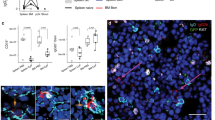
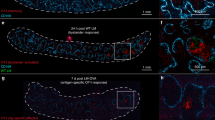
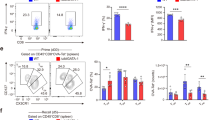
Abstract
The cellular basis of immunological memory remains a controversial issue. Here we show that basophils bound large amounts of intact antigens on their surface and were the main source of interleukins 6 and 4 in the spleen and bone marrow after restimulation with a soluble antigen. Depletion of basophils resulted in a much lower humoral memory response and greater susceptibility of immunized mice to sepsis induced by Streptococcus pneumoniae. Adoptive transfer of antigen-reactive basophils significantly increased specific antibody production, and activated basophils, together with CD4+ T cells, profoundly enhanced B cell proliferation and immunoglobulin production. These basophil-dependent effects on B cells required interleukins 6 and 4 and increased the capacity of CD4+ T cells to provide B cell help. Thus, basophils are important contributors to humoral memory immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
McHeyzer-Williams, L.J., Malherbe, L.P. & McHeyzer-Williams, M.G. Checkpoints in memory B-cell evolution. Immunol. Rev. 211, 255–268 (2006).
Zinkernagel, R.M. & Hengartner, H. Protective 'immunity' by pre-existent neutralizing antibody titers and preactivated T cells but not by so-called 'immunological memory'. Immunol. Rev. 211, 310–319 (2006).
Manz, R.A., Thiel, A. & Radbruch, A. Lifetime of plasma cells in the bone marrow. Nature 388, 133–134 (1997).
Julius, M.H., Masuda, T. & Herzenberg, L.A. Demonstration that antigen-binding cells are precursors of antibody-producing cells after purification with a fluorescence-activated cell sorter. Proc. Natl. Acad. Sci. USA 69, 1934–1938 (1972).
McHeyzer-Williams, L.J. & McHeyzer-Williams, M.G. Antigen-specific memory B cell development. Annu. Rev. Immunol. 23, 487–513 (2005).
Fagarasan, S. & Honjo, T. T-independent immune response: new aspects of B cell biology. Science 290, 89–92 (2000).
Mack, M. et al. Identification of antigen-capturing cells as basophils. J. Immunol. 174, 735–741 (2005).
Bell, J. & Gray, D. Antigen-capturing cells can masquerade as memory B cells. J. Exp. Med. 197, 1233–1244 (2003).
Wolniak, K.L., Noelle, R.J. & Waldschmidt, T.J. Characterization of (4-hydroxy-3-nitrophenyl)acetyl (NP)-specific germinal center B cells and antigen-binding B220− cells after primary NP challenge in mice. J. Immunol. 177, 2072–2079 (2006).
Driver, D.J., McHeyzer-Williams, L.J., Cool, M., Stetson, D.B. & McHeyzer-Williams, M.G. Development and maintenance of a B220− memory B cell compartment. J. Immunol. 167, 1393–1405 (2001).
McHeyzer-Williams, L.J., Cool, M. & McHeyzer-Williams, M.G. Antigen-specific B cell memory: expression and replenishment of a novel B220− memory B cell compartment. J. Exp. Med. 191, 1149–1166 (2000).
Prussin, C. & Metcalfe, D.D. 5. IgE, mast cells, basophils, and eosinophils. J. Allergy Clin. Immunol. 117, 450–456 (2006).
Gibbs, B.F. Human basophils as effectors and immunomodulators of allergic inflammation and innate immunity. Clin. Exp. Med. 5, 43–49 (2005).
Lantz, C.S. et al. Role for interleukin-3 in mast-cell and basophil development and in immunity to parasites. Nature 392, 90–93 (1998).
Kawakami, T. & Galli, S.J. Regulation of mast-cell and basophil function and survival by IgE. Nat. Rev. Immunol. 2, 773–786 (2002).
Valent, P. et al. Interleukin 3 activates human blood basophils via high-affinity binding sites. Proc. Natl. Acad. Sci. USA 86, 5542–5546 (1989).
Bischoff, S.C., Brunner, T., De Weck, A.L. & Dahinden, C.A. Interleukin 5 modifies histamine release and leukotriene generation by human basophils in response to diverse agonists. J. Exp. Med. 172, 1577–1582 (1990).
Bischoff, S.C., Krieger, M., Brunner, T. & Dahinden, C.A. Monocyte chemotactic protein 1 is a potent activator of human basophils. J. Exp. Med. 175, 1271–1275 (1992).
Yoshimoto, T. et al. IL-18, although antiallergic when administered with IL-12, stimulates IL-4 and histamine release by basophils. Proc. Natl. Acad. Sci. USA 96, 13962–13966 (1999).
Geiser, T., Dewald, B., Ehrengruber, M.U., Clark-Lewis, I. & Baggiolini, M. The interleukin-8-related chemotactic cytokines GROα, GROβ, and GROγ activate human neutrophil and basophil leukocytes. J. Biol. Chem. 268, 15419–15424 (1993).
Sokol, C.L., Barton, G.M., Farr, A.G. & Medzhitov, R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nat. Immunol. 9, 310–318 (2008).
Gauchat, J.F. et al. Induction of human IgE synthesis in B cells by mast cells and basophils. Nature 365, 340–343 (1993).
Mukai, K. et al. Basophils play a critical role in the development of IgE-mediated chronic allergic inflammation independently of T cells and mast cells. Immunity 23, 191–202 (2005).
Ben-Sasson, S.Z., Le Gros, G., Conrad, D.H., Finkelman, F.D. & Paul, W.E. Cross-linking Fc receptors stimulate splenic non-B, non-T cells to secrete interleukin 4 and other lymphokines. Proc. Natl. Acad. Sci. USA 87, 1421–1425 (1990).
Seder, R.A. et al. Mouse splenic and bone marrow cell populations that express high-affinity Fcε receptors and produce interleukon 4 are enriched in basophils. Proc. Natl. Acad. Sci. USA 88, 2835–2839 (1991).
Min, B. et al. Basophils produce IL-4 and accumulate in tissues after infection with a Th2-inducing parasite. J. Exp. Med. 200, 507–517 (2004).
Hessel, E.M. et al. Immunostimulatory oligonucleotides block allergic airway inflammation by inhibiting Th2 cell activation and IgE-mediated cytokine induction. J. Exp. Med. 202, 1563–1573 (2005).
Hida, S., Tadachi, M., Saito, T. & Taki, S. Negative control of basophil expansion by IRF-2 critical for the regulation of Th1/Th2 balance. Blood 106, 2011–2017 (2005).
Oh, K., Shen, T., Le Gros, G. & Min, B. Induction of Th2 type immunity in a mouse system reveals a novel immunoregulatory role of basophils. Blood 109, 2921–2927 (2007).
Khodoun, M.V., Orekhova, T., Potter, C., Morris, S. & Finkelman, F.D. Basophils initiate IL-4 production during a memory T-dependent response. J. Exp. Med. 200, 857–870 (2004).
Takai, T., Ono, M., Hikida, M., Ohmori, H. & Ravetch, J.V. Augmented humoral and anaphylactic responses in FcγRII-deficient mice. Nature 379, 346–349 (1996).
Kuhn, R., Rajewsky, K. & Muller, W. Generation and analysis of interleukin-4 deficient mice. Science 254, 707–710 (1991).
Maurer, D. et al. Expression of functional high affinity immunoglobulin E receptors (FcεRI) on monocytes of atopic individuals. J. Exp. Med. 179, 745–750 (1994).
Maurer, D. et al. Peripheral blood dendritic cells express FcεRI as a complex composed of FcεRIα- and FcεRIγ-chains and can use this receptor for IgE-mediated allergen presentation. J. Immunol. 157, 607–616 (1996).
Kita, H. et al. Does IgE bind to and activate eosinophils from patients with allergy? J. Immunol. 162, 6901–6911 (1999).
Langermann, S. et al. Protective humoral response against pneumococcal infection in mice elicited by recombinant bacille Calmette-Guerin vaccines expressing pneumococcal surface protein A. J. Exp. Med. 180, 2277–2286 (1994).
Swiatlo, E., King, J., Nabors, G.S., Mathews, B. & Briles, D.E. Pneumococcal surface protein A is expressed in vivo, and antibodies to PspA are effective for therapy in a murine model of pneumococcal sepsis. Infect. Immun. 71, 7149–7153 (2003).
Gor, D.O., Ding, X., Briles, D.E., Jacobs, M.R. & Greenspan, N.S. Relationship between surface accessibility for PpmA, PsaA, and PspA and antibody-mediated immunity to systemic infection by Streptococcus pneumoniae. Infect. Immun. 73, 1304–1312 (2005).
Hasbold, J., Corcoran, L.M., Tarlinton, D.M., Tangye, S.G. & Hodgkin, P.D. Evidence from the generation of immunoglobulin G–secreting cells that stochastic mechanisms regulate lymphocyte differentiation. Nat. Immunol. 5, 55–63 (2004).
Abbas, A.K., Murphy, K.M. & Sher, A. Functional diversity of helper T lymphocytes. Nature 383, 787–793 (1996).
Rincon, M., Anguita, J., Nakamura, T., Fikrig, E. & Flavell, R.A. Interleukin (IL)-6 directs the differentiation of IL-4-producing CD4+ T cells. J. Exp. Med. 185, 461–469 (1997).
Kopf, M., Herren, S., Wiles, M.V., Pepys, M.B. & Kosco-Vilbois, M.H. Interleukin 6 influences germinal center development and antibody production via a contribution of C3 complement component. J. Exp. Med. 188, 1895–1906 (1998).
Matsuda, T., Yamasaki, K., Taga, T., Hirano, T. & Kishimoto, T. Current concepts of B cell modulation. Int. Rev. Immunol. 5, 97–109 (1989).
Seder, R.A. & Paul, W.E. Acquisition of lymphokine-producing phenotype by CD4+ T cells. Annu. Rev. Immunol. 12, 635–673 (1994).
Dombrowicz, D. et al. Absence of FcεRIα chain results in upregulation of FcγRIII-dependent mast cell degranulation and anaphylaxis. Evidence of competition between FcεRI and FcγRIII for limiting amounts of FcR β and γ chains. J. Clin. Invest. 99, 915–925 (1997).
Miyajima, I. et al. Systemic anaphylaxis in the mouse can be mediated largely through IgG1 and FcγRIII. Assessment of the cardiopulmonary changes, mast cell degranulation, and death associated with active or IgE- or IgG1-dependent passive anaphylaxis. J. Clin. Invest. 99, 901–914 (1997).
Piccinni, M.P. et al. Human bone marrow non-B, non-T cells produce interleukin 4 in response to cross-linkage of Fcε and Fcγ receptors. Proc. Natl. Acad. Sci. USA 88, 8656–8660 (1991).
Kopf, M. et al. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature 368, 339–342 (1994).
Takai, T., Li, M., Sylvestre, D., Clynes, R. & Ravetch, J.V. FcR γ chain deletion results in pleiotrophic effector cell defects. Cell 76, 519–529 (1994).
Winter, C. et al. Lung-specific overexpression of CC chemokine ligand (CCL) 2 enhances the host defense to Streptococcus pneumoniae infection in mice: role of the CCL2–CCR2 axis. J. Immunol. 178, 5828–5838 (2007).
Acknowledgements
We thank K. Schmidbauer, M. Wondrak and N. Göbel for technical assistance, and D. Schlöndorff for critical reading of the manuscript. Supported by the Deutsche Forschungsgemeinschaft (M.M.) and the University Hospital Regensburg.
Author information
Authors and Affiliations
Contributions
A.D. contributed to the results in Figures 1,2,3,4,5; U.A.M., C.W., R.M., S.H. and D.E.B. contributed to the results in Figure 4c,d; M.R.G. contributed to the results in Figures 1 and 3; C.M. and L.A.K.-S., contributed to the results in Figures 6,7,8,9; M.N. and Y.T. contributed to the results in Figures 1 and 6,7,8,9; and M.M. contributed to the results in Figures 1,2,3,4,5,6,7,8,9.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15 and Supplementary Table 1 (PDF 682 kb)
Rights and permissions
About this article
Cite this article
Denzel, A., Maus, U., Gomez, M. et al. Basophils enhance immunological memory responses. Nat Immunol 9, 733–742 (2008). https://doi.org/10.1038/ni.1621
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1621
This article is cited by
-
PD-L1- and IL-4-expressing basophils promote pathogenic accumulation of T follicular helper cells in lupus
Nature Communications (2024)
-
Basophils absence predicts poor prognosis and indicates immunosuppression of patients in intensive care units
Scientific Reports (2023)
-
Immunoregulatory nanomedicine for respiratory infections
Nature Reviews Bioengineering (2023)
-
Prognostic significance of circulating basophil counts in patients who underwent esophagectomy for esophageal cancer
Langenbeck's Archives of Surgery (2023)
-
Comprehensive overview of COVID-19-related respiratory failure: focus on cellular interactions
Cellular & Molecular Biology Letters (2022)