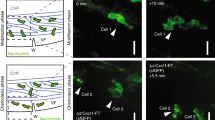
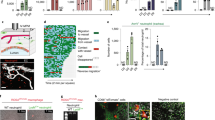
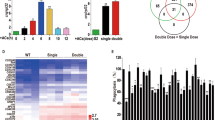
Abstract
Human immune cells have to penetrate an endothelial barrier during their beneficial pursuit of infection and their destructive infiltration of tissues in autoimmune diseases. This transmigration requires Rap1 GTPase to activate integrin affinity1. We define a new model system for this process by demonstrating, with live imaging and genetics, that during embryonic development Drosophila melanogaster immune cells penetrate an epithelial, Drosophila E-cadherin (DE-cadherin)-based tissue barrier. A mutant in RhoL, a GTPase homologue that is specifically expressed in haemocytes, blocks this invasive step but not other aspects of guided migration. RhoL mediates integrin adhesion caused by Drosophila Rap1 overexpression and moves Rap1 away from a concentration in the cytoplasm to the leading edge during invasive migration. These findings indicate that a programmed migratory step during Drosophila development bears striking molecular similarities to vertebrate immune cell transmigration during inflammation, and identify RhoL as a new regulator of invasion, adhesion and Rap1 localization. Our work establishes the utility of Drosophila for identifying novel components of immune cell transmigration and for understanding the in vivo interplay of immune cells with the barriers they penetrate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Abram, C. L. & Lowell, C. A. The ins and outs of leukocyte integrin signaling. Annu. Rev. Immunol. 2 7, 339–362 (2009).
Etzioni, A., Doerschuk, C. M. & Harlan, J. M. Of man and mouse: leukocyte and endothelial adhesion molecule deficiencies. Blood 94, 3281–3288 (1999).
Luster, A. D., Alon, R. & von Andrian, U. H. Immune cell migration in inflammation: present and future therapeutic targets. Nat. Immunol. 6, 1182–1190 (2005).
Kim, J. V., Kang, S. S., Dustin, M. L. & McGavern, D. B. Myelomonocytic cell recruitment causes fatal CNS vascular injury during acute viral meningitis. Nature 457, 191–195 (2009).
Shimonaka, M. et al. Rap1 translates chemokine signals to integrin activation, cell polarization, and motility across vascular endothelium under flow. J. Cell Biol. 161, 417–427 (2003).
Katagiri, K., Maeda, A., Shimonaka, M. & Kinashi, T. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat. Immunol. 4, 741–748 (2003).
Katagiri, K., Imamura, M. & Kinashi, T. Spatiotemporal regulation of the kinase Mst1 by binding protein RAPL is critical for lymphocyte polarity and adhesion. Nat. Immunol. 7, 919–928 (2006).
Evans, C. J., Hartenstein, V. & Banerjee, U. Thicker than blood: conserved mechanisms in Drosophila and vertebrate hematopoiesis. Dev. Cell 5, 673–690 (2003).
Wood, W. & Jacinto, A. Drosophila melanogaster embryonic haemocytes: masters of multitasking. Nat. Rev. Mol. Cell Biol. 8, 542–551 (2007).
Babcock, D. T. et al. Circulating blood cells function as a surveillance system for damaged tissue in Drosophila larvae. Proc. Natl Acad. Sci. USA 105, 10017–10022 (2008).
Stramer, B. et al. Live imaging of wound inflammation in Drosophila embryos reveals key roles for small GTPases during in vivo cell migration. J. Cell Biol. 168, 567–573 (2005).
Wood, W., Faria, C. & Jacinto, A. Distinct mechanisms regulate haemocyte chemotaxis during development and wound healing in Drosophila melanogaster. J. Cell Biol. 173, 405–416 (2006).
Pastor-Pareja, J. C., Wu, M. & Xu, T. An innate immune response of blood cells to tumors and tissue damage in Drosophila. Dis. Mod. Mech. 1, 144–154 (2008).
Bruckner, K. et al. The PDGF/VEGF receptor controls blood cell survival in Drosophila. Dev. Cell 7, 73–84 (2004).
Cho, N. K. et al. Developmental control of blood cell migration by the Drosophila VEGF pathway. Cell 108, 865–876 (2002).
Friedl, P. & Wolf, K. Tumour-cell invasion and migration: diversity and escape mechanisms. Nature Rev. Cancer 3, 362–374 (2003).
Murphy, A. M. & Montell, D. J. Cell type-specific roles for Cdc42, Rac, and RhoL in Drosophila oogenesis. J. Cell Biol. 133, 617–630 (1996).
Paladi, M. & Tepass, U. Function of Rho GTPases in embryonic blood cell migration in Drosophila. J. Cell Sci. 117, 6313–6326 (2004).
Page-McCaw, A., Serano, J., Sante, J. M. & Rubin, G. M. Drosophila matrix metalloproteinases are required for tissue remodeling, but not embryonic development. Dev. Cell 4, 95–106 (2003).
Srivastava, A., Pastor-Pareja, J. C., Igaki, T., Pagliarini, R. & Xu, T. Basement membrane remodeling is essential for Drosophila disc eversion and tumor invasion. Proc. Natl Acad. Sci. USA 104, 2721–2726 (2007).
Tepass, U. et al. Shotgun encodes Drosophila E-cadherin and is preferentially required during cell rearrangement in the neuroectoderm and other morphogenetically active epithelia. Genes Dev. 10, 672–685 (1996).
Pacquelet, A. & Rorth, P. Regulatory mechanisms required for DE-cadherin function in cell migration and other types of adhesion. J. Cell Biol. 170, 803–812 (2005).
Huelsmann, S., Hepper, C., Marchese, D., Knoll, C. & Reuter, R. The PDZ-GEF dizzy regulates cell shape of migrating macrophages via Rap1 and integrins in the Drosophila embryo. Development 133, 2915–2924 (2006).
Bogaert, T., Brown, N. & Wilcox, M. The Drosophila PS2 antigen is an invertebrate integrin that, like the fibronectin receptor, becomes localized to muscle attachments. Cell 51, 929–940 (1987).
Tepass, U., Fessler, L. I., Aziz, A. & Hartenstein, V. Embryonic origin of haemocytes and their relationship to cell death in Drosophila. Development 120, 1829–1837 (1994).
de Velasco, B., Mandal, L., Mkrtchyan, M. & Hartenstein, V. Subdivision and developmental fate of the head mesoderm in Drosophila melanogaster. Dev. Genes Evol. 216, 39–51 (2006).
Knox, A. L. & Brown, N. H. Rap1 GTPase regulation of adherens junction positioning and cell adhesion. Science 295, 1285–1288 (2002).
Bertrand, J. Y. et al. Characterization of purified intraembryonic hematopoietic stem cells as a tool to define their site of origin. Proc. Natl Acad. Sci. USA 102, 134–139 (2005).
Menasche, G., Kliche, S., Bezman, N. & Schraven, B. Regulation of T-cell antigen receptor-mediated inside-out signalling by cytosolic adapter proteins and Rap1 effector molecules. Immunol. Rev. 218, 82–91 (2007).
Tepass, U. & Hartenstein, V. The development of cellular junctions in the Drosophila embryo. Dev. Biol. 161, 563–596 (1994).
Choi, E. Y., Santoso, S. & Chavakis, T. Mechanisms of neutrophil transendothelial migration. Front Biosci. 14, 1596–1605 (2009).
Mor, A., Dustin, M. L. & Philips, M. R. Small GTPases and LFA-1 reciprocally modulate adhesion and signaling. Immunol. Rev. 218, 114–125 (2007).
de Toledo, M. et al. The GTP/GDP cycling of rho GTPase TCL is an essential regulator of the early endocytic pathway. Mol. Biol. Cell 14, 4846–4856 (2003).
Kawase, K. et al. GTP hydrolysis by the Rho family GTPase TC10 promotes exocytic vesicle fusion. Dev. Cell 11, 411–421 (2006).
Lammermann, T. et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008).
Lehmann, R. & Tautz, D. In situ hybridization to RNA. Methods in cell biology 44, 575–598 (1994).
Acknowledgements
We thank D. Jukam, P. Kunwar, A-M. Sudarov, and S. Wang for help during the GAL4 enhancer screen, and D. Demy for input. We thank all the people who shared their reagents: U. Heberlein, K. Brueckner, N. Perrimon, D. Montell, R. Reuter and A. Page-McCaw for stocks and T. Uemura, D. Branton, R, Dubreuil, H. Bellen, N. Lowe, D. Ryoo and J. Treisman for antibodies. We thank the Developmental Studies Hybridoma Bank developed with NICHD support and maintained by the University of Iowa for antibodies and the Bloomington Stock Center for flies. We are grateful for conversations with P. Rangan, P. Kunwar and many other members of the laboratory. We thank M. Dustin, S. Schwab and laboratory members for comments on the manuscript. We thank NICHD for supporting part of this work. R.L. is an investigator of the HHMI.
Author information
Authors and Affiliations
Contributions
Project conception and planning were conducted by D.S. with guidance from R.L. D.S. performed and analysed all experiments except the following: M.H. participated in the screen, and produced all the data in Supplementary Information, Fig. S2a, b and d except the production and analysis of the extent of the XA12 excision and O.M. produced Fig. 4c and assisted in staining for Fig. 5h–i. Experimental interpretation was conducted by D.S., M.H. and R.L. The manuscript was written by D.S. and edited by R.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1502 kb)
Supplementary Information
Supplementary Movie 1 (MOV 17415 kb)
Supplementary Information
Supplementary Movie 2 (MOV 2755 kb)
Supplementary Information
Supplementary Movie 3 (MOV 11101 kb)
Supplementary Information
Supplementary Movie 4 (MOV 2736 kb)
Supplementary Information
Supplementary Movie 5 (MOV 10777 kb)
Supplementary Information
Supplementary Movie 6 (MOV 9705 kb)
Supplementary Information
Supplementary Movie 7 (MOV 5555 kb)
Rights and permissions
About this article
Cite this article
Siekhaus, D., Haesemeyer, M., Moffitt, O. et al. RhoL controls invasion and Rap1 localization during immune cell transmigration in Drosophila. Nat Cell Biol 12, 605–610 (2010). https://doi.org/10.1038/ncb2063
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2063
This article is cited by
-
Hemocytes in Drosophila melanogaster embryos move via heterogeneous anomalous diffusion
Communications Physics (2022)
-
SCAR/WAVE-mediated processing of engulfed apoptotic corpses is essential for effective macrophage migration in Drosophila
Cell Death & Differentiation (2013)