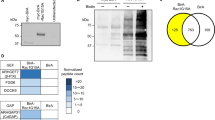
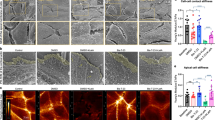
Abstract
The Rho family of small GTPases (RhoA, Rac1 and Cdc42) controls signal-transduction pathways that influence many aspects of cell behaviour, including cytoskeletal dynamics1,2,3. At the leading edge, Rac1 and Cdc42 promote cell motility through the formation of lamellipodia and filopodia, respectively. On the contrary, RhoA promotes the formation of contractile actin–myosin-containing stress fibres in the cell body and at the rear1,2,4. Here, we identify synaptopodin, an actin-associated protein, as a novel regulator of RhoA signalling and cell migration in kidney podocytes. We show that synaptopodin induces stress fibres by competitive blocking of Smurf1-mediated ubiquitination of RhoA, thereby preventing the targeting of RhoA for proteasomal degradation. Gene silencing of synaptopodin in kidney podocytes causes the loss of stress fibres and the formation of aberrant non-polarized filopodia and impairment of cell migration. Together, these data show that synaptopodin is essential for the integrity of the podocyte actin cytoskeleton and for the regulation of podocyte cell migration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Etienne-Manneville, S. & Hall, A. Rho GTPases in cell biology. Nature 420, 629–635 (2002).
Raftopoulou, M. & Hall, A. Cell migration: Rho GTPases lead the way. Dev. Biol. 265, 23–32 (2004).
Jaffe, A. B. & Hall, A. RHO GTPases: Biochemistry and Biology. Annu. Rev. Cell. Dev. Biol. 21, 247–269 (2005).
Jaffe, A. B. & Hall, A. Cell biology. Smurfing at the leading edge. Science 302, 1690–1691 (2003).
Mundel, P. et al. Synaptopodin: an actin-associated protein in telencephalic dendrites and renal podocytes. J. Cell Biol. 139, 193–204 (1997).
Asanuma, K. et al. Synaptopodin regulates the actin-bundling activity of α-actinin in an isoform-specific manner. J. Clin. Invest. 115, 1188–1198 (2005).
Deller, T. et al. Synaptopodin-deficient mice lack a spine apparatus and show deficits in synaptic plasticity. Proc. Natl Acad. Sci. USA 100, 10494–10499 (2003).
Mundel, P. et al. Rearrangements of the cytoskeleton and cell contacts induce process formation during differentiation of conditionally immortalized mouse podocyte cell lines. Exp. Cell Res. 236, 248–258 (1997).
Wang, H. R. et al. Regulation of cell polarity and protrusion formation by targeting RhoA for degradation. Science 302, 1775–1779 (2003).
Ozdamar, B. et al. Regulation of the polarity protein Par6 by TGFβ receptors controls epithelial cell plasticity. Science 307, 1603–1609 (2005).
Faul, C. et al. Promotion of importinα-mediated nuclear import by the phosphorylation-dependent binding of cargo protein to 14-3-3. J. Cell Biol. 169, 415–424 (2005).
Mundel, P. & Kriz, W. Structure and function of podocytes: an update. Anat. Embryol. 192, 385–397 (1995).
Somlo, S. & Mundel, P. Getting a foothold in nephrotic syndrome. Nature Genet. 24, 333–335 (2000).
Drenckhahn, D. & Franke, R. P. Ultrastructural organization of contractile and cytoskeletal proteins in glomerular podocytes of chicken, rat, and man. Lab. Invest. 59, 673–682 (1988).
Kaplan, J. M. et al. Mutations in ACTN4, encoding α-actinin-4, cause familial focal segmental glomerulosclerosis. Nature Genet. 24, 251–256 (2000).
Kavsak, P. et al. Smad7 binds to Smurf2 to form an E3 ubiquitin ligase that targets the TGFβ receptor for degradation. Mol. Cell 6, 1365–1375 (2000).
Acknowledgements
We thank S. Ratner and M. Donnelly for excellent technical assistance. We thank J. Wrana (University of Toronto, Toronto, Canada) for RhoAK6,7R cDNA, F. Gertler (MIT, Cambridge, MA) for anti-Mena antibodies, T. Takenawa (University of Tokyo, Tokyo, Japan) for GST–RhoAT19N cDNA, T. Imamura (The JFCR Cancer Institute, Tokyo, Japan) for Smurf1 (wild-type and C710A) cDNA, K. Tanaka (Tokyo Metropolitan Institute of Medical Science, Tokyo, Japan) for ubiquitin cDNA and M. Pollak (Brigham and Women's Hospital and Harvard Medical School, Boston, MA) for α-actinin-4 antibody. It is a pleasure to acknowledge helpful discussions with H. Yamaguchi (Albert Einstein College of Medicine, Bronx, New York, NY). K. Asanuma was supported by the National Kidney Foundation and the Alumini Association of Juntendo University (Juntendo University, Tokyo, Japan), and K. Kim by the Kidney and Urology Foundation of America. This work was supported by National Institutes of Health grants (DA18886, DK57683), and the George M. O'Brien Kidney Center (DK064236) to P.M.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1 and S2 (PDF 191 kb)
Rights and permissions
About this article
Cite this article
Asanuma, K., Yanagida-Asanuma, E., Faul, C. et al. Synaptopodin orchestrates actin organization and cell motility via regulation of RhoA signalling. Nat Cell Biol 8, 485–491 (2006). https://doi.org/10.1038/ncb1400
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1400
This article is cited by
-
The effect of mycophenolate mofetil on podocytes in nephrotoxic serum nephritis
Scientific Reports (2023)
-
Mycophenolic acid directly protects podocytes by preserving the actin cytoskeleton and increasing cell survival
Scientific Reports (2023)
-
SMURF1, a promoter of tumor cell progression?
Cancer Gene Therapy (2021)
-
Neurological involvement in monogenic podocytopathies
Pediatric Nephrology (2021)
-
Protective effects of DPP-4 inhibitor on podocyte injury in glomerular diseases
BMC Nephrology (2020)