Abstract
By rapidly depleting each of the essential budding yeast proteins of unknown function, we identified a novel factor that we call Inn1, which associates with the contractile actomyosin ring at the end of mitosis and is needed for cytokinesis. We show that Inn1 has a C2 domain at the amino terminus of the protein that is required for ingression of the plasma membrane, whereas the remainder of the protein recruits Inn1 to the actomyosin ring. The lethal effects of deleting the INN1 gene can be suppressed by artificial fusion of the C2 domain to other components of the actomyosin ring, restoring membrane ingression on contraction of the actomyosin ring. Our data indicate that recruitment of the C2 domain of Inn1 to the contractile actomyosin ring is crucial for ingression of the plasma membrane during cytokinesis in budding yeast.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Albertson, R., Riggs, B. & Sullivan, W. Membrane traffic: a driving force in cytokinesis. Trends Cell Biol. 15, 92–101 (2005).
Strickland, L. I. & Burgess, D. R. Pathways for membrane trafficking during cytokinesis. Trends Cell Biol. 14, 115–118 (2004).
Burgess, D. R. & Chang, F. Site selection for the cleavage furrow at cytokinesis. Trends Cell Biol. 15, 156–162 (2005).
D'Avino, P. P., Savoian, M. S. & Glover, D. M. Cleavage furrow formation and ingression during animal cytokinesis: a microtubule legacy. J. Cell Sci. 118, 1549–1558 (2005).
Tolliday, N., Bouquin, N. & Li, R. Assembly and regulation of the cytokinetic apparatus in budding yeast. Curr. Opin. Microbiol. 4, 690–695 (2001).
Balasubramanian, M. K., Bi, E. & Glotzer, M. Comparative analysis of cytokinesis in budding yeast, fission yeast and animal cells. Curr. Biol. 14, R806–R818 (2004).
Glotzer, M. The molecular requirements for cytokinesis. Science 307, 1735–1739 (2005).
Barr, F. A. & Gruneberg, U. Cytokinesis: placing and making the final cut. Cell 131, 847–860 (2007).
Giaever, G. et al. Functional profiling of the Saccharomyces cerevisiae genome. Nature 418, 387–391 (2002).
Winzeler, E. A. et al. Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285, 901–906 (1999).
Kanemaki, M., Sanchez-Diaz, A., Gambus, A. & Labib, K. Functional proteomic identification of DNA replication proteins by induced proteolysis in vivo. Nature 423, 720–725 (2003).
Dohmen, R. J., Wu, P. & Varshavsky, A. Heat-inducible degron: a method for constructing temperature-sensitive mutants. Science 263, 1273–1276 (1994).
Labib, K., Tercero, J. A. & Diffley, J. F. X. Uninterrupted MCM2-7 function required for DNA replication fork progression. Science 288, 1643–1647 (2000).
Cheeseman, I. M. et al. Implication of a novel multiprotein Dam1p complex in outer kinetochore function. J. Cell Biol. 155, 1137–1145 (2001).
Enquist-Newman, M. et al. Dad1p, third component of the Duo1p/Dam1p complex involved in kinetochore function and mitotic spindle integrity. Mol. Biol. Cell 12, 2601–2613 (2001).
Euskirchen, G. M. Nnf1p, Dsn1p, Mtw1p, and Nsl1p: a new group of proteins important for chromosome segregation in Saccharomyces cerevisiae. Eukaryot. Cell 1, 229–240 (2002).
Janke, C., Ortiz, J., Tanaka, T. U., Lechner, J. & Schiebel, E. Four new subunits of the Dam1–Duo1 complex reveal novel functions in sister kinetochore biorientation. EMBO J. 21, 181–193 (2002).
Li, Y. et al. The mitotic spindle is required for loading of the DASH complex onto the kinetochore. Genes Dev. 16, 183–197 (2002).
Pinsky, B. A., Tatsutani, S. Y., Collins, K. A. & Biggins, S. An Mtw1 complex promotes kinetochore biorientation that is monitored by the Ipl1/Aurora protein kinase. Dev. Cell 5, 735–745 (2003).
Scharfenberger, M. et al. Nsl1p is essential for the establishment of bipolarity and the localization of the Dam–Duo complex. EMBO J. 22, 6584–6597 (2003).
Lippincott, J. & Li, R. Dual function of Cyk2, a cdc15/PSTPIP family protein, in regulating actomyosin ring dynamics and septin distribution. J. Cell Biol. 143, 1947–1960 (1998).
Vallen, E. A., Caviston, J. & Bi, E. Roles of Hof1p, Bni1p, Bnr1p, and myo1p in cytokinesis in Saccharomyces cerevisiae. Mol. Biol. Cell 11, 593–611 (2000).
Dobbelaere, J. & Barral, Y. Spatial coordination of cytokinetic events by compartmentalization of the cell cortex. Science 305, 393–396 (2004).
Norden, C. et al. The NoCut pathway links completion of cytokinesis to spindle midzone function to prevent chromosome breakage. Cell 125, 85–98 (2006).
Whistler, J. L. & Rine, J. Ras2 and Ras1 protein phosphorylation in Saccharomyces cerevisiae. J. Biol. Chem. 272, 18790–18800 (1997).
Cho, W. & Stahelin, R. V. Membrane binding and subcellular targeting of C2 domains. Biochim. Biophys. Acta 1761, 838–849 (2006).
Cho, W. & Stahelin, R. V. Membrane-protein interactions in cell signaling and membrane trafficking. Annu. Rev. Biophys. Biomol. Struct. 34, 119–151 (2005).
Hurley, J. H. Membrane binding domains. Biochim. Biophys. Acta 1761, 805–811 (2006).
VerPlank, L. & Li, R. Cell cycle-regulated trafficking of Chs2 controls actomyosin ring stability during cytokinesis. Mol. Biol. Cell 16, 2529–2543 (2005).
Zhang, G., Kashimshetty, R., Ng, K. E., Tan, H. B. & Yeong, F. M. Exit from mitosis triggers Chs2p transport from the endoplasmic reticulum to mother-daughter neck via the secretory pathway in budding yeast. J. Cell Biol. 174, 207–220 (2006).
Roh, D. H., Bowers, B., Schmidt, M. & Cabib, E. The septation apparatus, an autonomous system in budding yeast. Mol. Biol. Cell 13, 2747–2759 (2002).
Chuang, J. S. & Schekman, R. W. Differential trafficking and timed localization of two chitin synthase proteins, Chs2p and Chs3p. J. Cell Biol. 135, 597–610 (1996).
Martens, S., Kozlov, M. M. & McMahon, H. T. How synaptotagmin promotes membrane fusion. Science 316, 1205–1208 (2007).
Aspenstrom, P., Fransson, A. & Richnau, N. Pombe Cdc15 homology proteins: regulators of membrane dynamics and the actin cytoskeleton. Trends Biochem. Sci. 31, 670–679 (2006).
Field, C. M. & Alberts, B. M. Anillin, a contractile ring protein that cycles from the nucleus to the cell cortex. J. Cell Biol. 131, 165–178 (1995).
Oegema, K., Savoian, M. S., Mitchison, T. J. & Field, C. M. Functional analysis of a human homologue of the Drosophila actin binding protein anillin suggests a role in cytokinesis. J. Cell Biol. 150, 539–552 (2000).
Echard, A., Hickson, G. R., Foley, E. & O'Farrell, P. H. Terminal cytokinesis events uncovered after an RNAi screen. Curr. Biol. 14, 1685–1693 (2004).
Somma, M. P., Fasulo, B., Cenci, G., Cundari, E. & Gatti, M. Molecular dissection of cytokinesis by RNA interference in Drosophila cultured cells. Mol. Biol. Cell 13, 2448–2460 (2002).
Straight, A. F., Field, C. M. & Mitchison, T. J. Anillin binds nonmuscle myosin II and regulates the contractile ring. Mol. Biol. Cell 16, 193–201 (2005).
Zhao, W. M. & Fang, G. Anillin is a substrate of anaphase-promoting complex/cyclosome (APC/C) that controls spatial contractility of myosin during late cytokinesis. J. Biol. Chem. 280, 33516–33524 (2005).
Losev, E. et al. Golgi maturation visualized in living yeast. Nature 441, 1002–1006 (2006).
Spurr, A. R. A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43 (1969).
Reynolds, E. S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 17, 208–212 (1963).
Gambus, A. et al. GINS maintains association of Cdc45 with MCM in replisome progression complexes at eukaryotic DNA replication forks. Nature Cell Biol. 8, 358–366 (2006).
Rost, B., Yachdav, G. & Liu, J. The PredictProtein server. Nucleic Acids Res 32, W321–W326 (2004).
Rizo, J. & Sudhof, T. C. C2-domains, structure and function of a universal Ca2+-binding domain. J. Biol. Chem. 273, 15879–15882 (1998).
Acknowledgements
This work was funded by Cancer Research U.K., from whom K.L. received a Senior Cancer Research Fellowship, and by the EMBO Young Investigator Programme. A.S.D. received a Marie Curie personal training fellowship from the European Union. We are very grateful to S. Bagley for much advice and help with time-lapse video microscopy, B. Glick for advice regarding the use of concanavalin A, J. Wiedenmann for the eQFP construct, and E. Schiebel for plasmids and helpful comments on the manuscript. A.S.D. thanks A. Sanchez-Sanfructuoso and J. Diaz-Marcos for their support.
Author information
Authors and Affiliations
Contributions
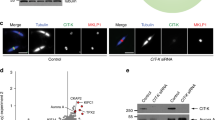
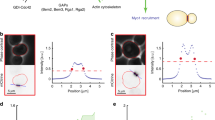
V. M. generated the strains that are described in Supplementary Information, Fig. S1a and b, and screened these strains for defects in cell cycle progression to generate the corrsponding data in Supplementary Information, Fig. 1c. S. M. performed the electron microscopy that is summarized in Fig. 4a. R. J. was responsible for the mass spectrometry data in Fig. 1e. G. P. made the initial observations of the localization of Inn–GFP and taught A. S. D. how to prepare samples for fluorescence microscopy. K. L. made the strains shown in Fig. 8a, b, c. A. S. D. performed all other experiments.
Corresponding author
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and S5 (PDF 2225 kb)
Supplementary Information
Supplementary Movie 1 (MOV 1307 kb)
Supplementary Information
Supplementary Movie 2 (MOV 282 kb)
Rights and permissions
About this article
Cite this article
Sanchez-Diaz, A., Marchesi, V., Murray, S. et al. Inn1 couples contraction of the actomyosin ring to membrane ingression during cytokinesis in budding yeast. Nat Cell Biol 10, 395–406 (2008). https://doi.org/10.1038/ncb1701
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1701
This article is cited by
-
Large-scale identification of genes involved in septal pore plugging in multicellular fungi
Nature Communications (2023)
-
PP2A-Cdc55 phosphatase regulates actomyosin ring contraction and septum formation during cytokinesis
Cellular and Molecular Life Sciences (2022)
-
The final cut: cell polarity meets cytokinesis at the bud neck in S. cerevisiae
Cellular and Molecular Life Sciences (2016)
-
Frontiers of protein expression control with conditional degrons
Pflügers Archiv - European Journal of Physiology (2013)