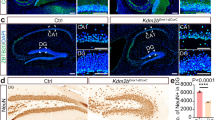
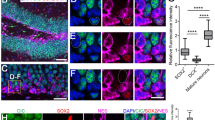
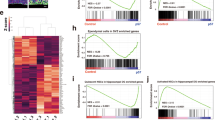
Abstract
The generation of new neurons from neural stem cells is restricted to two regions of the adult mammalian central nervous system: the subventricular zone of the lateral ventricle, and the subgranular zone of the hippocampal dentate gyrus1. In both regions, signals provided by the microenvironment regulate the maintenance, proliferation and neuronal fate commitment of the local stem cell population1. The identity of these signals is largely unknown. Here we show that adult hippocampal stem/progenitor cells (AHPs) express receptors and signalling components for Wnt proteins, which are key regulators of neural stem cell behaviour in embryonic development2. We also show that the Wnt/β-catenin pathway is active and that Wnt3 is expressed in the hippocampal neurogenic niche. Overexpression of Wnt3 is sufficient to increase neurogenesis from AHPs in vitro and in vivo. By contrast, blockade of Wnt signalling reduces neurogenesis from AHPs in vitro and abolishes neurogenesis almost completely in vivo. Our data show that Wnt signalling is a principal regulator of adult hippocampal neurogenesis and provide evidence that Wnt proteins have a role in adult hippocampal function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Alvarez-Buylla, A. & Lim, D. A. For the long run: maintaining germinal niches in the adult brain. Neuron 41, 683–686 (2004)
Kleber, M. & Sommer, L. Wnt signalling and the regulation of stem cell function. Curr. Opin. Cell Biol. 16, 681–687 (2004)
Maretto, S. et al. Mapping Wnt/β-catenin signalling during mouse development and in colorectal tumors. Proc. Natl Acad. Sci. USA 100, 3299–3304 (2003)
Song, H., Stevens, C. F. & Gage, F. H. Astroglia induce neurogenesis from adult neural stem cells. Nature 417, 39–44 (2002)
Moon, R. T. Canonical Wnt/β-catenin Signaling. Sci. STKE 2004, TR5 (2004)
Rattner, A. et al. A family of secreted proteins contains homology to the cysteine-rich ligand-binding domain of frizzled receptors. Proc. Natl Acad. Sci. USA 94, 2859–2863 (1997)
Couillard-Despres, S. et al. Doublecortin expression levels in adult brain reflect neurogenesis. Eur. J. Neurosci. 21, 1–14 (2005)
Veeman, M. T., Axelrod, J. D. & Moon, R. T. A second canon. Functions and mechanisms of β-catenin-independent Wnt signalling. Dev. Cell 5, 367–377 (2003)
Veeman, M. T., Slusarski, D. C., Kaykas, A., Louie, S. H. & Moon, R. T. Zebrafish prickle, a modulator of noncanonical Wnt/Fz signalling, regulates gastrulation movements. Curr. Biol. 13, 680–685 (2003)
DasGupta, R. & Fuchs, E. Multiple roles for activated LEF/TCF transcription complexes during hair follicle development and differentiation. Development 126, 4557–4568 (1999)
Sakurada, K., Ohshima-Sakurada, M., Palmer, T. D. & Gage, F. H. Nurr1, an orphan nuclear receptor, is a transcriptional activator of endogenous tyrosine hydroxylase in neural progenitor cells derived from the adult brain. Development 126, 4017–4026 (1999)
McBeath, R., Pirone, D. M., Nelson, C. M., Bhadriraju, K. & Chen, C. S. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev. Cell 6, 483–495 (2004)
Schwartz, K. A., Lanciloti, N. J., Moore, M. K., Campione, A. L. & Chandar, N. p53 transactivity during in vitro osteoblast differentiation in a rat osteosarcoma cell line. Mol. Carcinog. 25, 132–138 (1999)
Consiglio, A. et al. Robust in vivo gene transfer into adult mammalian neural stem cells by lentiviral vectors. Proc. Natl Acad. Sci. USA 101, 14835–14840 (2004)
Garcia-Castro, M. I., Marcelle, C. & Bronner-Fraser, M. Ectodermal Wnt function as a neural crest inducer. Science 297, 848–851 (2002)
Hoppler, S., Brown, J. D. & Moon, R. T. Expression of a dominant-negative Wnt blocks induction of MyoD in Xenopus embryos. Genes Dev. 10, 2805–2817 (1996)
Israsena, N., Hu, M., Fu, W., Kan, L. & Kessler, J. A. The presence of FGF2 signalling determines whether β-catenin exerts effects on proliferation or neuronal differentiation of neural stem cells. Dev. Biol. 268, 220–231 (2004)
Jamora, C., DasGupta, R., Kocieniewski, P. & Fuchs, E. Links between signal transduction, transcription and adhesion in epithelial bud development. Nature 422, 317–322 (2003)
Duncan, A. W. et al. Integration of Notch and Wnt signalling in hematopoietic stem cell maintenance. Nature Immunol. 6, 314–322 (2005)
Kleber, M. et al. Neural crest stem cell maintenance by combinatorial Wnt and BMP signalling. J. Cell Biol. 169, 309–320 (2005)
Lai, K., Kaspar, B. K., Gage, F. H. & Schaffer, D. V. Sonic hedgehog regulates adult neural progenitor proliferation in vitro and in vivo. Nature Neurosci. 6, 21–27 (2003)
Lee, J., Duan, W. & Mattson, M. P. Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J. Neurochem. 82, 1367–1375 (2002)
Cao, L. et al. VEGF links hippocampal activity with neurogenesis, learning and memory. Nature Genet. 36, 827–835 (2004)
Palmer, T. D., Takahashi, J. & Gage, F. H. The adult rat hippocampus contains primordial neural stem cells. Mol. Cell. Neurosci. 8, 389–404 (1997)
Kitamura, T. et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp. Hematol. 31, 1007–1014 (2003)
van Praag, H., Kempermann, G. & Gage, F. H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nature Neurosci. 2, 266–270 (1999)
Lein, E. S., Zhao, X. & Gage, F. H. Defining a molecular atlas of the hippocampus using DNA microarrays and high-throughput in situ hybridization. J. Neurosci. 24, 3879–3889 (2004)
Acknowledgements
We thank H. Clevers, R. Nusse, R.T. Moon, E. Fuchs, R. Marr and J. Nathans for cDNA and plasmids; S. Piccolo and B. Sosa-Pineda for BATGAL reporter mice; R. Summers, K. Nakashima, T. Kuwabara and H. Suh for discussions and suggestions; M.L. Gage for editorial comments; and L. Kitabayashi for technical assistance. D.C.L. and S.J. are supported in part by the Deutsche Forschungsgemeinschaft (DFG, Germany), S.A.C. was supported by NRSA, L.D. was supported by the Association for Medical Research (FRM, France), A.C. is a recipient of a Telethon postdoctoral fellowship. Additional support was provided by grants from the National Institute of Neurological Disorders and Stroke (NINDS) and National Institute on Aging (NIA), the Max Planck Research Award Program funded by the German Ministry for Education, Science, Research, and Technology, the Lookout Fund, the Christopher Reeves Paralysis Foundation, Defense Advanced Research Projects Agency and Project ALS (to F.H.G.), and from the NINDS and NIA (to H.J.S.). Author Contributions D.C.L. is the leading author. He contributed to the concept, designed and did the experiments, analysed the data, wrote the paper and provided partial financial support. S.A.C. contributed to the concept, designed and did the in vivo loss-of-function experiment, contributed reagents and revised the paper. H.J.S. contributed to the concept and the co-culture experiments. L.D. initiated the study, contributed to the concept and did part of the RT–PCR analysis. H.M. contributed to the co-culture experiments. A.C. generated the LV vectors. E.S.L. did the in situ hybridization. S.J. analysed the BATGAL mice. H.L. and A.R.D. provided technical support and contributed to analysis of the data. F.H.G. is the senior author. He contributed to the concept, analysed the data, revised the manuscript and provided the main financial support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure Legends
Legends to accompany the Supplementary Figures. (DOC 28 kb)
Supplementary Figure 1
Wnt proteins expressed by AHPs are not sufficient to elicit significant Wnt/β-catenin signalling in AHPs. (PDF 267 kb)
Supplementary Figure 2
Stimulation of Wnt/β-catenin signalling in AHPs by Wnt3. (PDF 176 kb)
Supplementary Figure 3
Wnt3 overexpression does not enhance the generation of glial cells. (PDF 199 kb)
Supplementary Figure 4
Time course of differentiation of AHPs into doublecortin-expressing cells. (PDF 169 kb)
Supplementary Figure 5
Suppression of Wnt//β-catenin signalling in AHPs by dnWnt. (PDF 168 kb)
Supplementary Figure 6
DnWnt overexpression does not increase cell death in vitro or in vivo. (PDF 198 kb)
Supplementary Methods
Detailed, additional description of methods. (DOC 26 kb)
Rights and permissions
About this article
Cite this article
Lie, DC., Colamarino, S., Song, HJ. et al. Wnt signalling regulates adult hippocampal neurogenesis. Nature 437, 1370–1375 (2005). https://doi.org/10.1038/nature04108
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04108
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.