Abstract
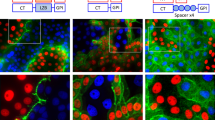
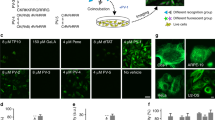
To study the physiological regulation and function of cell-cell gap junction communication in vivo, we developed a bioconjugate of caged dye, named dextran-CANPE-HCC, for imaging cell coupling in small model organisms. In vitro, the compound was photolyzed efficiently with robust fluorescence enhancement. Dextran-CANPE-HCC delivered into Caenorhabditis elegans oocytes was retained in cells throughout development. Using local uncaging, we photolyzed dextran-CANPE-HCC to release the small HCC dye and imaged the dynamics of intercellular dye transfer through gap junction channels, a technique we named Trojan–local activation of molecular fluorescent probes (LAMP). Early during embryonic development, the pattern of cell coupling undergoes dramatic remodeling and imaging revealed that the germ cell precursors, P2, P3 and P4, were isolated from the somatic cell communication compartment. As dextran-CANPE-HCC is chemically and metabolically stable, labeled worms showed very bright signal upon photoactivation after hatching, which allowed us to examine cell coupling in living worms noninvasively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Goodenough, D.A., Goliger, J.A. & Paul, D.L. Connexins, connexons, and intercellular communication. Annu. Rev. Biochem. 65, 475–502 (1996).
Bennett, M.V. & Zukin, R.S. Electrical coupling and neuronal synchronization in the Mammalian brain. Neuron 41, 495–511 (2004).
Harris, A.L. Emerging issues of connexin channels: biophysics fills the gap. Q. Rev. Biophys. 34, 325–472 (2001).
Nicholson, B.J. Gap junctions—from cell to molecule. J. Cell Sci. 116, 4479–4481 (2003).
Dakin, K., Zhao, Y.R. & Li, W.H. LAMP, a new imaging assay of gap junctional communication unveils that Ca2+ influx inhibits cell coupling. Nat. Methods 2, 55–62 (2005).
Dakin, K. & Li, W.H. Infrared-LAMP: two-photon uncaging and imaging of gap junctional communication in three dimensions. Nat. Methods 3, 959 (2006).
Zhao, Y. et al. New caged coumarin fluorophores with extraordinary uncaging cross sections suitable for biological imaging applications. J. Am. Chem. Soc. 126, 4653–4663 (2004).
White, J.G., Southgate, E., Thomson, J.N. & Brenner, S. The structure of the nervous system of the nematode Caenorhabditis elegans. Phil. Trans. R. Soc. Lond. B 314, 1–340 (1986).
Phelan, P. & Starich, T.A. Innexins get into the gap. Bioessays 23, 388–396 (2001).
Herve, J.C., Phelan, P., Bruzzone, R. & White, T.W. Connexins, innexins and pannexins: Bridging the communication gap. Biochim. Biophys. Acta 1719, 3–5 (2005).
Hua, V.B. et al. Sequence and phylogenetic analyses of 4 TMS junctional proteins of animals: connexins, innexins, claudins and occludins. J. Membr. Biol. 194, 59–76 (2003).
Mitchison, T.J., Sawin, K.E., Theriot, J.A., Gee, K. & Mallavarapu, A. Caged fluorescent probes. Methods Enzymol. 291, 63–78 (1998).
Squirrell, J.M., Wokosin, D.L., White, J.G. & Bavister, B.D. Long-term two-photon fluorescence imaging of mammalian embryos without compromising viability. Nat. Biotechnol. 17, 763–767 (1999).
Sulston, J.E., Schierenberg, E., White, J.G. & Thomson, J.N. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev. Biol. 100, 64–119 (1983).
Goodenough, D.A. & Paul, D.L. Beyond the gap: functions of unpaired connexon channels. Nat. Rev. Mol. Cell Biol. 4, 285–294 (2003).
Bao, Z. et al. Automated cell lineage tracing in. Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA (2006).
Strehlow, D. & Gilbert, W. A fate map for the 1st cleavages of the zebrafish. Nature 361, 451–453 (1993).
Wolke, U., Jezuit, E.A. & Priess, J.R. Actin-dependent cytoplasmic streaming in C. elegans oogenesis. Development 134, 2227–2236 (2007).
Rozental, R., Srinivas, M. & Spray, D.C. How to close a gap junction channel. in Connexins Methods and Protocols (eds. Bruzzone, R. & Giaume, C.) 447–476 (Humana Press, Totowa, New Jersey, 2001).
Simpson, I., Rose, B. & Loewenstein, W.R. Size limit of molecules permeating the junctional membrane channels. Science 195, 294–296 (1977).
Altun, Z.F. & Hall, D.H. Handbook of C. elegans anatomy. http://www.wormatlas.org/handbook/contents.htm. (2005).
Levin, M. Isolation and community: a review of the role of gap-junctional communication in embryonic patterning. J. Membr. Biol. 185, 177–192 (2002).
Bossinger, O. & Schierenberg, E. Cell-cell communication in the embryo of Caenorhabditis elegans. Dev. Biol. 151, 401–409 (1992).
Bossinger, O. & Schierenberg, E. The use of fluorescent marker dyes for studying intercellular communication in nematode embryos. Int. J. Dev. Biol. 40, 431–439 (1996).
Lopresti, V., Macagno, E.R. & Levinthal, C. Structure and development of neuronal connections in isogenic organisms: transient gap junctions between growing optic axons and lamina neuroblasts. Proc. Natl. Acad. Sci. USA 71, 1098–1102 (1974).
Wolszon, L.R., Gao, W.Q., Passani, M.B. & Macagno, E.R. Growth cone “collapse” in vivo: are inhibitory interactions mediated by gap junctions? J. Neurosci. 14, 999–1010 (1994).
Montoro, R.J. & Yuste, R. Gap junctions in developing neocortex: a review. Brain Res. Brain Res. Rev. 47, 216–226 (2004).
Starich, T.A., Miller, A., Nguyen, R.L., Hall, D.H. & Shaw, J.E. The Caenorhabditis elegans innexin INX-3 is localized to gap junctions and is essential for embryonic development. Dev. Biol. 256, 403–417 (2003).
Chuang, C.F., Vanhoven, M.K., Fetter, R.D., Verselis, V.K. & Bargmann, C.I. An innexin-dependent cell network establishes left-right neuronal asymmetry in C. elegans. Cell 129, 787–799 (2007).
Nagayama, S. et al. In vivo simultaneous tracing and Ca2+ imaging of local neuronal circuits. Neuron 53, 789–803 (2007).
Acknowledgements
Imaging experiments involving two-photon excitation were performed at the Live Cell Imaging Core Facility of University of Texas Southwestern Medical Center and directed by K. Luby-Phelps, who also provided critical comments on the manuscript. We thank the Caenorhabditis Genetics Center for providing worm strains and X. Wang (University of Texas Southwestern) for providing the microinjection apparatus. We thank the Welch Foundation (I-1510) and the US National Institutes of Health for financial support.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1–8 , Supplementary Methods (PDF 1579 kb)
Supplementary movie 1.
Dye transfer in an early 4-cell C. elegans embryo after uncaging EMS. (MOV 776 kb)
Supplementary movie 2.
Dye transfer in a late 4-cell C. elegans embryo after uncaging EMS. (MOV 1088 kb)
Supplementary movie 3.
Restricted dye diffusion into the P3 cell (interphase) of developing C. elegans embryos. (MOV 2528 kb)
Supplementary movie 4.
Restricted dye diffusion into the dividing P3 cell of developing C. elegans embryos. (MOV 1975 kb)
Supplementary movie 5.
Restricted dye diffusion into the P4 and D cells of developing C. elegans embryos. (MOV 2568 kb)
Supplementary movie 6.
Fluorescence enhancement after a global uncaging of a L1 larva labeled with dextran-CANPE-HCC. (MOV 1966 kb)
Supplementary movie 7.
Image dynamic cell-cell coupling in a living worm (L1 larva). (MOV 704 kb)
Rights and permissions
About this article
Cite this article
Guo, YM., Chen, S., Shetty, P. et al. Imaging dynamic cell-cell junctional coupling in vivo using Trojan-LAMP. Nat Methods 5, 835–841 (2008). https://doi.org/10.1038/nmeth.1238
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1238
This article is cited by
-
From the connectome to brain function
Nature Methods (2013)
-
Endogenous voltage gradients as mediators of cell-cell communication: strategies for investigating bioelectrical signals during pattern formation
Cell and Tissue Research (2013)
-
Inhibiting miRNA in Caenorhabditis elegans using a potent and selective antisense reagent
Silence (2010)
-
Spatial organization and signal transduction at intercellular junctions
Nature Reviews Molecular Cell Biology (2010)