Abstract
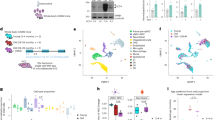
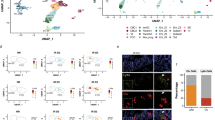
Restoration of regulated insulin secretion is the ultimate goal of therapy for type 1 diabetes. Here, we show that, unexpectedly, somatic ablation of Foxo1 in Neurog3+ enteroendocrine progenitor cells gives rise to gut insulin-positive (Ins+) cells that express markers of mature β cells and secrete bioactive insulin as well as C-peptide in response to glucose and sulfonylureas. Lineage tracing experiments showed that gut Ins+ cells arise cell autonomously from Foxo1-deficient cells. Inducible Foxo1 ablation in adult mice also resulted in the generation of gut Ins+ cells. Following ablation by the β-cell toxin streptozotocin, gut Ins+ cells regenerate and produce insulin, reversing hyperglycemia in mice. The data indicate that Neurog3+ enteroendocrine progenitors require active Foxo1 to prevent differentiation into Ins+ cells. Foxo1 ablation in gut epithelium may provide an approach to restore insulin production in type 1 diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Zhou, Q. & Melton, D.A. Pathways to new β cells. Cold Spring Harb. Symp. Quant. Biol. 73, 175–181 (2008).
Kroon, E. et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat. Biotechnol. 26, 443–452 (2008).
Bonal, C. & Herrera, P.L. Genes controlling pancreas ontogeny. Int. J. Dev. Biol. 52, 823–835 (2008).
Gradwohl, G., Dierich, A., LeMeur, M. & Guillemot, F. neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas. Proc. Natl. Acad. Sci. USA 97, 1607–1611 (2000).
Jensen, J. et al. Control of endodermal endocrine development by Hes-1. Nat. Genet. 24, 36–44 (2000).
Schwitzgebel, V.M. et al. Expression of neurogenin3 reveals an islet cell precursor population in the pancreas. Development 127, 3533–3542 (2000).
Lee, C.S., Perreault, N., Brestelli, J.E. & Kaestner, K.H. Neurogenin 3 is essential for the proper specification of gastric enteroendocrine cells and the maintenance of gastric epithelial cell identity. Genes Dev. 16, 1488–1497 (2002).
Schonhoff, S.E., Giel-Moloney, M. & Leiter, A.B. Neurogenin 3–expressing progenitor cells in the gastrointestinal tract differentiate into both endocrine and non-endocrine cell types. Dev. Biol. 270, 443–454 (2004).
Schonhoff, S.E., Giel-Moloney, M. & Leiter, A.B. Minireview: development and differentiation of gut endocrine cells. Endocrinology 145, 2639–2644 (2004).
Gu, G., Dubauskaite, J. & Melton, D.A. Direct evidence for the pancreatic lineage: NGN3+ cells are islet progenitors and are distinct from duct progenitors. Development 129, 2447–2457 (2002).
Xu, X. et al. β cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell 132, 197–207 (2008).
Hunt, R.K. & Jacobson, M. Specification of positional information in retinal ganglion cells of Xenopus: stability of the specified state. Proc. Natl. Acad. Sci. USA 69, 2860–2864 (1972).
Accili, D. & Arden, K.C. FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell 117, 421–426 (2004).
Hribal, M.L., Nakae, J., Kitamura, T., Shutter, J.R. & Accili, D. Regulation of insulin-like growth factor–dependent myoblast differentiation by Foxo forkhead transcription factors. J. Cell Biol. 162, 535–541 (2003).
Nakae, J. et al. The forkhead transcription factor Foxo1 regulates adipocyte differentiation. Dev. Cell 4, 119–129 (2003).
Paik, J.H. et al. FoxOs cooperatively regulate diverse pathways governing neural stem cell homeostasis. Cell Stem Cell 5, 540–553 (2009).
Kitamura, T. et al. A Foxo/Notch pathway controls myogenic differentiation and fiber type specification. J. Clin. Invest. 117, 2477–2485 (2007).
Kitamura, T. et al. Regulation of pancreatic juxtaductal endocrine cell formation by FoxO1. Mol. Cell. Biol. 29, 4417–4430 (2009).
Kitamura, T. et al. The forkhead transcription factor Foxo1 links insulin signaling to Pdx1 regulation of pancreatic β cell growth. J. Clin. Invest. 110, 1839–1847 (2002).
Okamoto, H. et al. Role of the forkhead protein FoxO1 in β cell compensation to insulin resistance. J. Clin. Invest. 116, 775–782 (2006).
Kawamori, D. et al. The forkhead transcription factor Foxo1 bridges the JNK pathway and the transcription factor PDX-1 through its intracellular translocation. J. Biol. Chem. 281, 1091–1098 (2006).
Kitamura, Y.I. et al. FoxO1 protects against pancreatic β cell failure through NeuroD and MafA induction. Cell Metab. 2, 153–163 (2005).
Al-Masri, M. et al. Effect of forkhead box O1 (FOXO1) on β cell development in the human fetal pancreas. Diabetologia 53, 699–711 (2010).
Lee, J.C. et al. Regulation of the pancreatic pro-endocrine gene neurogenin3. Diabetes 50, 928–936 (2001).
Fukuda, A. et al. Ectopic pancreas formation in Hes1-knockout mice reveals plasticity of endodermal progenitors of the gut, bile duct, and pancreas. J. Clin. Invest. 116, 1484–1493 (2006).
Kageyama, R., Ohtsuka, T. & Tomita, K. The bHLH gene Hes1 regulates differentiation of multiple cell types. Mol. Cells 10, 1–7 (2000).
van der Flier, L.G. & Clevers, H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu. Rev. Physiol. 71, 241–260 (2009).
Hingorani, S.R. et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell 4, 437–450 (2003).
Fujita, Y. et al. Glucose-dependent insulinotropic polypeptide is expressed in pancreatic islet α-cells and promotes insulin secretion. Gastroenterology 138, 1966–1975 (2010).
Wang, S. et al. Sustained Neurog3 expression in hormone-expressing islet cells is required for endocrine maturation and function. Proc. Natl. Acad. Sci. USA 106, 9715–9720 (2009).
Tuttle, R.L. et al. Regulation of pancreatic (-cell growth and survival by the serine/threonine protein kinase Akt1/PKBα. Nat. Med. 7, 1133–1137 (2001).
Nielsen, K. et al. β-cell maturation leads to in vitro sensitivity to cytotoxins. Diabetes 48, 2324–2332 (1999).
Sommer, L., Ma, Q. & Anderson, D.J. neurogenins, a novel family of atonal-related bHLH transcription factors, are putative mammalian neuronal determination genes that reveal progenitor cell heterogeneity in the developing CNS and PNS. Mol. Cell. Neurosci. 8, 221–241 (1996).
Gershengorn, M.C. et al. Epithelial-to-mesenchymal transition generates proliferative human islet precursor cells. Science 306, 2261–2264 (2004).
Naya, F.J. et al. Diabetes, defective pancreatic morphogenesis, and abnormal enteroendocrine differentiation in BETA2/neuroD-deficient mice. Genes Dev. 11, 2323–2334 (1997).
Gao, N., White, P. & Kaestner, K.H. Establishment of intestinal identity and epithelial-mesenchymal signaling by Cdx2. Dev. Cell 16, 588–599 (2009).
Nakamura, T., Tsuchiya, K. & Watanabe, M. Crosstalk between Wnt and Notch signaling in intestinal epithelial cell fate decision. J. Gastroenterol. 42, 705–710 (2007).
Essers, M.A. et al. Functional interaction between β-catenin and FOXO in oxidative stress signaling. Science 308, 1181–1184 (2005).
Sewalt, R.G., Gunster, M.J., van der Vlag, J., Satijn, D.P. & Otte, A.P. C-terminal binding protein is a transcriptional repressor that interacts with a specific class of vertebrate Polycomb proteins. Mol. Cell. Biol. 19, 777–787 (1999).
Hoffman, B.G., Zavaglia, B., Beach, M. & Helgason, C.D. Expression of Groucho/TLE proteins during pancreas development. BMC Dev. Biol. 8, 81 (2008).
Muhr, J., Andersson, E., Persson, M., Jessell, T.M. & Ericson, J. Groucho-mediated transcriptional repression establishes progenitor cell pattern and neuronal fate in the ventral neural tube. Cell 104, 861–873 (2001).
Sonoshita, M. et al. Suppression of colon cancer metastasis by Aes through inhibition of Notch signaling. Cancer Cell 19, 125–137 (2011).
Cheung, A.T. et al. Glucose-dependent insulin release from genetically engineered K cells. Science 290, 1959–1962 (2000).
Nielsen, L.B. et al. Co-localisation of the Kir6.2/SUR1 channel complex with glucagon-like peptide–1 and glucose-dependent insulinotrophic polypeptide expression in human ileal cells and implications for glycaemic control in new onset type 1 diabetes. Eur. J. Endocrinol. 156, 663–671 (2007).
D'Amour, K.A. et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 24, 1392–1401 (2006).
Mundell, N.A. & Labosky, P.A. Neural crest stem cell multipotency requires Foxd3 to maintain neural potential and repress mesenchymal fates. Development 138, 641–652 (2011).
Uhlenhaut, N.H. et al. Somatic sex reprogramming of adult ovaries to testes by FOXL2 ablation. Cell 139, 1130–1142 (2009).
Bjerknes, M. & Cheng, H. Neurogenin 3 and the enteroendocrine cell lineage in the adult mouse small intestinal epithelium. Dev. Biol. 300, 722–735 (2006).
Drucker, D.J. The biology of incretin hormones. Cell Metab. 3, 153–165 (2006).
Ridgway, J. et al. Inhibition of Dll4 signalling inhibits tumour growth by deregulating angiogenesis. Nature 444, 1083–1087 (2006).
Paik, J.H. et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 128, 309–323 (2007).
Okamoto, H. et al. Transgenic rescue of insulin receptor–deficient mice. J. Clin. Invest. 114, 214–223 (2004).
Sherman, B.M., Gorden, P., Roth, J. & Freychet, P. Circulating insulin: the proinsulin-like properties of “big” insulin in patients without islet cell tumors. J. Clin. Invest. 50, 849–858 (1971).
Golaz, J.L., Vonlaufen, N., Hemphill, A. & Burgener, I.A. Establishment and characterization of a primary canine duodenal epithelial cell culture. In Vitro Cell. Dev. Biol. Anim. 43, 176–185 (2007).
Gao, N. et al. Foxa2 controls vesicle docking and insulin secretion in mature β cells. Cell Metab. 6, 267–279 (2007).
Suzuki, A., Nakauchi, H. & Taniguchi, H. Glucagon-like peptide 1 (1–37) converts intestinal epithelial cells into insulin-producing cells. Proc. Natl. Acad. Sci. USA 100, 5034–5039 (2003).
Acknowledgements
We thank A. Efstratiadis (Biomedical Research Foundation, Academy of Athens, Athens, Greece) for Ins2-Gfp knock-in mice and A. Leiter (University of Massachusetts) for Neurog3-Cre mice. We thank L.S., J.Y.K.-M. and T.M. for advice and reagents and Q.X. for technical support. We also thank members of the Accili laboratory for insightful discussion and critical reading of the manuscript. This work was supported by grants from the US National Institutes of Health (DK58282 and DK64819), the Columbia University Diabetes Research Center (DK63608), the Druckenmiller Fellowship of the New York Stem Cell Foundation, the Brehm Coalition and the Russell Berrie Foundation.
Author information
Authors and Affiliations
Contributions
C.T. generated mice, designed and performed experiments and wrote the manuscript. S.X. generated mice and designed experiments. T.K. generated mice and performed immunohistochemistry. R.A.D. and D.A. designed experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 5241 kb)
Rights and permissions
About this article
Cite this article
Talchai, C., Xuan, S., Kitamura, T. et al. Generation of functional insulin-producing cells in the gut by Foxo1 ablation. Nat Genet 44, 406–412 (2012). https://doi.org/10.1038/ng.2215
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2215
This article is cited by
-
Stomach-derived human insulin-secreting organoids restore glucose homeostasis
Nature Cell Biology (2023)
-
MYCL-mediated reprogramming expands pancreatic insulin-producing cells
Nature Metabolism (2022)
-
A hierarchical transcription factor cascade regulates enteroendocrine cell diversity and plasticity in Drosophila
Nature Communications (2022)
-
Therapeutic strategies targeting FOXO transcription factors
Nature Reviews Drug Discovery (2021)
-
Insulin is expressed by enteroendocrine cells during human fetal development
Nature Medicine (2021)