Abstract
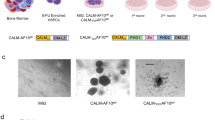
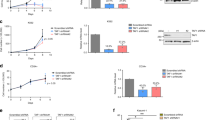
Chromosomal translocation is a common cause of leukaemia1 and the most common chromosome translocations found in leukaemia patients involve the mixed lineage leukaemia (MLL) gene2,3. AF10 is one of more than 30 MLL fusion partners in leukaemia4. We have recently demonstrated that the H3K79 methyltransferase hDOT1L contributes to MLL–AF10-mediated leukaemogenesis through its interaction with AF10 (ref. 5). In addition to MLL, AF10 has also been reported to fuse to CALM (clathrin-assembly protein-like lymphoid–myeloid) in patients with T-cell acute lymphoblastic leukaemia (T-ALL) and acute myeloid leukaemia (AML)6,7. Here, we analysed the molecular mechanism of leukaemogenesis by CALM–AF10. We demonstrate that CALM–AF10 fusion is both necessary and sufficient for leukaemic transformation. Additionally, we provide evidence that hDOT1L has an important role in the transformation process. hDOT1L contributes to CALM–AF10-mediated leukaemic transformation by preventing nuclear export of CALM–AF10 and by upregulating the Hoxa5 gene through H3K79 methylation. Thus, our study establishes CALM–AF10 fusion as a cause of leukaemia and reveals that mistargeting of hDOT1L and upregulation of Hoxa5 through H3K79 methylation is the underlying mechanism behind leukaemia caused by CALM–AF10 fusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Greaves, M. F. & Wiemels, J. Origins of chromosome translocations in childhood leukaemia. Nature Rev. Cancer 3, 639–649 (2003).
Daser, A. & Rabbitts, T. H. Extending the repertoire of the mixed-lineage leukemia gene MLL in leukemogenesis. Genes Dev. 18, 965–974 (2004).
Ayton, P. M. & Cleary, M. L. Molecular mechanisms of leukemogenesis mediated by MLL fusion proteins. Oncogene 20, 5695–5707 (2001).
Chaplin, T. et al. The t(10;11) translocation in acute myeloid leukemia (M5) consistently fuses the leucine zipper motif of AF10 onto the HRX gene. Blood 86, 2073–2076 (1995).
Okada, Y. et al. hDOT1L links histone methylation to leukemogenesis. Cell 121, 167–178 (2005).
Carlson, K. M. et al. Identification and molecular characterization of CALM/AF10fusion products in T cell acute lymphoblastic leukemia and acute myeloid leukemia. Leukemia 14, 100–104 (2000).
Dreyling, M. H. et al. MLL and CALM are fused to AF10 in morphologically distinct subsets of acute leukemia with translocation t(10;11): both rearrangements are associated with a poor prognosis. Blood 91, 4662–4667 (1998).
Feng, Q. et al. Methylation of H3-lysine 79 is mediated by a new family of HMTases without a SET domain. Curr. Biol. 12, 1052–1058 (2002).
DiMartino, J. F. et al. The AF10 leucine zipper is required for leukemic transformation of myeloid progenitors by MLL–AF10. Blood 99, 3780–3785 (2002).
Dreyling, M. H. et al. The t(10;11)(p13;q14) in the U937 cell line results in the fusion of the AF10 gene and CALM, encoding a new member of the AP-3 clathrin assembly protein family. Proc. Natl Acad. Sci. USA 93, 4804–4809 (1996).
Kobayashi, H., Thirman, M. J. & Rowley, J. D. U937 cell line has a t(10;11)(p13-14;q14-21) rather than a deletion of 11q. Genes Chromosomes Cancer 13, 217–218 (1995).
Dik, W. A. et al. CALM-AF10+ T-ALL expression profiles are characterized by overexpression of HOXA and BMI1 oncogenes. Leukemia 19, 1948–1957 (2005).
Drabkin, H. A. et al. Quantitative HOX expression in chromosomally defined subsets of acute myelogenous leukemia. Leukemia 16, 186–195 (2002).
Soulier, J. et al. HOXA genes are included in genetic and biologic networks defining human acute T-cell leukemia (T-ALL). Blood 106, 274–286 (2005).
Ayton, P. M. & Cleary, M. L. Transformation of myeloid progenitors by MLL oncoproteins is dependent on Hoxa7 and Hoxa9. Genes Dev. 17, 2298–2307 (2003).
Zeisig, B. B. et al. Hoxa9 and Meis1 are key targets for MLL-ENL-mediated cellular immortalization. Mol. Cell Biol. 24, 617–628 (2004).
Meyerholz, A. et al. Effect of clathrin assembly lymphoid myeloid leukemia protein depletion on clathrin coat formation. Traffic 6, 1225–1234 (2005).
Tebar, F., Bohlander, S. K. & Sorkin, A. Clathrin assembly lymphoid myeloid leukemia (CALM) protein: localization in endocytic-coated pits, interactions with clathrin, and the impact of overexpression on clathrin-mediated traffic. Mol. Biol. Cell 10, 2687–2702 (1999).
Vecchi, M. et al. Nucleocytoplasmic shuttling of endocytic proteins. J. Cell Biol. 153, 1511–1517 (2001).
So, C. W., Karsunky, H., Wong, P., Weissman, I. L. & Cleary, M. L. Leukemic transformation of hematopoietic progenitors by MLL-GAS7 in the absence of Hoxa7 or Hoxa9. Blood 103, 3192–3199 (2004).
Kobayashi, H. et al. Hematologic malignancies with the t(10;11) (p13;q21) have the same molecular event and a variety of morphologic or immunologic phenotypes. Genes Chromosomes Cancer 20, 253–259 (1997).
Crooks, G. M. et al. Constitutive HOXA5 expression inhibits erythropoiesis and increases myelopoiesis from human hematopoietic progenitors. Blood 94, 519–528 (1999).
Quentmeier, H. et al. Expression of HOX genes in acute leukemia cell lines with and without MLL translocations. Leuk. Lymphoma 45, 567–574 (2004).
Jeannotte, L., Lemieux, M., Charron, J., Poirier, F. & Robertson, E. J. Specification of axial identity in the mouse: role of the Hoxa-5 (Hox1.3) gene. Genes Dev. 7, 2085–2096 (1993).
Acknowledgements
We thank J.A. Frelinger for providing NOD–SCID mice and E. Kallin for critical reading of the manuscript. This work was supported by grants from the National Institutes of Health (NIH) to Y.Z. (GM68804) and L.S. (AI48407, HL72240). Y.O. is a research fellow of Japan Society for the Promotion of Science. L.J. holds a Chercheur National Award from the Fonds de la recherche en Sante du Quebec. Y.Z. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2 and S3 (PDF 512 kb)
Rights and permissions
About this article
Cite this article
Okada, Y., Jiang, Q., Lemieux, M. et al. Leukaemic transformation by CALM–AF10 involves upregulation of Hoxa5 by hDOT1L. Nat Cell Biol 8, 1017–1024 (2006). https://doi.org/10.1038/ncb1464
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1464
This article is cited by
-
Clinico-biological features of T-cell acute lymphoblastic leukemia with fusion proteins
Blood Cancer Journal (2022)
-
Therapeutic strategies against hDOT1L as a potential drug target in MLL-rearranged leukemias
Clinical Epigenetics (2020)
-
Epigenetic priming by Dot1l in lymphatic endothelial progenitors ensures normal lymphatic development and function
Cell Death & Disease (2020)
-
Epigenetic modifications of histones in cancer
Genome Biology (2019)
-
Targeting epigenetic regulators for cancer therapy: mechanisms and advances in clinical trials
Signal Transduction and Targeted Therapy (2019)