Abstract
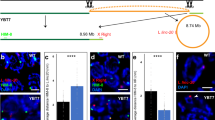
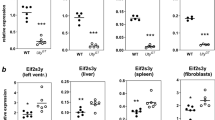
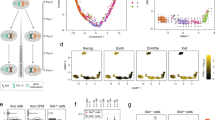
According to the prevailing view, mammalian X chromosomes are enriched in spermatogenesis genes expressed before meiosis1,2,3 and deficient in spermatogenesis genes expressed after meiosis2,3. The paucity of postmeiotic genes on the X chromosome has been interpreted as a consequence of meiotic sex chromosome inactivation (MSCI)—the complete silencing of genes on the XY bivalent at meiotic prophase4,5. Recent studies have concluded that MSCI-initiated silencing persists beyond meiosis6,7,8 and that most genes on the X chromosome remain repressed in round spermatids7. Here, we report that 33 multicopy gene families, representing ∼273 mouse X-linked genes, are expressed in the testis and that this expression is predominantly in postmeiotic cells. RNA FISH and microarray analysis show that the maintenance of X chromosome postmeiotic repression is incomplete. Furthermore, X-linked multicopy genes exhibit a similar degree of expression as autosomal genes. Thus, not only is the mouse X chromosome enriched for spermatogenesis genes functioning before meiosis, but in addition, ∼18% of mouse X-linked genes are expressed in postmeiotic cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Wang, P.J., McCarrey, J.R., Yang, F. & Page, D.C. An abundance of X-linked genes expressed in spermatogonia. Nat. Genet. 27, 422–426 (2001).
Khil, P.P., Smirnova, N.A., Romanienko, P.J. & Camerini-Otero, R.D. The mouse X chromosome is enriched for sex-biased genes not subject to selection by meiotic sex chromosome inactivation. Nat. Genet. 36, 642–646 (2004).
Reinke, V. Sex and the genome. Nat. Genet. 36, 548–549 (2004).
McKee, B.D. & Handel, M.A. Sex chromosomes, recombination, and chromatin conformation. Chromosoma 102, 71–80 (1993).
Turner, J.M. Meiotic sex chromosome inactivation. Development 134, 1823–1831 (2007).
Greaves, I.K., Rangasamy, D., Devoy, M., Marshall Graves, J.A. & Tremethick, D.J. The X and Y chromosomes assemble into H2A.Z-containing facultative heterochromatin following meiosis. Mol. Cell. Biol. 26, 5394–5405 (2006).
Namekawa, S.H. et al. Postmeiotic sex chromatin in the male germline of mice. Curr. Biol. 16, 660–667 (2006).
Turner, J.M., Mahadevaiah, S.K., Ellis, P.J., Mitchell, M.J. & Burgoyne, P.S. Pachytene asynapsis drives meiotic sex chromosome inactivation and leads to substantial postmeiotic repression in spermatids. Dev. Cell 10, 521–529 (2006).
Warburton, P.E., Giordano, J., Cheung, F., Gelfand, Y. & Benson, G. Inverted repeat structure of the human genome: the X-chromosome contains a preponderance of large, highly homologous inverted repeats that contain testes genes. Genome Res. 14, 1861–1869 (2004).
Eichler, E.E., Clark, R.A. & She, X. An assessment of the sequence gaps: unfinished business in a finished human genome. Nat. Rev. Genet. 5, 345–354 (2004).
Mroz, K., Carrel, L. & Hunt, P.A. Germ cell development in the XXY mouse: evidence that X chromosome reactivation is independent of sexual differentiation. Dev. Biol. 207, 229–238 (1999).
Mazeyrat, S. et al. A Y-encoded subunit of the translation initiation factor Eif2 is essential for mouse spermatogenesis. Nat. Genet. 29, 49–53 (2001).
Rice, W.R. Sex chromosomes and the evolution of sexual dimorphism. Evolution Int. J. Org. Evolution 38, 735–742 (1984).
Kimura, T. et al. Mouse germ cell-less as an essential component for nuclear integrity. Mol. Cell. Biol. 23, 1304–1315 (2003).
Govin, J. et al. Pericentric heterochromatin reprogramming by new histone variants during mouse spermiogenesis. J. Cell Biol. 176, 283–294 (2007).
Wang, P.J., Page, D.C. & McCarrey, J.R. Differential expression of sex-linked and autosomal germ-cell-specific genes during spermatogenesis in the mouse. Hum. Mol. Genet. 14, 2911–2918 (2005).
Westbrook, V.A. et al. Spermatid-specific expression of the novel X-linked gene product SPAN-X localized to the nucleus of human spermatozoa. Biol. Reprod. 63, 469–481 (2000).
Westbrook, V.A. et al. Hominoid-specific SPANXA/D genes demonstrate differential expression in individuals and protein localization to a distinct nuclear envelope domain during spermatid morphogenesis. Mol. Hum. Reprod. 12, 703–716 (2006).
Kuroda-Kawaguchi, T. et al. The AZFc region of the Y chromosome features massive palindromes and uniform recurrent deletions in infertile men. Nat. Genet. 29, 279–286 (2001).
Waterston, R.H. et al. Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562 (2002).
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. & Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Acknowledgements
We thank J. Alfoldi, D. Bellott, P. Burgoyne, H. Byers, J. Hughes, J. Lange, L. Reynard, S. Rozen and H. Skaletsky for helpful comments on the manuscript; D. Bellott, M. Gill, Y. Hu, J. Hughes and H. Skaletsky for technical advice; and P. Burgoyne and M. Mitchell for the Uty transgenic line. This work was supported by the Medical Research Council, US National Institutes of Health grant HD000257 (to D.C.P.), including fellowship F32HD052379 (to J.L.M.), and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
J.L.M., J.M.A.T., S.K.M. and P.E.W. identified amplicons and multicopy genes. J.L.M., J.M.A.T. and S.K.M. carried out RT-PCRs and RNA FISH. J.L.M. and P.J.P. did microarray and statistical analysis. The paper was written by J.L.M., J.M.A.T. and D.C.P.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Tables 1–5 (PDF 1283 kb)
Rights and permissions
About this article
Cite this article
Mueller, J., Mahadevaiah, S., Park, P. et al. The mouse X chromosome is enriched for multicopy testis genes showing postmeiotic expression. Nat Genet 40, 794–799 (2008). https://doi.org/10.1038/ng.126
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.126
This article is cited by
-
Characterization of repetitive DNA on the genome of the marsh rat Holochilus nanus (Cricetidae: Sigmodontinae)
Molecular Genetics and Genomics (2023)
-
Active DNA damage response signaling initiates and maintains meiotic sex chromosome inactivation
Nature Communications (2022)
-
X-linked palindromic gene families 4930567H17Rik and Mageb5 are dispensable for male mouse fertility
Scientific Reports (2022)
-
Meiotic drive in house mice: mechanisms, consequences, and insights for human biology
Chromosome Research (2022)
-
The X chromosome and male infertility
Human Genetics (2021)