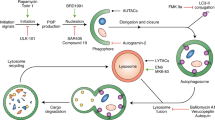
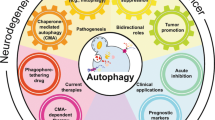
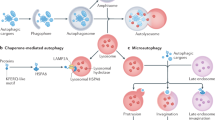
Abstract
Autophagy is an evolutionarily conserved mechanism for protein degradation that is critical for the maintenance of homeostasis in man. Autophagy has unexpected pleiotropic functions that favor survival of the cell, including nutrient supply under starvation, cleaning of the cellular interior, defense against infection and antigen presentation. Moreover, defective autophagy is associated with a diverse range of disease states, including neurodegeneration, cancer and Crohn's disease. Here we discuss the roles of mammalian autophagy in health and disease and highlight recent advances in pharmacological manipulation of autophagic pathways as a therapeutic strategy for a variety of pathological conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Yoshimori, T. Autophagy: a regulated bulk degradation process inside cells. Biochem. Biophys. Res. Commun. 313, 453–458 (2004).
Yu, L. et al. Termination of autophagy and reformation of lysosomes regulated by mTOR. Nature 465, 942–946 (2010).
Nakatogawa, H., Suzuki, K., Kamada, Y. & Ohsumi, Y. Dynamics and diversity in autophagy mechanisms: lessons from yeast. Nat. Rev. Mol. Cell Biol. 10, 458–467 (2009).
Hosokawa, N. et al. Nutrient-dependent mTORC1 association with the ULK1-Atg13–FIP200 complex required for autophagy. Mol. Biol. Cell 20, 1981–1991 (2009).
Kroemer, G., Marino, G. & Levine, B. Autophagy and the integrated stress response. Mol. Cell 40, 280–293 (2010).
Simonsen, A. & Tooze, S.A. Coordination of membrane events during autophagy by multiple class III PI3-kinase complexes. J. Cell Biol. 186, 773–782 (2009).
Noda, T., Matsunaga, K., Taguchi-Atarashi, N. & Yoshimori, T. Regulation of membrane biogenesis in autophagy via PI3P dynamics. Semin. Cell Dev. Biol. 21, 671–676 (2010).
Fujita, N. et al. The Atg16L complex specifies the site of LC3 lipidation for membrane biogenesis in autophagy. Mol. Biol. Cell 19, 2092–2100 (2008).
Fujita, N. et al. An Atg4B mutant hampers the lipidation of LC3 paralogues and causes defects in autophagosome closure. Mol. Biol. Cell 19, 4651–4659 (2008).
Sou, Y.S. et al. The Atg8 conjugation system is indispensable for proper development of autophagic isolation membranes in mice. Mol. Biol. Cell 19, 4762–4775 (2008).
Webber, J.L. & Tooze, S.A. New insights into the function of Atg9. FEBS Lett. 584, 1319–1326 (2010).
Matsunaga, K. et al. Autophagy requires endoplasmic reticulum targeting of the PI3-kinase complex via Atg14L. J. Cell Biol. 190, 511–521 (2010).
Yousefi, S. et al. Calpain-mediated cleavage of Atg5 switches autophagy to apoptosis. Nat. Cell Biol. 8, 1124–1132 (2006).
Zhao, Z. et al. Autophagosome-independent essential function for the autophagy protein Atg5 in cellular immunity to intracellular pathogens. Cell Host Microbe 4, 458–469 (2008).
Saitoh, T. et al. Atg9a controls dsDNA-driven dynamic translocation of STING and the innate immune response. Proc. Natl. Acad. Sci. USA 106, 20842–20846 (2009).
Radoshevich, L. et al. ATG12 conjugation to ATG3 regulates mitochondrial homeostasis and cell death. Cell 142, 590–600 (2010).
Tooze, S.A. & Yoshimori, T. The origin of the autophagosomal membrane. Nat. Cell Biol. 12, 831–835 (2010).
Hailey, D.W. et al. Mitochondria supply membranes for autophagosome biogenesis during starvation. Cell 141, 656–667 (2010).
Ravikumar, B., Moreau, K., Jahreiss, L., Puri, C. & Rubinsztein, D.C. Plasma membrane contributes to the formation of pre-autophagosomal structures. Nat. Cell Biol. 12, 747–757 (2010).
Tsukamoto, S., Kuma, A. & Mizushima, N. The role of autophagy during the oocyte-to-embryo transition. Autophagy 4, 1076–1078 (2008).
Tsukamoto, S. et al. Autophagy is essential for preimplantation development of mouse embryos. Science 321, 117–120 (2008).
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009).
Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036 (2004).
Heilbronn, L.K. & Ravussin, E. Calorie restriction and aging: review of the literature and implications for studies in humans. Am. J. Clin. Nutr. 78, 361–369 (2003).
Vellai, T. Autophagy genes and ageing. Cell Death Differ. 16, 94–102 (2009).
Simonsen, A. et al. Promoting basal levels of autophagy in the nervous system enhances longevity and oxidant resistance in adult Drosophila. Autophagy 4, 176–184 (2008).
Zheng, S. et al. Deletion of the huntingtin polyglutamine stretch enhances neuronal autophagy and longevity in mice. PLoS Genet. 6, e1000838 (2010).
Harrison, D.E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Morselli, E. et al. The life span-prolonging effect of sirtuin-1 is mediated by autophagy. Autophagy 6, 186–188 (2010).
Armour, S.M. et al. Inhibition of mammalian S6 kinase by resveratrol suppresses autophagy. Aging (Albany NY) 1, 515–528 (2009).
Opipari, A.W. Jr. et al. Resveratrol-induced autophagocytosis in ovarian cancer cells. Cancer Res. 64, 696–703 (2004).
Tavernarakis, N., Pasparaki, A., Tasdemir, E., Maiuri, M.C. & Kroemer, G. The effects of p53 on whole organism longevity are mediated by autophagy. Autophagy 4, 870–873 (2008).
Ravikumar, B. et al. Regulation of Mammalian autophagy in physiology and pathophysiology. Physiol. Rev. 90, 1383–1435 (2010).
Thurston, T.L., Ryzhakov, G., Bloor, S., von Muhlinen, N. & Randow, F. The TBK1 adaptor and autophagy receptor NDP52 restricts the proliferation of ubiquitin-coated bacteria. Nat. Immunol. 10, 1215–1221 (2009).
Parkes, M. et al. Sequence variants in the autophagy gene IRGM and multiple other replicating loci contribute to Crohn's disease susceptibility. Nat. Genet. 39, 830–832 (2007).
Rioux, J.D. et al. Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nat. Genet. 39, 596–604 (2007).
Saitoh, T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature 456, 264–268 (2008).
Stappenbeck, T.S. et al. Crohn disease: A current perspective on genetics, autophagy and immunity. Autophagy 7 1–20 (2011).
Mizushima, N. & Hara, T. Intracellular quality control by autophagy: how does autophagy prevent neurodegeneration? Autophagy 2, 302–304 (2006).
García-Arencibia, M., Hochfeld, W.E., Toh, P.P. & Rubinsztein, D.C. Autophagy, a guardian against neurodegeneration. Semin. Cell Dev. Biol. 21, 691–698 (2010).
Ravikumar, B. et al. Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat. Genet. 36, 585–595 (2004).
Zhang, L. et al. Small molecule regulators of autophagy identified by an image-based high-throughput screen. Proc. Natl. Acad. Sci. USA 104, 19023–19028 (2007).
Williams, A. et al. Novel targets for Huntington's disease in an mTOR-independent autophagy pathway. Nat. Chem. Biol. 4, 295–305 (2008).
Rose, C. et al. Rilmenidine attenuates toxicity of polyglutamine expansions in a mouse model of Huntington's disease. Hum. Mol. Genet. 19, 2144–2153 (2010).
Lipinski, M.M. et al. Genome-wide analysis reveals mechanisms modulating autophagy in normal brain aging and in Alzheimer's disease. Proc. Natl. Acad. Sci. USA 107, 14164–14169 (2010).
Mariño, G. et al. Tissue-specific autophagy alterations and increased tumorigenesis in mice deficient in Atg4C/autophagin-3. J. Biol. Chem. 282, 18573–18583 (2007).
Qu, X. et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J. Clin. Invest. 112, 1809–1820 (2003).
Takahashi, Y. et al. Bif-1 interacts with Beclin 1 through UVRAG and regulates autophagy and tumorigenesis. Nat. Cell Biol. 9, 1142–1151 (2007).
White, E., Karp, C., Strohecker, A.M., Guo, Y. & Mathew, R. Role of autophagy in suppression of inflammation and cancer. Curr. Opin. Cell Biol. 22, 212–217 (2010).
Han, J. et al. Involvement of protective autophagy in TRAIL resistance of apoptosis-defective tumor cells. J. Biol. Chem. 283, 19665–19677 (2008).
Colell, A. et al. GAPDH and autophagy preserve survival after apoptotic cytochrome c release in the absence of caspase activation. Cell 129, 983–997 (2007).
Sotelo, J., Briceno, E. & Lopez-Gonzalez, M.A. Adding chloroquine to conventional treatment for glioblastoma multiforme: a randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 144, 337–343 (2006).
Komatsu, M. & Ichimura, Y. Selective autophagy regulates various cellular functions. Genes Cells 15, 923–933 (2010).
Wullschleger, S., Loewith, R. & Hall, M.N. TOR signaling in growth and metabolism. Cell 124, 471–484 (2006).
Sancak, Y. et al. Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141, 290–303 (2010).
Mizushima, N. The role of the Atg1/ULK1 complex in autophagy regulation. Curr. Opin. Cell Biol. 22, 132–139 (2010).
Criollo, A. et al. The IKK complex contributes to the induction of autophagy. EMBO J. 29, 619–631 (2010).
Ravikumar, B., Duden, R. & Rubinsztein, D.C. Aggregate-prone proteins with polyglutamine and polyalanine expansions are degraded by autophagy. Hum. Mol. Genet. 11, 1107–1117 (2002).
Noda, T. & Ohsumi, Y. Tor, a phosphatidylinositol kinase homologue, controls autophagy in yeast. J. Biol. Chem. 273, 3963–3966 (1998).
Kim, D.H. et al. mTOR interacts with raptor to form a nutrient-sensitive complex that signals to the cell growth machinery. Cell 110, 163–175 (2002).
Klionsky, D.J. & Emr, S.D. Autophagy as a regulated pathway of cellular degradation. Science 290, 1717–1721 (2000).
Balgi, A.D. et al. Screen for chemical modulators of autophagy reveals novel therapeutic inhibitors of mTORC1 signaling. PLoS ONE 4, e7124 (2009).
Feldman, M.E. et al. Active-site inhibitors of mTOR target rapamycin-resistant outputs of mTORC1 and mTORC2. PLoS Biol. 7, e38 (2009).
Thoreen, C.C. et al. An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J. Biol. Chem. 284, 8023–8032 (2009).
Cantley, L.C. The phosphoinositide 3-kinase pathway. Science 296, 1655–1657 (2002).
Datta, S.R. et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91, 231–241 (1997).
Backer, J.M. The regulation and function of Class III PI3Ks: novel roles for Vps34. Biochem. J. 410, 1–17 (2008).
Laane, E. et al. Cell death induced by dexamethasone in lymphoid leukemia is mediated through initiation of autophagy. Cell Death Differ. 16, 1018–1029 (2009).
Grandér, D., Kharaziha, P., Laane, E., Pokrovskaja, K. & Panaretakis, T. Autophagy as the main means of cytotoxicity by glucocorticoids in hematological malignancies. Autophagy 5, 1198–1200 (2009).
Alcalay, M. et al. The promyelocytic leukemia gene product (PML) forms stable complexes with the retinoblastoma protein. Mol. Cell. Biol. 18, 1084–1093 (1998).
Blommaart, E.F., Krause, U., Schellens, J.P., Vreeling-Sindelarova, H. & Meijer, A.J. The phosphatidylinositol 3-kinase inhibitors wortmannin and LY294002 inhibit autophagy in isolated rat hepatocytes. Eur. J. Biochem. 243, 240–246 (1997).
Seglen, P.O. & Gordon, P.B. 3-Methyladenine: specific inhibitor of autophagic/lysosomal protein degradation in isolated rat hepatocytes. Proc. Natl. Acad. Sci. USA 79, 1889–1892 (1982).
Raynaud, F.I. et al. Pharmacologic characterization of a potent inhibitor of class I phosphatidylinositide 3-kinases. Cancer Res. 67, 5840–5850 (2007).
Liu, Q., Thoreen, C., Wang, J., Sabatini, D. & Gray, N.S. mTOR mediated anti-cancer drug discovery. Drug Discov. Today Ther. Strateg. 6, 47–55 (2009).
Degtyarev, M. et al. Akt inhibition promotes autophagy and sensitizes PTEN-null tumors to lysosomotropic agents. J. Cell Biol. 183, 101–116 (2008).
Meley, D. et al. AMP-activated protein kinase and the regulation of autophagic proteolysis. J. Biol. Chem. 281, 34870–34879 (2006).
Meijer, A.J. & Codogno, P. AMP-activated protein kinase and autophagy. Autophagy 3, 238–240 (2007).
Williams, R.S., Cheng, L., Mudge, A.W. & Harwood, A.J. A common mechanism of action for three mood-stabilizing drugs. Nature 417, 292–295 (2002).
Sarkar, S. et al. Lithium induces autophagy by inhibiting inositol monophosphatase. J. Cell Biol. 170, 1101–1111 (2005).
Hallcher, L.M. & Sherman, W.R. The effects of lithium ion and other agents on the activity of myo-inositol-1-phosphatase from bovine brain. J. Biol. Chem. 255, 10896–10901 (1980).
Inhorn, R.C. & Majerus, P.W. Properties of inositol polyphosphate 1-phosphatase. J. Biol. Chem. 263, 14559–14565 (1988).
Shaltiel, G. et al. Valproate decreases inositol biosynthesis. Biol. Psychiatry 56, 868–874 (2004).
Xia, H.G. et al. Control of basal autophagy by calpain1 mediated cleavage of ATG5. Autophagy 6, 61–66 (2010).
Roufogalis, B.D., Minocherhomjee, A.M. & Al-Jobore, A. Pharmacological antagonism of calmodulin. Can. J. Biochem. Cell Biol. 61, 927–933 (1983).
Sczekan, S.R. & Strumwasser, F. Antipsychotic drugs block IP3-dependent Ca(2+)-release from rat brain microsomes. Biol. Psychiatry 40, 497–502 (1996).
Crowe, J.H. Trehalose as a “chemical chaperone”: fact and fantasy. Adv. Exp. Med. Biol. 594, 143–158 (2007).
Sarkar, S., Davies, J.E., Huang, Z., Tunnacliffe, A. & Rubinsztein, D.C. Trehalose, a novel mTOR-independent autophagy enhancer, accelerates the clearance of mutant huntingtin and alpha-synuclein. J. Biol. Chem. 282, 5641–5652 (2007).
Rodríguez-Navarro, J.A. et al. Trehalose ameliorates dopaminergic and tau pathology in parkin deleted/tau overexpressing mice through autophagy activation. Neurobiol. Dis. 39, 423–438 (2010).
Sarkar, S. & Rubinsztein, D.C. Small molecule enhancers of autophagy for neurodegenerative diseases. Mol. Biosyst. 4, 895–901 (2008).
Bommareddy, A. et al. Atg5 regulates phenethyl isothiocyanate-induced autophagic and apoptotic cell death in human prostate cancer cells. Cancer Res. 69, 3704–3712 (2009).
Lefranc, F., Facchini, V. & Kiss, R. Proautophagic drugs: a novel means to combat apoptosis-resistant cancers, with a special emphasis on glioblastomas. Oncologist 12, 1395–1403 (2007).
Sarkar, S. et al. A rational mechanism for combination treatment of Huntington's disease using lithium and rapamycin. Hum. Mol. Genet. 17, 170–178 (2008).
Miller, S. et al. Shaping development of autophagy inhibitors with the structure of the lipid kinase Vps34. Science 327, 1638–1642 (2010).
Lu, X. et al. Designed semisynthetic protein inhibitors of Ub/Ubl E1 activating enzymes. J. Am. Chem. Soc. 132, 1748–1749 (2010).
Kumanomidou, T. et al. The crystal structure of human Atg4b, a processing and de-conjugating enzyme for autophagosome-forming modifiers. J. Mol. Biol. 355, 612–618 (2006).
Sugawara, K. et al. Structural basis for the specificity and catalysis of human Atg4B responsible for mammalian autophagy. J. Biol. Chem. 280, 40058–40065 (2005).
Hopkins, A.L. & Groom, C.R. The druggable genome. Nat. Rev. Drug Discov. 1, 727–730 (2002).
Overington, J.P., Al-Lazikani, B. & Hopkins, A.L. How many drug targets are there? Nat. Rev. Drug Discov. 5, 993–996 (2006).
Acknowledgements
We are grateful for funding from the Medical Research Council (Programme Grant to D.C.R.; Skills Gap Award to A.F.), the Wellcome Trust (Senior Fellowship to D.C.R.), the UK National Institute for Health Research, Biomedical Research Centre at Addenbrooke's Hospital, Special Coordination Funds for Promoting Science and Technology of the Ministry of Education, Culture, Sports, Science and Technology of Japan, and the Takeda Science Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
David Rubinsztein is an inventor on patents describing autophagy-inducing drugs as potential therapeutics.
Rights and permissions
About this article
Cite this article
Fleming, A., Noda, T., Yoshimori, T. et al. Chemical modulators of autophagy as biological probes and potential therapeutics. Nat Chem Biol 7, 9–17 (2011). https://doi.org/10.1038/nchembio.500
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.500
This article is cited by
-
Metformin and Trehalose-Modulated Autophagy Exerts a Neurotherapeutic Effect on Parkinsonʼs Disease
Molecular Neurobiology (2023)
-
Autophagy Contributes to Oxidative Stress-Induced Apoptosis in Porcine Granulosa Cells
Reproductive Sciences (2021)
-
Autophagy: a promising therapeutic target for improving mesenchymal stem cell biological functions
Molecular and Cellular Biochemistry (2021)
-
Endocytic regulation of cellular ion homeostasis controls lysosome biogenesis
Nature Cell Biology (2020)
-
Effects of Cadmium and/or Lead on Autophagy and Liver Injury in Rats
Biological Trace Element Research (2020)