Abstract
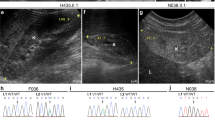
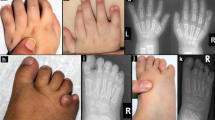
Noonan syndrome is characterized by short stature, facial dysmorphia and a wide spectrum of congenital heart defects1,2. Mutations of PTPN11, KRAS and SOS1 in the RAS-MAPK pathway cause ∼60% of cases of Noonan syndrome3,4,5,6,7,8,9. However, the gene(s) responsible for the remainder are unknown. We have identified five different mutations in RAF1 in ten individuals with Noonan syndrome; those with any of four mutations causing changes in the CR2 domain of RAF1 had hypertrophic cardiomyopathy (HCM), whereas affected individuals with mutations leading to changes in the CR3 domain did not. Cells transfected with constructs containing Noonan syndrome–associated RAF1 mutations showed increased in vitro kinase and ERK activation, and zebrafish embryos with morpholino knockdown of raf1 demonstrated the need for raf1 for the development of normal myocardial structure and function. Thus, our findings implicate RAF1 gain-of-function mutations as a causative agent of a human developmental disorder, representing a new genetic mechanism for the activation of the MAPK pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Noonan, J.A. Hypertelorism with Turner phenotype. A new syndrome with associated congenital heart disease. Am. J. Dis. Child. 116, 373–380 (1968).
Allanson, J.E. Noonan syndrome. J. Med. Genet. 24, 9–13 (1987).
Tartaglia, M. et al. Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome. Nat. Genet. 29, 465–468 (2001).
Tartaglia, M. et al. PTPN11 mutations in Noonan syndrome: molecular spectrum, genotype-phenotype correlation, and phenotypic heterogeneity. Am. J. Hum. Genet. 70, 1555–1563 (2002).
Fragale, A., Tartaglia, M., Wu, J. & Gelb, B.D. Noonan syndrome-associated SHP2/PTPN11 mutants cause EGF-dependent prolonged GAB1 binding and sustained ERK2/MAPK1 activation. Hum. Mutat. 23, 267–277 (2004).
Schubbert, S. et al. Germline KRAS mutations cause Noonan syndrome. Nat. Genet. 38, 331–336 (2006).
Carta, C. et al. Germline missense mutations affecting KRAS isoform B are associated with a severe Noonan syndrome phenotype. Am. J. Hum. Genet. 79, 129–135 (2006).
Roberts, A.E. et al. Germline gain-of-function mutations in SOS1 cause Noonan syndrome. Nat. Genet. 39, 70–74 (2007).
Tartaglia, M. et al. Gain-of-function SOS1 mutations cause a distinctive form of Noonan syndrome. Nat. Genet. 39, 75–79 (2007).
Bentires-Alj, M., Kontaridis, M.I. & Neel, B.G. Stops on the Ras pathway in human genetic disease. Nat. Med. 12, 283–285 (2006).
Kratz, C.P. et al. Germline mutations in components of the Ras signaling pathway in Noonan syndrome and related disorders. Cell Cycle 5, 1607–1611 (2006).
Gelb, B.D. & Tartaglia, M. Noonan syndrome and related disorders: dysregulated RAS-mitogen activated protein kinase signal transduction. Hum. Mol. Genet. 15, R220–R226 (2006).
Aoki, Y. et al. Germline mutations in HRAS proto-oncogene cause Costello syndrome. Nat. Genet. 37, 1038–1040 (2005).
Niihori, T. et al. Germline KRAS and BRAF mutations in cardio-facio-cutaneous syndrome. Nat. Genet. 38, 294–296 (2006).
Rodriguez-Viciana, P. et al. Germline mutations in genes within the MAPK pathway cause cardio-facio-cutaneous syndrome. Science 311, 1287–1290 (2006).
Tartaglia, M. et al. PTPN11 mutations in Noonan Syndrome: Molecular Spectrum, Genotype-Phenotype Correlation, and Phenotypic Heterogeneity. Am. J. Hum. Genet. 70, 1555–1563 (2002).
Marshall, C.J. MAP kinase kinase kinase, MAP kinase kinase and MAP kinase. Curr. Opin. Genet. Dev. 4, 82–89 (1994).
Schubbert, S. et al. Hyperactive Ras in developmental disorders and cancer. Nat. Rev. Cancer 7, 295–308 (2007).
Garnett, M.J. & Marais, R. Guilty as charged: B-RAF is a human oncogene. Cancer Cell 6, 313–319 (2004).
Heidecker, G. et al. Mutational activation of c-raf-1 and definition of the minimal transforming sequence. Mol. Cell. Biol. 10, 2503–2512 (1990).
Zebisch, A. et al. Two transforming C-RAF germ-line mutations identified in patients with therapy-related acute myeloid leukemia. Cancer Res. 66, 3401–3408 (2006).
Emuss, V. et al. Mutations of C-RAF are rare in human cancer because C-RAF has a low basal kinase activity compared with B-RAF. Cancer Res. 65, 9719–9726 (2005).
Chan, E.Y. et al. Mutations in conserved regions 1, 2, and 3 of Raf-1 that activate transforming activity. Mol. Carcinog. 33, 189–197 (2002).
Dumaz, N. & Marais, R. Protein Kinase A Blocks Raf-1 Activity by Stimulating 14–3-3 Binding and Blocking Raf-1 Interaction with Ras. J. Biol. Chem. 278, 29819–29823 (2003).
Harris, I.S. et al. Raf-1 kinase is required for cardiac hypertrophy and cardiomyocyte survival in response to pressure overload. Circulation 110, 718–723 (2004).
Yamazaki, T. et al. Protein kinase A and protein kinase C synergistically activate the Raf-1 kinase/mitogen-activated protein kinase cascade in neonatal rat cardiomyocytes. J. Mol. Cell. Cardiol. 29, 2491–2501 (1997).
Bueno, O.F. et al. The MEK1–ERK1/2 signaling pathway promotes compensated cardiac hypertrophy in transgenic mice. EMBO J. 19, 6341–6350 (2000).
Kimmel, C.B. et al. Stages of embryonic development of the zebrafish. Dev. Dyn. 203, 253–310 (1995).
Komoike, Y. et al. Zebrafish Polycomb group gene ph2α is required for epiboly and tailbud formation acting downstream of FGF signaling. Biochem. Biophys. Res. Commun. 328, 858–866 (2005).
Acknowledgements
The authors are grateful to the patients, their family members and the referring physicians. We thank B. Nadal-Ginard for his critical reading of the manuscript and for discussions. We also thank S. Arai and S. Imamura for generating Epstein-Barr virus–transformed lymphoblastoid cell lines of the subjects; K. Hirayama-Yamada, K. Komatsu, H. Nagao and K. Kihara for technical assistance and B. Levene for English correction of the manuscript. H.Y. was supported by a research fellowship from the Japan Society for the Promotion of Science. This project was supported by the Encouraging Development of Strategic Research Centers, Special Coordination Funds for Promoting Science and Technology, Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Contributions
M.A.R. and R.M. designed experiments; M.A.R. performed sequencing and biochemical analysis; plasmid construction was done by M.A.R., T.N. and H.Y.; patient phenotyping was performed by M.F., M.K., K.M., H.K., M.N., Y.F., M.M., K.M., M.T., H.H., J.M. and R.M.; zebrafish experiments were contributed by Y.K., R.A. and T.H.; M.A.R. and R.M. wrote the paper and R.M. organized and supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figs 1-3, Supplementary Tables 1-2, Supp Methods (PDF 377 kb)
Rights and permissions
About this article
Cite this article
Razzaque, M., Nishizawa, T., Komoike, Y. et al. Germline gain-of-function mutations in RAF1 cause Noonan syndrome. Nat Genet 39, 1013–1017 (2007). https://doi.org/10.1038/ng2078
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng2078
This article is cited by
-
The role of CRAF in cancer progression: from molecular mechanisms to precision therapies
Nature Reviews Cancer (2024)
-
High frequency of hotspot mutation in PTPN11 gene among Moroccan patients with Noonan syndrome
Journal of Applied Genetics (2024)
-
Novel effects of Ras-MAPK pathogenic variants on the developing human brain and their link to gene expression and inhibition abilities
Translational Psychiatry (2023)
-
RASopathies and sigmoid-shaped ventricular septum morphology: evidence of a previously unappreciated cardiac phenotype
Pediatric Research (2023)
-
An Assessment of the Therapeutic Landscape for the Treatment of Heart Disease in the RASopathies
Cardiovascular Drugs and Therapy (2023)