Abstract
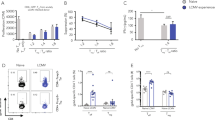
How vaccines control the development of antigen-specific effector and memory T helper cells is central to protective immunity but remains poorly understood. Here we found that protein vaccination selected high-affinity, CXCR5+ICOShi follicular B-helper T cells (TFH cells) that developed in draining lymphoid tissue to regulate B cell responses. In the memory phase, reservoirs of antigen-specific CXCR5+ICOSlo TFH cells persisted with less effector activity but accelerated antigen-recall ability. This new compartment of memory TFH cells was retained in draining lymphoid sites with antigen-specific memory B cells, persistent complexes of peptide and major histocompatibility complex class II and continued expression of CD69. Thus, protein vaccination promotes B cell immunity by selecting high-affinity effector TFH cells and creating lymphoid reservoirs of antigen-specific memory TFH cells in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
McHeyzer-Williams, L.J. & McHeyzer-Williams, M.G. Antigen-specific memory B cell development. Annu. Rev. Immunol. 23, 487–513 (2005).
Breitfeld, D. et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J. Exp. Med. 192, 1545–1552 (2000).
Campbell, D.J., Kim, C.H. & Butcher, E.C. Separable effector T cell populations specialized for B cell help or tissue inflammation. Nat. Immunol. 2, 876–881 (2001).
Schaerli, P. et al. CXC chemokine receptor 5 expression defines follicular homing T cells with B cell helper function. J. Exp. Med. 192, 1553–1562 (2000).
Vinuesa, C.G., Tangye, S.G., Moser, B. & Mackay, C.R. Follicular B helper T cells in antibody responses and autoimmunity. Nat. Rev. Immunol. 5, 853–865 (2005).
Ansel, K.M., McHeyzer-Williams, L.J., Ngo, V.N., McHeyzer-Williams, M.G. & Cyster, J.G. In vivo-activated CD4 T cells upregulate CXC chemokine receptor 5 and reprogram their response to lymphoid chemokines. J. Exp. Med. 190, 1123–1134 (1999).
Walker, L.S. et al. Compromised OX40 function in CD28-deficient mice is linked with failure to develop CXC chemokine receptor 5-positive CD4 cells and germinal centers. J. Exp. Med. 190, 1115–1122 (1999).
Chtanova, T. et al. T follicular helper cells express a distinctive transcriptional profile, reflecting their role as non-Th1/Th2 effector cells that provide help for B cells. J. Immunol. 173, 68–78 (2004).
Kim, C.H. et al. Unique gene expression program of human germinal center T helper cells. Blood 104, 1952–1960 (2004).
Mak, T.W. et al. Costimulation through the inducible costimulator ligand is essential for both T helper and B cell functions in T cell–dependent B cell responses. Nat. Immunol. 4, 765–772 (2003).
Obst, R., van Santen, H.M., Mathis, D. & Benoist, C. Antigen persistence is required throughout the expansion phase of a CD4+ T cell response. J. Exp. Med. 201, 1555–1565 (2005).
Celli, S., Garcia, Z. & Bousso, P. CD4 T cells integrate signals delivered during successive DC encounters in vivo . J. Exp. Med. 202, 1271–1278 (2005).
Jelley-Gibbs, D.M. et al. Unexpected prolonged presentation of influenza antigens promotes CD4 T cell memory generation. J. Exp. Med. 202, 697–706 (2005).
Kundig, T.M. et al. On T cell memory: arguments for antigen dependence. Immunol. Rev. 150, 63–90 (1996).
Zammit, D.J., Turner, D.L., Klonowski, K.D., Lefrancois, L. & Cauley, L.S. Residual antigen presentation after influenza virus infection affects CD8 T cell activation and migration. Immunity 24, 439–449 (2006).
McHeyzer-Williams, M.G. & Davis, M.M. Antigen-specific development of primary and memory T cells in vivo . Science 268, 106–111 (1995).
McHeyzer-Williams, L.J., Panus, J.F., Mikszta, J.A. & McHeyzer-Williams, M.G. Evolution of antigen-specific T cell receptors in vivo: Preimmune and antigen-driven selection of preferred complementarity-determining region 3 (CDR3) motifs. J. Exp. Med. 189, 1823–1837 (1999).
Crawford, F., Kozono, H., White, J., Marrack, P. & Kappler, J. Detection of antigen-specific T cells with multivalent soluble class II MHC covalent peptide complexes. Immunity 8, 675–682 (1998).
Malherbe, L. et al. Selective activation and expansion of high-affinity CD4+ T cells in resistant mice upon infection with Leishmania major . Immunity 13, 771–782 (2000).
Malherbe, L., Hausl, C., Teyton, L. & McHeyzer-Williams, M.G. Clonal selection of helper T cells is determined by an affinity threshold with no further skewing of TCR binding properties. Immunity 21, 669–679 (2004).
Savage, P.A., Boniface, J.J. & Davis, M.M. A kinetic basis for T cell receptor repertoire selection during an immune response. Immunity 10, 485–492 (1999).
Sallusto, F., Geginat, J. & Lanzavecchia, A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol. 22, 745–763 (2004).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
McHeyzer-Williams, L.J., Cool, M. & McHeyzer-Williams, M.G. Antigen-specific B cell memory: expression and replenishment of a novel B220− memory B cell compartment. J. Exp. Med. 191, 1149–1166 (2000).
Lopez-Cabrera, M. et al. Molecular cloning, expression, and chromosomal localization of the human earliest lymphocyte activation antigen AIM/CD69, a new member of the C-type animal lectin superfamily of signal-transmitting receptors. J. Exp. Med. 178, 537–547 (1993).
Nakayama, T. et al. The generation of mature, single-positive thymocytes in vivo is dysregulated by CD69 blockade or overexpression. J. Immunol. 168, 87–94 (2002).
Rosen, H., Alfonso, C., Surh, C.D. & McHeyzer-Williams, M.G. Rapid induction of medullary thymocyte phenotypic maturation and egress inhibition by nanomolar sphingosine 1-phosphate receptor agonist. Proc. Natl. Acad. Sci. USA 100, 10907–10912 (2003).
Shiow, L.R. et al. CD69 acts downstream of interferon-α/β to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature 440, 540–544 (2006).
Bikah, G., Pogue-Caley, R.R., McHeyzer-Williams, L.J. & McHeyzer-Williams, M.G. Regulating T helper cell immunity through antigen responsiveness and calcium entry. Nat. Immunol. 1, 402–412 (2000).
Gett, A.V., Sallusto, F., Lanzavecchia, A. & Geginat, J. T cell fitness determined by signal strength. Nat. Immunol. 4, 355–360 (2003).
Iezzi, G., Karjalainen, K. & Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8, 89–95 (1998).
Busch, D.H. & Pamer, E.G. T cell affinity maturation by selective expansion during infection. J. Exp. Med. 189, 701–710 (1999).
Fasso, M. et al. T cell receptor (TCR)-mediated repertoire selection and loss of TCR Vβ diversity during the initiation of a CD4+ T cell response in vivo . J. Exp. Med. 192, 1719–1730 (2000).
Kedl, R.M., Schaefer, B.C., Kappler, J.W. & Marrack, P. T cells down-modulate peptide-MHC complexes on APCs in vivo . Nat. Immunol. 3, 27–32 (2002).
Rasheed, A.U., Rahn, H.P., Sallusto, F., Lipp, M. & Muller, G. Follicular B helper T cell activity is confined to CXCR5hiICOShi CD4 T cells and is independent of CD57 expression. Eur. J. Immunol. 36, 1892–1903 (2006).
Forster, R., Emrich, T., Kremmer, E. & Lipp, M. Expression of the G-protein–coupled receptor BLR1 defines mature, recirculating B cells and a subset of T-helper memory cells. Blood 84, 830–840 (1994).
Masopust, D., Vezys, V., Marzo, A.L. & Lefrancois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001).
Reinhardt, R.L., Khoruts, A., Merica, R., Zell, T. & Jenkins, M.K. Visualizing the generation of memory CD4 T cells in the whole body. Nature 410, 101–105 (2001).
MacLennan, I.C.M. & Gray, D. Antigen-driven selection of virgin and memory B cells. Immunol. Rev. 91, 61–85 (1986).
Gray, D., MacLennan, I.C., Bazin, H. & Khan, M. Migrant μ+δ+ and static μ+δ− B lymphocyte subsets. Eur. J. Immunol. 12, 564–569 (1982).
Lau, L.L., Jamieson, B.D., Somasundaram, T. & Ahmed, R. Cytotoxic T-cell memory without antigen. Nature 369, 648–652 (1994).
Maruyama, M., Lam, K.P. & Rajewsky, K. Memory B-cell persistence is independent of persisting immunizing antigen. Nature 407, 636–642 (2000).
Swain, S.L., Hu, H. & Huston, G. Class II-independent generation of CD4 memory T cells from effectors. Science 286, 1381–1383 (1999).
Mitchell, J. & Abbot, A. Ultrastructure of the antigen-retaining reticulum of lymph node follicles as shown by high-resolution autoradiography. Nature 208, 500–502 (1965).
Kim, C.H. et al. Subspecialization of CXCR5+ T cells: B helper activity is focused in a germinal center-localized subset of CXCR5+ T cells. J. Exp. Med. 193, 1373–1381 (2001).
Furukawa, K., Akasako-Furukawa, A., Shirai, H., Nakamura, H. & Azuma, T. Junctional amino acids determine the maturation pathway of an antibody. Immunity 11, 329–338 (1999).
Esplugues, E. et al. Enhanced antitumor immunity in mice deficient in CD69. J. Exp. Med. 197, 1093–1106 (2003).
Lauzurica, P. et al. Phenotypic and functional characteristics of hematopoietic cell lineages in CD69-deficient mice. Blood 95, 2312–2320 (2000).
Baldridge, J.R. & Crane, R.T. Monophosphoryl lipid A (MPL) formulations for the next generation of vaccines. Methods 19, 103–107 (1999).
McHeyzer-Williams, L.J. & McHeyzer-Williams, M.G. Developmentally distinct Th cells control plasma cell production in vivo . Immunity 20, 231–242 (2004).
Acknowledgements
We thank D. Nemazee, W. Havran and L. Sherman for critical appraisal of the manuscript; J. Shandraw for technical assistance; M. Davis (Stanford University) for the original 5C.C7αβ transgenic mice and the I-Ek tetramer Escherichia coli–expressed preparations; and B. Blazar (University of Minnesota) for the original B10.BR-Thy-1.1 congenic mice. Supported by the National Institutes of Health (AI47231, AI040215 and AI059475 to M.G.M.-W.), the National Institutes of Health National Service Research Award (M.D.E.), the European Molecular Biology Organization (L.M.) and the Arthritis Foundation (R.R.P.-C). This is manuscript 18458 from The Scripps Research Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Dynamics of LN and spleen response to subcutaneous-priming with PCC. (PDF 729 kb)
Supplementary Fig. 2
LN and spleen response to intraperitoneal-priming with PCC. (PDF 542 kb)
Supplementary Fig. 3
CFA induces PCC-specific effector TFH cells in draining LN. (PDF 930 kb)
Supplementary Fig. 4
HEL induces antigen-specific effector TFH in draining LN. (PDF 804 kb)
Supplementary Fig. 5
NP-KLH induces antigen-specific B cell response in draining LN (day 14). (PDF 1558 kb)
Rights and permissions
About this article
Cite this article
Fazilleau, N., Eisenbraun, M., Malherbe, L. et al. Lymphoid reservoirs of antigen-specific memory T helper cells. Nat Immunol 8, 753–761 (2007). https://doi.org/10.1038/ni1472
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1472
This article is cited by
-
Memory B cells
Nature Reviews Immunology (2024)
-
Same yet different — how lymph node heterogeneity affects immune responses
Nature Reviews Immunology (2023)
-
IL-21 Enhances the Immune Protection Induced by the Vibrio vulnificus Hemolysin A Protein
Inflammation (2022)
-
Dysregulation of circulating follicular helper T cells in type 2 diabetic patients with diabetic retinopathy
Immunologic Research (2021)
-
Function and dysfunction of plasma cells in intestine
Cell & Bioscience (2019)