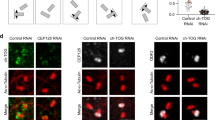
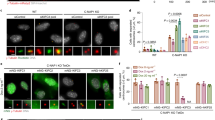
Abstract
Regulated centrosome biogenesis is required for accurate cell division and for maintaining genome integrity1. Centrosomes consist of a centriole pair surrounded by a protein network known as pericentriolar material1 (PCM). PCM assembly is a tightly regulated, critical step that determines the size and capability of centrosomes2,3,4. Here, we report a role for tubulin in regulating PCM recruitment through the conserved centrosomal protein Sas-4. Tubulin directly binds to Sas-4; together they are components of cytoplasmic complexes of centrosomal proteins5,6. A Sas-4 mutant, which cannot bind tubulin, enhances centrosomal protein complex formation and has abnormally large centrosomes with excessive activity. These results suggest that tubulin negatively regulates PCM recruitment. Whereas tubulin–GTP prevents Sas-4 from forming protein complexes, tubulin–GDP promotes it. Thus, the regulation of PCM recruitment by tubulin depends on its GTP/GDP-bound state. These results identify a role for tubulin in regulating PCM recruitment independent of its well-known role as a building block of microtubules7. On the basis of its guanine-bound state, tubulin can act as a molecular switch in PCM recruitment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Nigg, E. A. & Raff, J. W. Centrioles, centrosomes, and cilia in health and disease. Cell 139, 663–678 (2009).
Conduit, P. T. et al. Centrioles regulate centrosome size by controlling the rate of Cnn incorporation into the PCM. Curr. Biol. 20, 2178–2186 (2010).
Kirkham, M., Muller-Reichert, T., Oegema, K., Grill, S. & Hyman, A. A. SAS-4 is a C. elegans centriolar protein that controls centrosome size. Cell 112, 575–587 (2003).
Piehl, M., Tulu, U. S., Wadsworth, P. & Cassimeris, L. Centrosome maturation: measurement of microtubule nucleation throughout the cell cycle by using GFP-tagged EB1. Proc. Natl Acad. Sci. USA 101, 1584–1588 (2004).
Gopalakrishnan, J. et al. Sas-4 provides a scaffold for cytoplasmic complexes and tethers them in a centrosome. Nat. Commun. 2, 359 (2011).
Hung, L. Y., Chen, H. L., Chang, C. W., Li, B. R. & Tang, T. K. Identification of a novel microtubule-destabilizing motif in CPAP that binds to tubulin heterodimers and inhibits microtubule assembly. Mol. Biol. Cell 15, 2697–2706 (2004).
Desai, A. & Mitchison, T. J. Microtubule polymerization dynamics. Annu. Rev. Cell Dev. Biol. 13, 83–117 (1997).
Dammermann, A., Maddox, P. S., Desai, A. & Oegema, K. SAS-4 is recruited to a dynamic structure in newly forming centrioles that is stabilized by the γ-tubulin-mediated addition of centriolar microtubules. J. Cell Biol. 180, 771–785 (2008).
Hung, L. Y., Tang, C. J. & Tang, T. K. Protein 4.1 R-135 interacts with a novel centrosomal protein (CPAP) which is associated with the γ-tubulin complex. Mol. Cell Biol. 20, 7813–7825 (2000).
Pelletier, L., O’Toole, E., Schwager, A., Hyman, A. A. & Muller-Reichert, T. Centriole assembly in Caenorhabditis elegans. Nature 444, 619–623 (2006).
Leidel, S. & Gonczy, P. SAS-4 is essential for centrosome duplication in Celegans and is recruited to daughter centrioles once per cell cycle. Dev. Cell 4, 431–439 (2003).
Hsu, W. B. et al. Functional characterization of the microtubule-binding and -destabilizing domains of CPAP and d-SAS-4. Exp. Cell Res. 314, 2591–2602 (2008).
Tang, C. J., Fu, R. H., Wu, K. S., Hsu, W. B. & Tang, T. K. CPAP is a cell-cycle regulated protein that controls centriole length. Nat. Cell Biol. 11, 825–831 (2009).
Basto, R. et al. Flies without Centrioles. Cell 125, 1375–1386 (2006).
Blachon, S. et al. Drosophila asterless and vertebrate Cep152 Are orthologs essential for centriole duplication. Genetics 180, 2081–2094 (2008).
Blachon, S. et al. A proximal centriole-like structure is present in Drosophila spermatids and can serve as a model to study centriole duplication. Genetics 182, 133–144 (2009).
Kohlmaier, G. et al. Overly long centrioles and defective cell division upon excess of the SAS-4-related protein CPAP. Curr. Biol. 19, 1012–1018 (2009).
Schmidt, T. I. et al. Control of centriole length by CPAP and CP110. Curr. Biol. 19, 1005–1011 (2009).
Li, K. et al. Drosophila centrosomin protein is required for male meiosis and assembly of the flagellar axoneme. J. Cell Biol. 141, 455–467 (1998).
Rogers, G. C., Rusan, N. M., Peifer, M. & Rogers, S. L. A multicomponent assembly pathway contributes to the formation of acentrosomal microtubule arrays in interphase Drosophila cells. Mol. Biol. Cell 19, 3163–3178 (2008).
Choi, Y. K., Liu, P., Sze, S. K., Dai, C. & Qi, R. Z. CDK5RAP2 stimulates microtubule nucleation by the γ-tubulin ring complex. J. Cell Biol. 191, 1089–1095 (2010).
Rusan, N. & Peifer, M. A role for a novel centrosome cycle in asymmetric cell division. J. Cell Biol. 177, 13–33 (2007).
Giansanti, M., Gatti, M. & Bonaccorsi, S. The role of centrosomes and astral microtubules during asymmetric division of Drosophila neuroblasts. Development 128, 1137–1182 (2001).
Sandoval, I. V., Jameson, J. L., Niedel, J., MacDonald, E. & Cuatrecasas, P. Role of nucleotides in tubulin polymerization: effect of guanosine 5’-methylene diphosphonate. Proc. Natl Acad. Sci. USA 75, 3178–3182 (1978).
Cormier, A. et al. The PN2-3 domain of centrosomal P4.1-associated protein implements a novel mechanism for tubulin sequestration. J. Biol. Chem. 284, 6909–6917 (2009).
Mozziconacci, J., Sandblad, L., Wachsmuth, M., Brunner, D. & Karsenti, E. Tubulin dimers oligomerize before their incorporation into microtubules. PLoS One 3, e3821 (2008).
Hiller, G. & Weber, K. Radioimmunoassay for tubulin: a quantitative comparison of the tubulin content of different established tissue culture cells and tissues. Cell 14, 795–804 (1978).
David-Pfeuty, T., Simon, C. & Pantaloni, D. Effect of antimitotic drugs on tubulin GTPase activity and self-assembly. J. Biol. Chem. 254, 11696–11702 (1979).
Cabrera-Vera, T. M. et al. Insights into G protein structure, function, and regulation. Endocr. Rev. 24, 765–781 (2003).
Takai, Y., Sasaki, T. & Matozaki, T. Small GTP-binding proteins. Physiol. Rev. 81, 153–208 (2001).
Melki, R., Carlier, M. F., Pantaloni, D. & Timasheff, S. N. Cold depolymerization of microtubules to double rings: geometric stabilization of assemblies. Biochemistry 28, 9143–9152 (1989).
Kobayashi, T. & Dynlacht, B. D. Regulating the transition from centriole to basal body. J. Cell Biol. 193, 435–444 (2011).
Janulevicius, A., van Pelt, J. & van Ooyen, A. Compartment volumeinfluences microtubule dynamic instability: a model study. Biophys. J. 90, 788–798 (2006).
Zhai, Y., Kronebusch, P. J., Simon, P. M. & Borisy, G. G. Microtubule dynamics at the G2/M transition: abrupt breakdown of cytoplasmic microtubules at nuclear envelope breakdown and implications for spindle morphogenesis. J. Cell Biol. 135, 201–214 (1996).
Schiff, P. B. & Horwitz, S. B. Taxol stabilizes microtubules in mouse fibroblast cells. Proc. Natl Acad. Sci. USA 77, 1561–1565 (1980).
Oegema, K. et al. Characterization of two related Drosophila γ-tubulin complexes that differ in their ability to nucleate microtubules. J. Cell Biol. 144, 721–733 (1999).
Zhou, P., Lugovskoy, A. A. & Wagner, G. A solubility-enhancement tag (SET) for NMR studies of poorly behaving proteins. J. Biomol. NMR 20, 11–14 (2001).
Moritz, M. et al. Three-dimensional structural characterization of centrosomes from early Drosophila embryos. J. Cell Biol. 130, 1149–1159 (1995).
Gopalakrishnan, J. et al. Self-assembling SAS-6 multimer is a core centriole building block. J. Biol. Chem. 285, 8759–8770 (2010).
Wodarz, A., Ramrath, A., Kuchinke, U. & Knust, E. Bazooka provides an apicalcue for Inscuteable localization in Drosophila neuroblasts. Nature 402, 544–547 (1999).
Acknowledgements
We would like to thank J. Iwasa for scientific illustrations; T. Mitchision A. Johnson, I. Cheeseman and J. Malicki for scientific discussions; T. Kaufman, J. Raff, B. Raynaud-Messina and T. K. Tang (Institute of Biomedical Sciences, Taipei, Taiwan) for reagents; R. Reed laboratory (Harvard Medical School, USA), F. Eric, A. Hari, R. Rodriguez for technical help with biophysical experiments; E. Koundakjian for scientific editing and discussions; and electron microscopy facility staff at HMS for help with electron microscopy analyses. This work was supported by a grant (R01GM098394) from the National Institute of General Medical Sciences.
Author information
Authors and Affiliations
Contributions
J.G. and T.A.R. conceived the project. J.G. performed most of the experiments described herein. T.A-R. supervised the project. Y-F.C. performed phase and electron microscopy analyses. A.H. performed biochemical complex analyses. M.L.B. generated constructs and took part in the biochemical purification of recombinant proteins. D.A.L. and N.M.R. advised on and discussed larval brain analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 826 kb)
Rights and permissions
About this article
Cite this article
Gopalakrishnan, J., Frederick Chim, YC., Ha, A. et al. Tubulin nucleotide status controls Sas-4-dependent pericentriolar material recruitment. Nat Cell Biol 14, 865–873 (2012). https://doi.org/10.1038/ncb2527
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2527
This article is cited by
-
Molecular basis for CPAP-tubulin interaction in controlling centriolar and ciliary length
Nature Communications (2016)
-
Centrosome function and assembly in animal cells
Nature Reviews Molecular Cell Biology (2015)