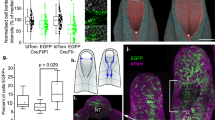
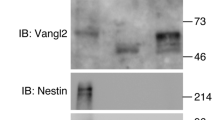
Abstract
Craniorachischisis is a rare but severe birth defect that results in a completely open neural tube. Mouse mutants in planar cell polarity (PCP) signalling components have deficits in the morphological movements of convergent extension that result in craniorachischisis. Using a forward genetic screen in mice, we identified Sec24b, a cargo-sorting member of the core complex of the endoplasmic reticulum (ER)-to-Golgi transport vesicle COPII, as critical for neural tube closure. Sec24bY613 mutant mice exhibit craniorachischisis, deficiencies in convergent extension and other PCP-related phenotypes. Vangl2, a key component of the PCP-signalling pathway critical for convergent extension, is selectively sorted into COPII vesicles by Sec24b. Moreover, Sec24bY613 genetically interacts with a loss-of-function Vangl2 allele (Vangl2LP), causing a marked increase in the prevalence of spina bifida. Interestingly, the Vangl2 looptail point mutants Vangl2D255E and Vangl2S464N, known to cause defects in convergent extension, fail to sort into COPII vesicles and are trapped in the ER. Thus, during COPII vesicle formation, Sec24b shows cargo specificity for a core PCP component, Vangl2, of which proper ER-to-Golgi transport is essential for the establishment of PCP, convergent extension and closure of the neural tube.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kibar, Z., Capra, V. & Gros, P. Toward understanding the genetic basis of neural tube defects. Clin. Genet. 71, 295–310 (2007).
De Marco, P., Merello, E., Mascelli, S. & Capra, V. Current perspectives on the genetic causes of neural tube defects. Neurogenetics 7, 201–221 (2006).
Kibar, Z. Identification of a new chemically induced allele (Lpm1Jus) at the loop-tail locus: morphology, histology, and genetic mapping. Genomics 72, 331–337 (2001).
Montcouquiol, M. et al. Identification of Vangl2 and Scrb1 as planar polarity genes in mammals. Nature 423, 173–177 (2003).
Wang, Y., N., G. & Nathans, J. The role of Frizzled3 and Frizzled6 in neural tube closure and in the planar polarity of inner-ear sensory hair cells. J. Neurosci. 26, 2147–2156 (2006).
Curtin, J. Mutation of Celsr1 disrupts planar polarity of inner ear hair cells and causes severe neural tube defects in the mouse. Curr. Biol. 13, 1129–1133 (2003).
Hamblet, N. et al. Dishevelled 2 is essential for cardiac outflow tract development, somite segmentation and neural tube closure. Development 129, 5827–5838 (2002).
Etheridge, S. L. et al. Murine dishevelled 3 functions in redundant pathways with dishevelled 1 and 2 in normal cardiac outflow tract, cochlea, and neural tube development. PLoS Genet. 4, e1000259 (2008).
Lu, X. et al. PTK7/CCK-4 is a novel regulator of planar cell polarity in vertebrates. Nature 430, 93–98 (2004).
Murdoch, J. et al. Circletail, a new mouse mutant with severe neural tube defects: chromosomal localization and interaction with the loop-tail mutation. Genomics 78, 55–63 (2001).
Murdoch, J. et al. Disruption of scribble (Scrb1) causes severe neural tube defects in the circletail mouse. Hum. Mol. Genet. 12, 87–98 (2003).
Ybot-Gonzalez, P. et al. Convergent extension, planar-cell-polarity signalling and initiation of mouse neural tube closure. Development 134, 789–799 (2007).
Mlodzik, M. Planar cell polarization: do the same mechanisms regulate Drosophila tissue polarity and vertebrate gastrulation? Trends Genet. 18, 564–571 (2002).
Wallingford, J., Fraser, S. E. & Harland, R. M. Convergent extension: the molecular control of polarized cell movement during embryonic development. Dev. Cell 2, 695–706 (2002).
Kasarskis, A., Manova, K. & Anderson, K. V. A phenotype-based screen for embryonic lethal mutations in the mouse. Proc. Natl Acad. Sci. USA 95, 7485–7490 (1998).
Wendeler, M., Paccaud, J. & Hauri, H. Role of Sec24 isoforms in selective export of membrane proteins from the endoplasmic reticulum. EMBO Rep. 8, 258–264 (2007).
Shimoni, Y. et al. Lst1p and Sec24p cooperate in sorting of the plasma membrane ATPase into COPII vesicles in Saccharomyces cerevisiae. J. Cell Biol. 151, 973–984 (2000).
Miller, E., Antonny, B., Hamamoto, S. & Schekman, R. Cargo selection into COPII vesicles is driven by the Sec24p subunit. EMBO J. 21, 6105–6113 (2002).
Miller, E. A. et al. Multiple cargo binding sites on the COPII subunit Sec24p ensure capture of diverse membrane proteins into transport vesicles. Cell 114, 497–509 (2003).
Bi, X., Corpina, R. A. & Goldberg, J. Structure of the Sec23/24-Sar1 pre-budding complex of the COPII vesicle coat. Nature 419, 271–277 (2002).
Wallingford, J. & Harland, R. Neural tube closure requires Dishevelled-dependent convergent extension of the midline. Development 129, 5815–5825 (2002).
Jones, C. & Chen, P. Planar cell polarity signaling in vertebrates. Bioessays 29, 120–132 (2007).
Copp, A. J., Checiu, I. & Henson, J. N. Developmental basis of severe neural tube defects in the loop-tail (Lp) mutant mouse: use of microsatellite DNA markers to identify embryonic genotype. Dev. Biol. 165, 20–29 (1994).
Qian, D. et al. Wnt5a functions in planar cell polarity regulation in mice. Dev. Biol. 306, 121–133 (2007).
Kim, J. et al. Biogenesis of γ-secretase early in the secretory pathway. J. Cell Biol. 179, 951–963 (2007).
Torban, E. et al. Tissue, cellular and sub-cellular localization of the Vangl2 protein during embryonic development: effect of the Lp mutation. Gene Expr. Patterns 7, 346–354 (2007).
Justice, M. et al. Mamm. Effects of ENU dosage on mouse strains. Genome 11, 484–488 (2000).
Justice, M. J., Noveroske, J. K., Weber, J. S., Zheng, B. & Bradley, A. Mouse ENU mutagenesis. Hum. Mol. Genet. 8, 1955–1963 (1999).
Weber, J. S., Salinger, A. & Justice, M. J. Optimal N-ethyl-N-nitrosourea (ENU) doses for inbred mouse strains. Genesis 26, 230–233 (2000).
Wilson, R. et al. The translocation, folding, assembly and redox-dependent degradation of secretory and membrane proteins in semi-permeabilized mammalian cells. Biochem. J. 307, 679–687 (1995).
Kim, J., Hamamoto, S., Ravazzola, M., Orci, L. & Schekman, R. Uncoupled packaging of amyloid precursor protein and presenilin 1 into coat protein complex II vesicles. J. Biol. Chem. 280, 7758–7768 (2005).
Fromme, J. et al. The genetic basis of a craniofacial disease provides insight into COPII coat assembly Dev. Cell 13, 623–634 (2007).
Acknowledgements
We thank J. Nathans, T. Makita, A. Kolodkin, and members of the Ginty laboratory for discussions and critical reading of this manuscript; P. Gros for providing the Vangl2 antibody; J. C. Fromme, B. Kleizen and A. Schindler for Anti-Sec13 and Anti-Sec22 antiserum; C.Chan for recombinant Sec23A/Sec24A and Sec23A/Sec24C; B. Yang and Y. Wu for excellent technical help. This work was funded by NIH grants NS34814 (D.D.G.) and the Johns Hopkins Mind Brain Institute. D.D.G. and R.S. are investigators of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
J.M. and D.D.G. designed the forward genetic screen. J.M. developed and implemented the screen, identified the novel mouse line with craniorachischisis, did the positional cloning to map the mutation to Sec24bY613, characterized PCP-related phenotypes in the Sec24bY613 mutants and showed that Vangl2LP and Sec24bY613 genetically interact. D.J. did the experiments showing that Sec24bY613 cannot form an oligomer with Sec23, and the COPII vesicle sorting experiments showing that Vangl2 is selectively sorted by Sec24b and that mutant forms of Vangl2 fail to properly sort into these vesicles. K.W. identified and analysed the missexpression of Vangl2 in Sec24bY613 mutant neuroepithelium. Y.W. aided in experimental planning of PCP phenotype analysis and performed some cochlear hair cell orientation analysis. S.S. aided in the implementation of the forward genetic screen that identified Sec24bY613 and the mapping of the point mutation. R.S. conducted experimental design and analysis of the role of Sec24b in the sorting and expression of Vangl2 and Vangl2-mutant proteins. D.D.G. conducted experimental design and analysis of the forward genetic screen, mutation mapping, and mouse phenotypic analysis. J.M. and D.D.G. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1938 kb)
Rights and permissions
About this article
Cite this article
Merte, J., Jensen, D., Wright, K. et al. Sec24b selectively sorts Vangl2 to regulate planar cell polarity during neural tube closure. Nat Cell Biol 12, 41–46 (2010). https://doi.org/10.1038/ncb2002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2002
This article is cited by
-
Unraveling the genetic architecture of congenital vertebral malformation with reference to the developing spine
Nature Communications (2024)
-
Functional interaction between Vangl2 and N-cadherin regulates planar cell polarization of the developing neural tube and cochlear sensory epithelium
Scientific Reports (2023)
-
In vitro reconstitution of COPII vesicles from Arabidopsis thaliana suspension-cultured cells
Nature Protocols (2023)
-
USP39 is essential for mammalian epithelial morphogenesis through upregulation of planar cell polarity components
Communications Biology (2022)
-
Wnt/planar cell polarity signaling controls morphogenetic movements of gastrulation and neural tube closure
Cellular and Molecular Life Sciences (2022)