Abstract
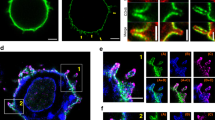
The reorientation of the T cell microtubule-organizing center (MTOC) toward the antigen-presenting cell enables the directional secretion of cytokines and lytic factors. By single-cell photoactivation of the T cell antigen receptor, we show that MTOC polarization is driven by localized accumulation of diacylglycerol (DAG). MTOC reorientation was closely preceded first by production of DAG and then by recruitment of the microtubule motor protein dynein. Blocking DAG production or disrupting the localization of DAG impaired MTOC recruitment. Localized DAG accumulation was also required for cytotoxic T cell–mediated killing. Furthermore, photoactivation of DAG itself was sufficient to induce transient polarization. Our data identify a DAG-dependent pathway that signals through dynein to control microtubule polarity in T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Breart, B., Lemaitre, F., Celli, S. & Bousso, P. Two-photon imaging of intratumoral CD8+ T cell cytotoxic activity during adoptive T cell therapy in mice. J. Clin. Invest. 118, 1390–1397 (2008).
Butz, E.A. & Bevan, M.J. Massive expansion of antigen-specific CD8+ T cells during an acute virus infection. Immunity 8, 167–175 (1998).
Murali-Krishna, K. et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8, 177–187 (1998).
Bromley, S.K. et al. The immunological synapse. Annu. Rev. Immunol. 19, 375–396 (2001).
Huse, M., Lillemeier, B.F., Kuhns, M.S., Chen, D.S. & Davis, M.M. T cells use two directionally distinct pathways for cytokine secretion. Nat. Immunol. 7, 247–255 (2006).
Kupfer, A., Mosmann, T.R. & Kupfer, H. Polarized expression of cytokines in cell conjugates of helper T cells and splenic B cells. Proc. Natl. Acad. Sci. USA 88, 775–779 (1991).
Stinchcombe, J.C., Bossi, G., Booth, S. & Griffiths, G.M. The immunological synapse of CTL contains a secretory domain and membrane bridges. Immunity 15, 751–761 (2001).
Huse, M., Quann, E.J. & Davis, M.M. Shouts, whispers, and the kiss of death: directional secretion in T cells. Nat. Immunol. 9, 1105–1111 (2008).
Chang, J.T. et al. Asymmetric T lymphocyte division in the initiation of adaptive immune responses. Science 315, 1687–1691 (2007).
Martin-Cofreces, N.B. et al. MTOC translocation modulates IS formation and controls sustained T cell signaling. J. Cell Biol. 182, 951–962 (2008).
Depoil, D. et al. Immunological synapses are versatile structures enabling selective T cell polarization. Immunity 22, 185–194 (2005).
Huse, M. et al. Spatial and temporal dynamics of T cell receptor signaling with a photoactivatable agonist. Immunity 27, 76–88 (2007).
Sedwick, C.E. et al. TCR, LFA-1, and CD28 play unique and complementary roles in signaling T cell cytoskeletal reorganization. J. Immunol. 162, 1367–1375 (1999).
Koretzky, G.A., Abtahian, F. & Silverman, M.A. SLP76 and SLP65: complex regulation of signalling in lymphocytes and beyond. Nat. Rev. Immunol. 6, 67–78 (2006).
Sommers, C.L., Samelson, L.E. & Love, P.E. LAT: a T lymphocyte adapter protein that couples the antigen receptor to downstream signaling pathways. Bioessays 26, 61–67 (2004).
Kuhne, M.R. et al. Linker for activation of T cells, ζ-associated protein-70, and Src homology 2 domain-containing leukocyte protein-76 are required for TCR-induced microtubule-organizing center polarization. J. Immunol. 171, 860–866 (2003).
Lowin-Kropf, B., Shapiro, V.S. & Weiss, A. Cytoskeletal polarization of T cells is regulated by an immunoreceptor tyrosine-based activation motif-dependent mechanism. J. Cell Biol. 140, 861–871 (1998).
Spitaler, M., Emslie, E., Wood, C.D. & Cantrell, D. Diacylglycerol and protein kinase D localization during T lymphocyte activation. Immunity 24, 535–546 (2006).
Combs, J. et al. Recruitment of dynein to the Jurkat immunological synapse. Proc. Natl. Acad. Sci. USA 103, 14883–14888 (2006).
Zhong, X.P., Guo, R., Zhou, H., Liu, C. & Wan, C.K. Diacylglycerol kinases in immune cell function and self-tolerance. Immunol. Rev. 224, 249–264 (2008).
Huang, X.P., Sreekumar, R., Patel, J.R. & Walker, J.W. Response of cardiac myocytes to a ramp increase of diacylglycerol generated by photolysis of a novel caged diacylglycerol. Biophys. J. 70, 2448–2457 (1996).
Suzuki, A.Z. et al. Coumarin-4-ylmethoxycarbonyls as phototriggers for alcohols and phenols. Org. Lett. 5, 4867–4870 (2003).
Bunnell, S.C., Kapoor, V., Trible, R.P., Zhang, W. & Samelson, L.E. Dynamic actin polymerization drives T cell receptor-induced spreading: a role for the signal transduction adaptor LAT. Immunity 14, 315–329 (2001).
Negulescu, P.A., Krasieva, T.B., Khan, A., Kerschbaum, H.H. & Cahalan, M.D. Polarity of T cell shape, motility, and sensitivity to antigen. Immunity 4, 421–430 (1996).
Jones, G.A. & Carpenter, G. The regulation of phospholipase C-gamma 1 by phosphatidic acid. Assessment of kinetic parameters. J. Biol. Chem. 268, 20845–20850 (1993).
Baier, G. The PKC gene module: molecular biosystematics to resolve its T cell functions. Immunol. Rev. 192, 64–79 (2003).
Irie, A. et al. Protein kinase D2 contributes to either IL-2 promoter regulation or induction of cell death upon TCR stimulation depending on its activity in Jurkat cells. Int. Immunol. 18, 1737–1747 (2006).
Ebinu, J.O. et al. RasGRP links T-cell receptor signaling to Ras. Blood 95, 3199–3203 (2000).
Caloca, M.J., Delgado, P., Alarcon, B. & Bustelo, X.R. Role of chimaerins, a group of Rac-specific GTPase activating proteins, in T-cell receptor signaling. Cell. Signal. 20, 758–770 (2008).
Resnick, M.S. et al. Differential downstream functions of protein kinase Cη and -θ in EL4 mouse thymoma cells. J. Biol. Chem. 273, 27654–27661 (1998).
Siliceo, M. et al. Beta2-chimaerin provides a diacylglycerol-dependent mechanism for regulation of adhesion and chemotaxis of T cells. J. Cell Sci. 119, 141–152 (2006).
Sims, T.N. et al. Opposing effects of PKCθ and WASp on symmetry breaking and relocation of the immunological synapse. Cell 129, 773–785 (2007).
Sanjuan, M.A., Jones, D.R., Izquierdo, M. & Merida, I. Role of diacylglycerol kinase α in the attenuation of receptor signaling. J. Cell Biol. 153, 207–220 (2001).
Etienne-Manneville, S. & Hall, A. Cell polarity: Par6, aPKC and cytoskeletal crosstalk. Curr. Opin. Cell Biol. 15, 67–72 (2003).
Manneville, J.B. & Etienne-Manneville, S. Positioning centrosomes and spindle poles: looking at the periphery to find the centre. Biol. Cell 98, 557–565 (2006).
Li, R. & Gundersen, G.G. Beyond polymer polarity: how the cytoskeleton builds a polarized cell. Nat. Rev. Mol. Cell Biol. 9, 860–873 (2008).
Real, E., Faure, S., Donnadieu, E. & Delon, J. Cutting edge: atypical PKCs regulate T lymphocyte polarity and scanning behavior. J. Immunol. 179, 5649–5652 (2007).
Gomez, T.S. et al. Formins regulate the actin-related protein 2/3 complex-independent polarization of the centrosome to the immunological synapse. Immunity 26, 177–190 (2007).
Stowers, L., Yelon, D., Berg, L.J. & Chant, J. Regulation of the polarization of T cells toward antigen-presenting cells by Ras-related GTPase CDC42. Proc. Natl. Acad. Sci. USA 92, 5027–5031 (1995).
Devreotes, P. & Janetopoulos, C. Eukaryotic chemotaxis: distinctions between directional sensing and polarization. J. Biol. Chem. 278, 20445–20448 (2003).
Purbhoo, M.A., Irvine, D.J., Huppa, J.B. & Davis, M.M. T cell killing does not require the formation of a stable mature immunological synapse. Nat. Immunol. 5, 524–530 (2004).
Acknowledgements
We thank G. Altan-Bonnet (Memorial Sloan-Kettering Cancer Center) and O. Feinerman (Memorial Sloan-Kettering Cancer Center) for assistance with MATLAB programming, RMA-s cells and other reagents; T. Pentcheva-Hoang (Memorial Sloan-Kettering Cancer Center) and J. Allison (Memorial Sloan-Kettering Cancer Center) for CH12 cells and help with imaging experiments; Q. Li (Duke University) for helper T cell cDNA; W. Sha (University of California, Berkeley) and M.S. Kuhns (Stanford University) for pMSCV; R. Tsien (University of California, San Diego) and M.W. Davidson (Florida State University) for Tag-RFP-T; S.S. Yi and the Sloan-Kettering Institute Microchemistry Core Facility for peptide synthesis; D. Sant'Angelo, G. Altan-Bonnet, A. Hall, P. Abeyweera, D.A. Schaer and K. Pham for advice and critical reading of the manuscript; B. Driscoll for technical assistance; the laboratories of M.O. Li and B. Dupont for reagents and comments; and M.M. Davis for advice and support at the start of this project. Supported by the Spanish Ministry of Science and Innovation (E.M.), the Ministry of Education, Culture, Sports, Science and Technology of Japan (T.F.), the Searle Scholars Program (M.H.) and the Cancer Research Institute (M.H.).
Author information
Authors and Affiliations
Contributions
E.J.Q. and M.H. designed the experiments, collected the data and analyzed the results; E.M. helped develop the DGK studies; T.F. provided caged diC8; and M.H. wrote the manuscript with input from E.J.Q. and E.M.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–2 and Supplementary Movie Legends (PDF 1035 kb)
Supplementary Movie 1
Inhibition of PLC-γ blocks MTOC reorientation in TH cells. (MOV 1391 kb)
Supplementary Movie 2
The control compound U-73343 does not affect MTOC reorientation. (MOV 2226 kb)
Supplementary Movie 3
PMA inhibits MTOC reorientation. (MOV 2270 kb)
Supplementary Movie 4
Maintenance of MTOC polarization under control conditions. (MOV 756 kb)
Supplementary Movie 5
PMA impairs the maintenance of MTOC polarization. (MOV 525 kb)
Supplementary Movie 6
Ionomycin does not affect the maintenance of MTOC polarization. (MOV 417 kb)
Supplementary Movie 7
DAG accumulation precedes and is spatially correlated with MTOC reorientation. (MOV 216 kb)
Supplementary Movie 8
Dynein accumulation precedes and is spatially correlated with MTOC reorientation. (MOV 2517 kb)
Supplementary Movie 9
DAG accumulation precedes and is spatially correlated with dynein recruitment. (MOV 194 kb)
Supplementary Movie 10
DGK inhibition impairs the stable accumulation of DAG and the polarization of the MTOC. (MOV 2776 kb)
Supplementary Movie 11
DGK inhibition impairs the stable accumulation of dynein and the polarization of the MTOC. (MOV 399 kb)
Supplementary Movie 12
Localized DAG signaling is sufficient to induce transient reorientation of the MTOC. (MOV 3642 kb)
Rights and permissions
About this article
Cite this article
Quann, E., Merino, E., Furuta, T. et al. Localized diacylglycerol drives the polarization of the microtubule-organizing center in T cells. Nat Immunol 10, 627–635 (2009). https://doi.org/10.1038/ni.1734
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1734
This article is cited by
-
High-throughput spatiotemporal monitoring of single-cell secretions via plasmonic microwell arrays
Nature Biomedical Engineering (2023)
-
Extended-Synaptotagmin-1 and -2 control T cell signaling and function
EMBO Reports (2023)
-
Mechanically active integrins target lytic secretion at the immune synapse to facilitate cellular cytotoxicity
Nature Communications (2022)
-
Comprehensive landscape of extracellular vesicle-derived RNAs in cancer initiation, progression, metastasis and cancer immunology
Molecular Cancer (2020)
-
CLIP-170 is essential for MTOC repositioning during T cell activation by regulating dynein localisation on the cell surface
Scientific Reports (2018)