
Sze, J. Y., Victor, M., Loer, C., Shi, Y. & Ruvkun, G. Food and metabolic signalling defects in a Caenorhabditis elegans serotonin-synthesis mutant. Nature 403, 560–564 (2000)
Liu, L. X. et al. High-throughput isolation of Caenorhabditis elegans deletion mutants. Genome Res. 9, 859–867 (1999)
Kamath, R. S., Martinez-Campos, M., Zipperlen, P., Fraser, A. G. & Ahringer, J. Effectiveness of specific RNA-mediated interference through ingested double-stranded RNA in Caenorhabditis elegans. Genome Biol. 2, 1–10 (2000)
Brown, M. S. & Goldstein, J. L. A proteolytic pathway that controls the cholesterol content of membranes, cells, and blood. Proc. Natl Acad. Sci. USA 96, 11041–11048 (1999)
Rolland, V. et al. Evidence of increased glyceraldehyde-3-phosphate dehydrogenase and fatty acid synthetase promoter activities in transiently transfected adipocytes from genetically obese rats. J. Biol. Chem. 270, 1102–1106 (1995)
Olswang, Y. et al. A mutation in the peroxisome proliferator-activated receptor γ binding site in the gene for the cytosolic form of phosphoenolpyruvate carboxykinase reduces adipose tissue size and fat content in mice. Proc. Natl Acad. Sci. USA 99, 625–630 (2002)
Adibi, S. A. The oligopeptide transporter (Pept-1) in human intestine: biology and function. Gastroenterology 113, 332–340 (1997)
Chawla, A., Repa, J. J., Evans, R. M. & Mangelsdorf, D. J. Nuclear receptors and lipid physiology: opening the X-files. Science 294, 1866–1870 (2001)
Yamagata, K. et al. Mutations in the hepatocyte nuclear factor-4α gene in maturity-onset diabetes of the young (MODY1). Nature 384, 458–460 (1996)
Good, D. J. et al. Hypogonadism and obesity in mice with a targeted deletion of the Nhlh2 gene. Nature Genet 15, 397–401 (1997)
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402, 804–809 (1999)
Wolkow, C. A., Kimura, K. D., Lee, M. S. & Ruvkun, G. Regulation of C. elegans life-span by insulin-like signaling in the nervous system. Science 290, 147–150 (2000)
Brockmann, G. A. & Bevoval, M. R. Using mouse models to dissect the genetics of obesity. Trends Genet. 18, 367–376 (2002)
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974)
Johnstone, I. L. C. elegans: A Practical Approach (ed. Hope, I. A.) 201–225 (Oxford Univ. Press, Oxford, 1999)
Watts, J. L. & Browse, J. Genetic dissection of polyunsaturated fatty acid synthesis in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 99, 5854–5859 (2002)
Acknowledgements
We thank M. Basson and C. Johnson for the tub-1(nr2004) strain. K.A is grateful to A. Pasquinelli and other members of the Ruvkun laboratory for discussions and suggestions. K.A. was supported by a fellowship from the Damon-Runyon Walter-Winchell Cancer Research Fund; J.W. by a NIH grant; A.G.F. by a US Army Breast Cancer Research Fellowship; R.S.K. by a Howard Hughes Medical Institute Predoctoral Fellowship; and J.A. by a Wellcome Trust Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Ashrafi, K., Chang, F., Watts, J. et al. Genome-wide RNAi analysis of Caenorhabditis elegans fat regulatory genes. Nature 421, 268–272 (2003). https://doi.org/10.1038/nature01279
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01279
This article is cited by
-
Image segmentation and separation of spectrally similar dyes in fluorescence microscopy by dynamic mode decomposition of photobleaching kinetics
BMC Bioinformatics (2022)
-
Caenorhabditis elegans as a powerful tool in natural product bioactivity research
Applied Biological Chemistry (2022)
-
Neuropeptidergic regulation of compulsive ethanol seeking in C. elegans
Scientific Reports (2022)
-
Intracellular lipid surveillance by small G protein geranylgeranylation
Nature (2022)
-
Identification of healthspan-promoting genes in Caenorhabditis elegans based on a human GWAS study
Biogerontology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.