Abstract
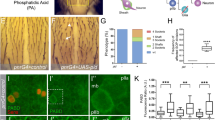
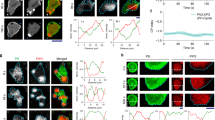
Dishevelled (Dsh) is a cytoplasmic multidomain protein that is required for all known branches of the Wnt signalling pathway1,2,3. The Frizzled/planar cell polarity (Fz/PCP) signalling branch requires an asymmetric cortical localization of Dsh, but this process remains poorly understood. Using a genome-wide RNA interference (RNAi) screen in Drosophila melanogaster cells, we show that Dsh membrane localization is dependent on the Na+/H+ exchange activity of the plasma membrane exchanger Nhe2. Manipulating Nhe2 expression levels in the eye causes PCP defects, and Nhe2 interacts genetically with Fz. Our data show that the binding and surface recruitment of Dsh by Fz is pH- and charge-dependent. We identify a polybasic stretch within the Dsh DEP domain that binds to negatively charged phospholipids and appears to be mechanistically important. Dsh recruitment by Fz can be abolished by converting these basic amino-acid residues into acidic ones, as in the mutant, DshKR/E. In vivo, the DshKR/E(2×) mutant with two substituted residues fails to associate with the membrane during active PCP signalling but rescues canonical Wnt signalling defects in a dsh-background. These results suggest that direct interaction between Fz and Dsh is stabilized by a pH and charge-dependent interaction of the DEP domain with phospholipids. This stabilization is particularly important for the PCP signalling branch and, thus, promotes specific pathway selection in Wnt signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
24 February 2009
In the version of this article initially published online, Fig. 5s had an erroneous 8th transmembrane domain on Frizzled. Supplementary Methods were also missing from the Supplementary Information. These errors have been corrected for the HTML and PDF versions of the article.
References
Boutros, M., Paricio, N., Strutt, D. I. & Mlodzik, M. Dishevelled activates JNK and discriminates between JNK pathways in planar polarity and wingless signaling. Cell 94, 109–118 (1998).
Seifert, J. R. & Mlodzik, M. Frizzled/PCP signalling: a conserved mechanism regulating cell polarity and directed motility. Nature Rev. Genet. 8, 126–138 (2007).
Veeman, M. T., Axelrod, J. D. & Moon, R. T. A second canon. Functions and mechanisms of β-catenin-independent Wnt signaling. Dev. Cell 5, 367–377 (2003).
Axelrod, J. D. Unipolar membrane association of Dishevelled mediates Frizzled planar cell polarity signaling. Genes Dev. 15, 1182–1187 (2001).
Bilic, J. et al. Wnt induces LRP6 signalosomes and promotes dishevelled-dependent LRP6 phosphorylation. Science 316, 1619–1622 (2007).
Wong, H-C. et al. Direct binding of the PDZ domain of Dishevelled to a conserved internal sequence in the C-terminal region of Frizzled. Mol. Cell 12, 1251–1260 (2003).
Rothbacher, U. et al. Dishevelled phosphorylation, subcellular localization and multimerization regulate its role in early embryogenesis. EMBO J. 19, 1010–1022 (2000).
Wong, H. C. et al. Structural basis of the recognition of the dishevelled DEP domain in the Wnt signaling pathway. Nature Struct. Biol. 7, 1178–1184 (2000).
Boutros, M., Mihaly, J., Bouwmeester, T. & Mlodzik, M. Signaling specificity by Frizzled receptors in Drosophila. Science 288, 1825–1828 (2000).
Boutros, M. et al. Genome-wide RNAi analysis of growth and viability in Drosophila cells. Science 303, 832–835 (2004).
Ossipova, O., Dhawan, S., Sokol, S. & Green, J. B. Distinct PAR-1 proteins function in different branches of Wnt signaling during vertebrate development. Dev. Cell 8, 829–841 (2005).
Putney, L. K., Denker, S. P. & Barber, D. L. The changing face of the Na+/H+ exchanger, NHE1: structure, regulation, and cellular actions. Annu. Rev. Pharmacol. Toxicol. 42, 527–552 (2002).
Donowitz, M. & Li, X. Regulatory binding partners and complexes of NHE3. Physiol. Rev. 87, 825–872 (2007).
Ro, H. A. & Carson, J. H. pH microdomains in oligodendrocytes. J. Biol. Chem. 279, 37115–37123 (2004).
Frantz, C., Karydis, A., Nalbant, P., Hahn, K. M. & Barber, D. L. Positive feedback between Cdc42 activity and H+ efflux by the Na-H exchanger NHE1 for polarity of migrating cells. J. Cell Biol. 179, 403–410 (2007).
Wang, D., King, S. M., Quill, T. A., Doolittle, L. K. & Garbers, D. L. A new sperm-specific Na+/H+ exchanger required for sperm motility and fertility. Nature Cell Biol. 5, 1117–1122 (2003).
Yeung, T. et al. Membrane phosphatidylserine regulates surface charge and protein localization. Science 319, 210–213 (2008).
Giannakou, M. E. & Dow, J. A. Characterization of the Drosophila melanogaster alkali-metal/proton exchanger (NHE) gene family. J. Exp. Biol. 204, 3703–3716 (2001).
Mustonen, P., Lehtonen, J., Koiv, A. & Kinnunen, P. K. Effects of sphingosine on peripheral membrane interactions: comparison of adriamycin, cytochrome c, and phospholipase A2. Biochemistry 32, 5373–5380 (1993).
Kooijman, E. E. et al. An electrostatic/hydrogen bond switch as the basis for the specific interaction of phosphatidic acid with proteins. J. Biol. Chem. 282, 11356–11364 (2007).
Lemmon, M. A. Membrane recognition by phospholipid-binding domains. Nature Rev. Mol. Cell Biol. 9, 99–111 (2008).
Penton, A., Wodarz, A. & Nusse, R. A mutational analysis of dishevelled in Drosophila defines novel domains in the dishevelled protein as well as novel suppressing alleles of axin. Genetics 161, 747–762 (2002).
Zeng, X. et al. Initiation of Wnt signaling: control of Wnt coreceptor Lrp6 phosphorylation/activation via frizzled, dishevelled and axin functions. Development 135, 367–375 (2008).
Jenny, A., Darken, R. S., Wilson, P. A. & Mlodzik, M. Prickle and Strabismus form a functional complex to generate a correct axis during planar cell polarity signaling. EMBO J. 22, 4409–4420 (2003).
Bizzarri, R. et al. Development of a novel GFP-based ratiometric excitation and emission pH indicator for intracellular studies. Biophys. J. 90, 3300–3314 (2006).
Schwarz-Romond, T., Merrifield, C., Nichols, B. J. & Bienz, M. The Wnt signalling effector Dishevelled forms dynamic protein assemblies rather than stable associations with cytoplasmic vesicles. J. Cell Sci. 118, 5269–5277 (2005).
Lang, K., Wagner, C., Haddad, G., Burnekova, O. & Geibel, J. Intracellular pH activates membrane-bound Na(+)/H(+) exchanger and vacuolar H(+)-ATPase in human embryonic kidney (HEK) cells. Cell. Physiol. Biochem. 13, 257–262 (2003).
Sandvig, K., Olsnes, S., Petersen, O. W. & van Deurs, B. Acidification of the cytosol inhibits endocytosis from coated pits. J. Cell Biol. 105, 679–689 (1987).
Fang, Y., Vilella-Bach, M., Bachmann, R., Flanigan, A. & Chen, J. Phosphatidic acid-mediated mitogenic activation of mTOR signaling. Science 294, 1942–1945 (2001).
Acknowledgements
We thank Bloomington Stock Center for fly strains, S. Grinstein, J. Orlowski, T. Kirchhausen and R. Bizzarri for cDNA constructs. We are grateful to A. Jenny and Y. Wang for the cloning of cDNA constructs, and U. Weber for analysis of the embryonic/early larval lethal phenotype. We thank C. Iomini, R. Krauss, S. Sokol and D. del Alamo for reading the manuscript, members of the Mlodzik laboratory for discussions and S. Okello, G. Garcia and M. Stricker for technical support. The work has been supported by NIH grant to M.M. RO1 GM62917. M.S. was a recipient of EMBO and DFG long-term fellowships.
Author information
Authors and Affiliations
Contributions
M.S. coordinated the project, designed and conducted the RNAi screen, performed the experimental work and data analysis and wrote the manuscript. W.J.G. performed the pupal dissections. W.J.G. and D.G. assisted with experiments. T.J.K. helped with the RNAi screen assay. R.R. and L.S.M. performed pH measurements. Y.S., H.-J.L. and J.Z. purified the DEP domain and performed electrostatic potential calculations. A.-L.W., Y.F. and J.C. performed SUV lipid binding assays. J.T.D. provided Nhe2 tools. M.B. designed and conducted the RNAi screen. M.M. coordinated the project, assisted with planning the experiments and data analysis and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1209 kb)
Rights and permissions
About this article
Cite this article
Simons, M., Gault, W., Gotthardt, D. et al. Electrochemical cues regulate assembly of the Frizzled/Dishevelled complex at the plasma membrane during planar epithelial polarization. Nat Cell Biol 11, 286–294 (2009). https://doi.org/10.1038/ncb1836
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1836
This article is cited by
-
Intracellular pH dynamics regulates intestinal stem cell lineage specification
Nature Communications (2023)
-
Analysing bioelectrical phenomena in the Drosophila ovary with genetic tools: tissue-specific expression of sensors for membrane potential and intracellular pH, and RNAi-knockdown of mechanisms involved in ion exchange
BMC Developmental Biology (2020)
-
Electrochemical patterns during Drosophila oogenesis: ion-transport mechanisms generate stage-specific gradients of pH and membrane potential in the follicle-cell epithelium
BMC Developmental Biology (2019)
-
Non-canonical Wnt induces chondrocyte de-differentiation through Frizzled 6 and DVL-2/B-raf/CaMKIIα/syndecan 4 axis
Cell Death & Differentiation (2018)
-
Planar cell polarity in development and disease
Nature Reviews Molecular Cell Biology (2017)