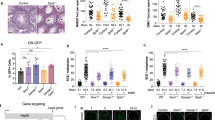
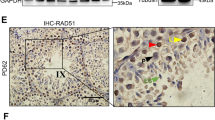
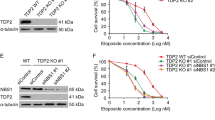
Abstract
Mice that are deficient in either the Pms2 or Msh2 DNA mismatch repair genes have microsatellite instability and a predisposition to tumours. Interestingly, Pms2–deficient males display sterility associated with abnormal chromosome pairing in meiosis. Here mice deficient in another mismatch repair gene, Mlh1, possess not only microsatellite instability but are also infertile (both males and females). Mlh 1 –deficient spermatocytes exhibit high levels of prematurely separated chromosomes and arrest in first division meiosis. We also show that Mlh1 appears to localize to sites of crossing over on meiotic chromosomes/Together these findings suggest that Mlh1 is involved in DNA mismatch repair and meiotic crossing over.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Modrich, P. Mechanisms and biological effects of mismatch repair. Annu. Rev. Genet. 25, 229–253 (1991).
Au, K.G., Welsh, K. & Modrich, P. Initiation of methyl-directed mismatch repair. J. Biol. Chem. 267, 12142–12148 (1992).
Reenan, R.A.G. & Kolodner, R.D. Isolation and characterization of two Saccharomyces cerevisiae genes encoding homologs of the bacterial HexA and MutS mismatch repair proteins. Genetics 132, 963–973 (1992).
Kramer, W., Kramer, B., Williamson, M.S. & Fogel, S. Cloning and nucleotide sequence of DNA mismatch repair gene PMS1 from Saccharomyces cerevisiae: homology of PMS1 to procaryotic MutL and HexB. J. Bacteriol. 171, 5339–5346 (1989).
Prolla, T., Christie, D.-M. & Liskay, R.M. Dual requirement in yeast DNA mismatch repair for MLH1 and PMS1, two homologs of the bacterial mutL gene. Mol. Cell. Biol. 14, 407–415 (1994).
Bronner, C.E. et al. Mutation in the DNA mismatch repair gene homologue hMLH1 is associated with hereditary nonpolyposis colon cancer. Nature 368, 258–261 (1993).
Fishel, R.A. et al. The human mutator gene homolog MSH2 and its association with hereditary nonpolyposis colon cancer. Cell 75, 1027–1038 (1993).
Leach, F.S. et al. Mutations of a mutS homolog in hereditary nonpolyposis colorectal cancer. Cell 75, 1215–1225 (1993).
Papadopoulos, N. et al. Mutation of a mutL homolog in hereditary colon cancer. Science 263, 1625–1629 (1994).
Nicolaides, N.C. et al. Mutations of two PMS homologues in hereditary nonpolyposis colon cancer. Nature 371, 75–60 (1994).
New, L., Liu, K. & Grouse, G.F. The yeast gene MSH3 defines a new class of eukaryotic MutS homologues. Mol. Gen. Genet. 239, 97–108 (1993).
Marsischky, G.T., Filosi, N., Kane, M.F. & Kolodner, R. Redundancy of Saccharomyces cerevisiae MSH3 and MSH6 in MSH2-dependent mismatch repair. Genes Dev. 10, 407–420 (1996).
Bishop, D.K., Andersen, J. & Kolodner, R.D. Specificity of mismatch repair following transformation of Saccharomyces cerevisiae with heteroduplex plasmid DNA. Proc. Natl. Acad. Sci. USA 86, 3713–3717 (1989).
Williamson, M.S., Game, J.C. & Fogel, S. Metotic gene conversion mutants in Saccharomyces cerevisiae. I. Isolation and characterization of pms1-1 and pms1-2. Genetics 110, 609–646 (1985).
Prolla, T.A., Pang, Q., Alani, E., Kolodner, R.D. & Liskay, R.M. Interactions between the MSH2, MLH1 and PMS1 proteins during the initiation of DNA mismatch repair. Science 265, 1091–1093 (1994).
Baker, S.M. et al. Male mice defective in the DNA mismatch repair gene PMS2 exhibit abnormal chromosome synapsis in meiosis. Cell 82, 309–319 (1995).
Reitmair, A.H. et al. MSH2 deficient mice are viable and susceptible to lymphoid tumours. Nature Genet. 11, 64–70 (1995).
de Wind, N., Decker, M., Bems, A., Radman, M. & te Riele, H. Inactivation of the mouse MSH2 gene results in postreplication mismatch repair deficiency, methylation tolerance, hyperrecombination and predisposition to tumorigenesis. Cell 82, 321–330 (1995).
Fishel, R., Ewel, A. & Lescoe, M.K. Purified human MSH2 protein binds to DNA containing mismatched nucleotides. Cancer Res. 54, 5539–5542 (1994).
Li, G.-M. & Modrich, P. Restoration of mismatch repair to nuclear extracts of H6 colorectal tumor cells by a heterodimer of human MutL homologs. Proc. Natl. Acad. Sci. USA 92, 1950–1954 (1995).
Petes, T.D., Malone, R.E. & Symington, L.S. Recombination in yeast. in The Molecular and Cellular Biology of the Yeast Saccharomyces vol. 1, (eds Broach, J., Jones, E. & Pringle, J.) 407–521 (Cold Spring Harbor Laboratory, Cold Spring Harbor, 1991).
Detloff, P., White, M.A. & Petes, T.D. Analysis of a gene conversion gradient at the HIS4locus in Saccharomyces cerevisiae. Genetics 132, 113–123 (1992).
Alani, E., Reenan, R.A.G. & Kolodner, R. Interaction between mismatch repair and genetic recombination in Saccharomyces cerevisiae. Genetics 137, 19–39 (1994).
Worth, L. Jr., Clark, S., Radman, M. & Modrich, P. Mismatch repair proteins MutS and MutL inhibit RecA-catalyzed strand transfer between diverged DMAs. Proc. Natl. Acad. Sci. USA 91, 3238–3241 (1994).
Datta, A., Adjiri, A., New, L., Grouse, G.F. & Jinks-Robertson, S. Mrtotic crossovers between diverged sequences are regulated by mismatch repair proteins in Saccharomyces cerevisiae. Mol. Cell. Biol. 16, 1085–1093 (1996).
Petit, M.-A., Dimpfl, J., Radman, M. & Echols, H. Control of large chromosomal duplications in Escherichia coli by the mismatch repair system. Genetics 129, 327–332 (1991).
Selva, E.M., New, L., Grouse, G.F. & Lahue, R.S. Mismatch correction acts as a barrier to homeologous recombination in Saccharomyces cerevisiae. Genetics 139, 1175–1188 (1995).
Hunter, N., Chambers, S.R., Louis, E.J. & Borts, R.H. The mismatch repair system contributes to meiotic sterility in an interspecific yeast hybrid. EMBO J. 15, 1726–1733 (1996).
Ross-Macdonald, P. & Roeder, G.S. Mutation of a meiosis-specific MutS homolog decreases crossing over but not mismatch correction. Cell 79, 1069–1080 (1994).
Hollingsworth, N.M., Ponte, L. & Halsey, C. MSH5, a novel MutS homolog, facilitates meiotic reciprocal recombination between homologs in Saccharomyces cerevisiae but not mismatch repair. Genes Dev. 9, 1728–1739 (1995).
Darlington, C.D. in Recent Advances in Cytology.(Churchill, London, 1937).
Hawley, R.S. Exchange and chromosomal segregation in eukaryotes. in Meiosis (eds Moens, RB.) 497–527 (Academic Press, New York, 1987).
Carpenter, A.T. Gene conversion, recombination nodules, and the initiation of meiotic synapsis. Bioessays 6, 232–236 (1987).
Ashley, T. Mammalian meiotic recombination: a reexamination. Hum. Genet. 94, 587–593 (1994).
Leeflang, R.E., Hubert, R., Zhang, L. & Arnheim, N. Single sperm typing. in Current Protocols in Human Genetics vol 1. (eds Dracopoli, N.C. et al.) 1.6.1–1.6.15 (John Wiley and Sons, New York, 1994).
Ashley, T. et al. Dynamic changes in Rad51 distribution on chromatin during meiosis in male and female vertebrates. Chromosome 104, 19–28 (1995).
Burgoyne, P.S. Genetic homology and crossing over in the X and Y chromosomes of mammals. Hum. Genet. 61, 85–90 (1982).
Rouyer, F. et al. A gradient of sex linkage in the pseudoautosomal region of the human sex chromosomes. Nature 319, 291–295 (1986).
Petit, C., Levillers, J. & Weissenbach, J. Physical mapping of the pseudoautosomal region: comparison with genetic linkage map. EMBO J. 7, 2369–2379 (1988).
Rappold, G.A. & Lehrach, H. A long range restriction map of the pseudoautosomal region by partial digest PFGE analysis from the telomere. Nud. Acids Res. 16, 5361–5377 (1988).
Soriano, P. et al. High rate of recombination and double crossovers in the mouse pseudoautosomal region during male meiosis. Proc. Natl. Acad. Sci. USA 84, 7218–7220 (1987).
Polani, R.E. & Jagiello, G.M., smata, meiotic univalents, and age in relation to aneuploid imbalance in mice. Cytogenet. Cell Genet. 16, 505–529 (1976).
Moses, M.J. & Poorman, P.A., Synapsis, synaptic adjustment and DNA synthesis in mouse oocytes. in Chromosomes Today vol. 8. (eds Bennett, M.D., Gropp, A. & Wolf, U.) 90–103 (George Alien & Unwin, Boston, 1984).
Moses, M.J. Composition and role of the synaptonemal complex. Symp. Soc. Exp. Biol. 38, 245–270 (1984).
Solari, A.J. Sex chromosome pairing and fertility in the heterogametic sex of mammals and birds. in Fertility and Chromosome Pairing: Recent Studies in Plants and Animals(eds Gillies, C. B.) 77–135 (CRC Press, Boca Raton, 1989).
Moses, M.J. Synaptonemal complex karyotyping in spermatocytes of the Chinese hamster (Cricetulus). II. Morphology of the XY pair in spread preparations. Chromosoma 60, 127–137 (1977).
Dietrich, A.J.J. & Mulder, P.J.P. A light and electron microscopic analysis of meiotic prophase in female mice. Chromosoma 88, 377–385 (1983).
Dietrich, A.J.J. & Mulder, P.J.P. A light microscopic study of the development and behaviour of the synaptonemal complex in spermatocytes of the mouse. Chromosoma 83, 409–418 (1981).
Goetz, R., Chandley, A.C. & Speed, R.M. Morphological and temporal sequence of meiotic prophase development at puberty in the male mouse. J. Cell Science 65, 249–263 (1984).
Speed, R.M. Meiosis in the foetal mouse ovary. I. An analysis at the light microscope level using surface spreading. Chromosoma 85, 427–437 (1982).
Roeder, G.S. Chromosome synapsis and genetic recombination: their roles in meiotic chromosome segregation. Trends Genet. 6, 385–389 (1990).
West, S.C. Enzymes and molecular mechanisms of genetic recombination. Annu. Rev. Biochem. 61, 603–640 (1992).
Jones, S.N., Roe, A.E., Donehower, L.A. & Bradley, A. Rescue of embryonic lethality in Mdm2-deficient mice by absence of p53. Nature 378, 206–208 (1995).
Ramirez-Solis, R., Davis, A.C. & Bradley, A. Gene targeting in mouse embryonic stem cells. in Guide to Techniques in Mouse Development (eds Wassarman, P. M. & DePamhilis, M.L) 855–878 (Academic, New York, 1993).
Bradley, A. Production and analysis of chimaeric mice. in Teratocarcinomas and Embryonic Stem Cells: A Practical Approach.(eds Robertson, E. J.) 113–151 (IRL, Oxford, 1987).
McMahon, A.R. & Bradley, A., Wnt-1 (int-1) proto-oncogene is required for development of a large region of the mouse brain. Cell 62, 1073–1085 (1990).
Haaf, T. et al. Nuclear foci of mammalian Rad51 recombination protein in somatic cells after DNA damage and it's localization in synaptonemal complexes. Proc. Natl. Acad. Sci. USA 92, 2298–2302 (1995).
Galfre, G. et al. Antibodies to major histocompatibility antigens produced by hybrid cell lines. Nature 266, 550–552 (1977).
de St. Groth, S.F. Monoclonal antibodies and how to make them. Transplant. Proc. 12, 447–450 (1980).
Moens, R.B. et al. Synaptonemal complex antigen localization and conservation. J. Cell Bid. 105, 93–103 (1987).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baker, S., Plug, A., Prolla, T. et al. Involvement of mouse Mlh1 in DNA mismatch repair and meiotic crossing over. Nat Genet 13, 336–342 (1996). https://doi.org/10.1038/ng0796-336
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0796-336
This article is cited by
-
Seeding the meiotic DNA break machinery and initiating recombination on chromosome axes
Nature Communications (2024)
-
DNA double-strand break genetic variants in patients with premature ovarian insufficiency
Journal of Ovarian Research (2023)
-
FIGNL1 AAA+ ATPase remodels RAD51 and DMC1 filaments in pre-meiotic DNA replication and meiotic recombination
Nature Communications (2023)
-
A novel recombination protein C12ORF40/REDIC1 is required for meiotic crossover formation
Cell Discovery (2023)
-
N-acetylcysteine rescues meiotic arrest during spermatogenesis in mice exposed to BDE-209
Environmental Science and Pollution Research (2023)