Abstract
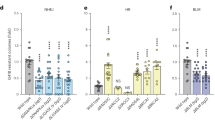
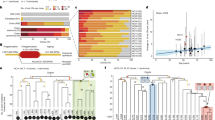
The L1 retrotransposon has had an immense impact on the size and structure of the human genome through a variety of mechanisms, including insertional mutagenesis1,2. To study retrotransposition in a living organism, we created a mouse model of human L1 retrotransposition. Here we show that L1 elements can retrotranspose in male germ cells, and that expression of a human L1 element under the control of its endogenous promoter is restricted to testis and ovary. In the mouse line with the highest level of L1 expression, we found two de novo L1 insertions in 135 offspring. Both insertions were structurally indistinguishable from natural endogenous insertions. This suggests that an individual L1 element can have substantial mutagenic potential. In addition to providing a valuable in vivo model of retrotransposition in mammals, these mice are an important step in the development of a new random mutagenesis system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
The International Human Genome Sequencing Consortium. Nature 409, 860–921 (2001).
Ostertag, E.M. & Kazazian, H.H. Jr. Biology of mammalian L1 retrotransposons. Ann. Rev. Genet. 35, 501–538 (2001).
Sassaman, D.M. et al. Many human L1 elements are capable of retrotransposition. Nature Genet. 16, 37–43 (1997).
Moran, J.V. et al. High frequency retrotransposition in cultured mammalian cells. Cell 87, 917–927 (1996).
Branciforte, D. & Martin, S.L. Developmental and cell type specificity of LINE-1 expression in mouse testis: implications for transposition. Mol. Cell. Biol. 14, 2584–2592 (1994).
Trelogan, S.A. & Martin, S.L. Tightly regulated, developmentally specific expression of the first open reading frame from LINE-1 during mouse embryogenesis. Proc. Natl Acad. Sci. USA 92, 1520–1524 (1995).
Nakanishi, T. et al. Real-time observation of acrosomal dispersal from mouse sperm using GFP as a marker protein. FEBS Lett. 449, 277–283 (1999).
Ostertag, E.M. et al. Determination of L1 retrotransposition kinetics in cultured cells. Nucleic Acids Res. 28, 1418–1423 (2000).
Schwahn, U. et al. Positional cloning of the gene for X-linked retinitis pigmentosa 2. Nature Genet. 19, 327–332 (1998).
Kimberland, M.L. et al. Full-length human L1 insertions retain the capacity for high-frequency retrotransposition in cultured cells. Hum. Mol. Genet. 8, 1557–1560 (1999).
Romrell, L.J., Bellvé, A.R. & Fawcett, D.W. Separation of mouse spermatogenic cells by sedimentation velocity. A morphological characterization. Dev. Biol. 49, 119–131 (1976).
Welch, J.E., Schatte, E.C., O'Brien, D.A. & Eddy, E.M. Expression of a glyceraldehyde 3-phosphate dehydrogenase gene specific to mouse spermatogenic cells. Biol. Reprod. 46, 869–878 (1992).
Steger, K. Transcriptional and translational regulation of gene expression in haploid spermatids. Anat. Embryol. 199, 471–487 (1999).
Swergold, G.D. Identification, characterization, and cell specificity of a human LINE-1 promoter. Mol. Cell. Biol. 10, 6718–6729 (1990).
Loeb, D.D. et al. The sequence of a large L1Md element reveals a tandemly repeated 5′ end and several features found in retrotransposons. Mol. Cell. Biol. 6, 168–182 (1986).
Padgett, R.W., Hutchison, C.A. 3rd & Edgell, M.H. The F-type 5′ motif of mouse L1 elements: a major class of L1 termini similar to the A-type in organization but unrelated in sequence. Nucleic Acids Res. 16, 739–749 (1988).
Naas, T.P. et al. An actively retrotranposing, novel subfamily of mouse L1 elements. EMBO J. 17, 590–597 (1998).
Luan, D.D., Korman, M.H., Jakubczak, J.L. & Eickbush, T.H. Reverse transcription of R2Bm RNA is primed by a nick at the chromosomal target site: a mechanism for non-LTR retrotransposable elements. Cell 72, 595–605 (1993).
Ostertag, E.M. & Kazazian, H.H. Jr. Twin Priming, a proposed mechanism for the creation of inversions in L1 retrotransposition. Genet. Res. 11, 2059–2065 (2001).
Feng, Q., Moran, J.V., Kazazian, H.H. Jr. & Boeke, J.D. Human L1 retrotransposon encodes a conserved endonuclease required for retrotransposition. Cell 87, 905–916 (1996).
Jurka, J. Sequence patterns indicate an enzymatic involvement in integration of mammalian retroposons. Proc. Natl Acad. Sci. USA 94, 1872–1877 (1997).
Cost, G.J. & Boeke, J.D. Targeting of human retrotransposon integration is directed by the specificity of the L1 endonuclease for regions of unusual DNA structure. Biochem. 37, 18081–18093 (1998).
Boissinot, S., Chevret, P. & Furano, A.V. L1 (LINE-1) retrotransposon evolution and amplification in recent human history. Mol. Biol. Evol. 17, 915–928 (2000).
Kazazian, H.H. Jr. An estimated frequency of endogenous insertional mutations in humans. Nature Genet. 22, 130 (1999).
Li, X. et al. Frequency of recent retrotransposition events in the human factor IX gene. Hum. Mutat. 17, 511–519 (2001).
Jensen, S., Gassama, M.P. & Heidmann, T. Taming of transposable elements by homology-dependent gene silencing. Nature Genet. 21, 209–212 (1999).
Hohjoh, H. & Singer, M.F. Cytoplasmic ribonucleoprotein complexes containing human LINE-1 protein and RNA. EMBO J. 15, 630–639 (1996).
Kolosha, V.O. & Martin, S.L. In vitro properties of the first ORF protein from mouse LINE-1 support its role in ribonucleoprotein particle formation during retrotransposition. Proc. Natl Acad. Sci. USA 94, 10155–10160 (1997).
Mathias, S.L., Scott, A.F., Kazazian, H.H., Boeke, J.D. & Gabriel, A. Reverse transcriptase encoded by a human transposable element. Science 254, 1808–1810 (1991).
Fanning, T. & Singer, M. The line-1 DNA-sequences in 4 mammalian orders predicts proteins that conserve homologies to retrovirus proteins. Nucleic Acids Res. 15, 2251–2260 (1987).
Acknowledgements
We thank A. Farley, K.-S. Kim and M.C. Cha for technical assistance; the Transgenic and DNA Sequencing Cores of the University of Pennsylvania for generation of transgenic mouse lines and for DNA sequences, respectively; and J. Moran and B. Brouha for critical reading of the manuscript. E.M.O. was supported by a Howard Hughes Predoctoral Fellowship, and H.H.K. was supported by grants from the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Ostertag, E., DeBerardinis, R., Goodier, J. et al. A mouse model of human L1 retrotransposition. Nat Genet 32, 655–660 (2002). https://doi.org/10.1038/ng1022
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1022
This article is cited by
-
Control of LINE-1 Expression Maintains Genome Integrity in Germline and Early Embryo Development
Reproductive Sciences (2022)
-
A potential new mechanism for pregnancy loss: considering the role of LINE-1 retrotransposons in early spontaneous miscarriage
Reproductive Biology and Endocrinology (2020)
-
dnmt1 function is required to maintain retinal stem cells within the ciliary marginal zone of the zebrafish eye
Scientific Reports (2020)
-
Targeted gene knockin in zebrafish using the 28S rDNA-specific non-LTR-retrotransposon R2Ol
Mobile DNA (2019)
-
Insertion of a chimeric retrotransposon sequence in mouse Axin1 locus causes metastable kinky tail phenotype
Mobile DNA (2019)