Abstract
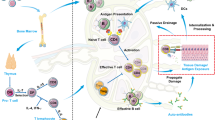
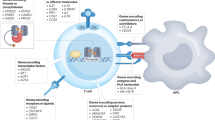
Approximately 5% of people in developed countries suffer from 1 of ∼80 classified autoimmune diseases. The sheer scale of the clinical problem captures the interests of health policy makers, academics, funding bodies and pharmaceutical companies in equal measure. In recent decades, immunologists have gained a good understanding of disease pathogenesis, which has led to the development of various potential therapies. The next challenge is to establish which therapies have superior efficacy, sustainability and safety. Therapeutic trials that depend on clinical end points are long lasting and expensive. In this Opinion article we offer a perspective on the future of clinical trial design in which the process is significantly shortened by making greater use of laboratory measures to determine therapeutic outcome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Descotes, J. Immunotoxicology: role in the safety assessment of drugs. Drug Saf. 28, 127–136 (2005).
Mandrup-Poulsen, T. et al. Disappearance and reappearance of islet cell cytoplasmic antibodies in cyclosporin-treated insulin-dependent diabetics. Lancet 1, 599–602 (1985).
Heidt, S. et al. Calcineurin inhibitors affect B cell antibody responses indirectly by interfering with T cell help. Clin. Exp. Immunol. 18 Nov 2009 (doi:10.1111/j.1365-2249.2009.04051.x).
Huurman, V. A. et al. Immunological efficacy of heat shock protein 60 peptide DiaPep277 therapy in clinical type I diabetes. Clin. Exp. Immunol. 152, 488–497 (2008).
Roep, B. O. et al. Auto- and alloimmune reactivity to human islet allografts transplanted into type 1 diabetic patients. Diabetes 48, 484–490 (1999).
Bougneres, P. F. et al. Limited duration of remission of insulin dependency in children with recent overt type I diabetes treated with low-dose cyclosporin. Diabetes 39, 1264–1272 (1990).
Herold, K. C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002).
Keymeulen, B. et al. Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N. Engl. J. Med. 352, 2598–2608 (2005).
Cernea, S. & Herold, K. C. Monitoring of antigen-specific CD8 T cells in patients with type 1 diabetes treated with antiCD3 monoclonal antibodies. Clin. Immunol. 16 Oct 2009 (doi:10.1016/j.clim.2009.09.005).
Herold, K. C. et al. Activation of human T cells by FcR nonbinding anti-CD3 mAb, hOKT3γ1(Ala–Ala). J. Clin. Invest. 111, 409–418 (2003).
Bisikirska, B., Colgan, J., Luban, J., Bluestone, J. A. & Herold, K. C. TCR stimulation with modified anti-CD3 mAb expands CD8+ T cell population and induces CD8+CD25+ Tregs. J. Clin. Invest. 115, 2904–2913 (2005).
Pescovitz, M. D. et al. Rituximab, B-lymphocyte depletion, and preservation of β-cell function. N. Engl. J. Med. 361, 2143–2152 (2009).
Ludvigsson, J. et al. GAD treatment and insulin secretion in recent-onset type 1 diabetes. N. Engl. J. Med. 359, 1909–1920 (2008).
Staeva-Vieira, T., Peakman, M. & von Herrath, M. Translational mini-review series on type 1 diabetes: immune-based therapeutic approaches for type 1 diabetes. Clin. Exp. Immunol. 148, 17–31 (2007).
Peakman, M. et al. Characterization of preparations of GAD65, proinsulin, and the islet tyrosine phosphatase IA-2 for use in detection of autoreactive T-cells in type 1 diabetes: report of phase II of the Second International Immunology of Diabetes Society Workshop for Standardization of T-cell assays in type 1 diabetes. Diabetes 50, 1749–1754 (2001).
Roep, B. O. et al. Autoreactive T cell responses in insulin-dependent (type 1) diabetes mellitus. Report of the first international workshop for standardization of T cell assays. J. Autoimmun. 13, 267–282 (1999).
Babel, N., Reinke, P. & Volk, H. D. Lymphocyte markers and prediction of long-term renal allograft acceptance. Curr. Opin. Nephrol. Hypertens. 18, 489–494 (2009).
Seyfert-Margolis, V. et al. Analysis of T-cell assays to measure autoimmune responses in subjects with type 1 diabetes: results of a blinded controlled study. Diabetes 55, 2588–2594 (2006).
Schloot, N. C. et al. Comparison of cytokine ELISpot assay formats for the detection of islet antigen autoreactive T cells. Report of the third immunology of diabetes society T-cell workshop. J. Autoimmun. 21, 365–376 (2003).
Janetzki, S. et al. “MIATA” — minimal information about T cell assays. Immunity 31, 527–528 (2009).
Herold, K. C. et al. Validity and reproducibility of measurement of islet autoreactivity by T-cell assays in subjects with early type 1 diabetes. Diabetes 58, 2588–2595 (2009).
Weis, J. H. Allergy test might have avoided drug-trial disaster. Nature 441, 150 (2006).
Bresson, D. et al. Anti-CD3 and nasal proinsulin combination therapy enhances remission from recent-onset autoimmune diabetes by inducing Tregs. J. Clin. Invest. 116, 1371–1381 (2006).
Valle, A. et al. Rapamycin prevents and breaks the anti-CD3-induced tolerance in NOD mice. Diabetes 58, 875–881 (2009).
Hayday, A. C. & Peakman, M. The habitual, diverse and surmountable obstacles to human immunology research. Nature Immunol. 9, 575–580 (2008).
Arechiga, A. F. et al. Cutting edge: the PTPN22 allelic variant associated with autoimmunity impairs B cell signaling. J. Immunol. 182, 3343–3347 (2009).
Dendrou, C. A. et al. Cell-specific protein phenotypes for the autoimmune locus IL2RA using a genotype-selectable human bioresource. Nature Genet. 41, 1011–1015 (2009).
Tabrizi, M. A. & Roskos, L. K. Preclinical and clinical safety of monoclonal antibodies. Drug Discov. Today 12, 540–547 (2007).
Stebbings, R. et al. “Cytokine storm” in the phase I trial of monoclonal antibody TGN1412: better understanding the causes to improve preclinical testing of immunotherapeutics. J. Immunol. 179, 3325–3331 (2007).
Thrower, S. L. et al. Proinsulin peptide immunotherapy in type 1 diabetes: report of a first-in-man phase I safety study. Clin. Exp. Immunol. 155, 156–165 (2009).
Roep, B. O., Atkinson, M. & von Herrath, M. Satisfaction (not) guaranteed: re-evaluating the use of animal models of type 1 diabetes. Nature Rev. Immunol. 4, 989–997 (2004).
van de Linde, P. et al. Mechanisms of antibody immunotherapy on clonal islet reactive T cells. Hum. Immunol. 67, 264–273 (2006).
Pearson, T. et al. Non-obese diabetic-recombination activating gene-1 (NOD–Rag1null) interleukin (IL)-2 receptor common γ chain (IL2rγnull) null mice: a radioresistant model for human lymphohaematopoietic engraftment. Clin. Exp. Immunol. 154, 270–284 (2008).
Shultz, L. D., Ishikawa, F. & Greiner, D. L. Humanized mice in translational biomedical research. Nature Rev. Immunol. 7, 118–130 (2007).
Nicoletti, F. et al. Serum concentrations of the interferon-γ-inducible chemokine IP-10/CXCL10 are augmented in both newly diagnosed Type I diabetes mellitus patients and subjects at risk of developing the disease. Diabetologia 45, 1107–1110 (2002).
Pfleger, C. et al. Relation of circulating concentrations of chemokine receptor CCR5 ligands to C-peptide, proinsulin and HbA1c and disease progression in type 1 diabetes. Clin. Immunol. 128, 57–65 (2008).
Pfleger, C. et al. Association of IL-1ra and adiponectin with C-peptide and remission in patients with type 1 diabetes. Diabetes 57, 929–937 (2008).
Lai, Y., Chen, C. & Linn, T. Innate immunity and heat shock response in islet transplantation. Clin. Exp. Immunol. 157, 1–8 (2009).
Padmos, R. C. et al. Distinct monocyte gene-expression profiles in autoimmune diabetes. Diabetes 57, 2768–2773 (2008).
Allen, J. S. et al. Plasmacytoid dendritic cells are proportionally expanded at diagnosis of type 1 diabetes and enhance islet autoantigen presentation to T-cells through immune complex capture. Diabetes 58, 138–145 (2009).
Beyan, H. et al. Altered monocyte cyclo-oxygenase response in non-obese diabetic mice. Clin. Exp. Immunol. 155, 304–310 (2009).
Arif, S. et al. Autoreactive T cell responses show proinflammatory polarization in diabetes but a regulatory phenotype in health. J. Clin. Invest. 113, 451–463 (2004).
Voltarelli, J. C. et al. Autologous nonmyeloablative hematopoietic stem cell transplantation in newly diagnosed type 1 diabetes mellitus. JAMA 297, 1568–1576 (2007).
Simon, G. et al. Murine antithymocyte globulin therapy alters disease progression in NOD mice by a time-dependent induction of immunoregulation. Diabetes 57, 405–414 (2008).
Lawson, J. M. et al. Increased resistance to CD4+CD25hi regulatory T cell-mediated suppression in patients with type 1 diabetes. Clin. Exp. Immunol. 154, 353–359 (2008).
Lindley, S. et al. Defective suppressor function in CD4+CD25+ T-cells from patients with type 1 diabetes. Diabetes 54, 92–99 (2005).
Schneider, A. et al. The effector T cells of diabetic subjects are resistant to regulation via CD4+FOXP3+ regulatory T cells. J. Immunol. 181, 7350–7355 (2008).
Mannering, S. I. et al. The A-chain of insulin is a hot-spot for CD4+ T cell epitopes in human type 1 diabetes. Clin. Exp. Immunol. 156, 226–231 (2009).
Spencer, J. & Peakman, M. Post-mortem analysis of islet pathology in type 1 diabetes illuminates the life and death of the β cell. Clin. Exp. Immunol. 155, 125–127 (2009).
Planas, R. et al. Gene expression profiles for the human pancreas and purified islets in type 1 diabetes: new findings at clinical onset and in long-standing diabetes. Clin. Exp. Immunol. 159, 23–44 (2009).
Hu, C., Wong, F. S. & Wen, L. Translational mini-review series on B cell-directed therapies: B cell-directed therapy for autoimmune diseases. Clin. Exp. Immunol. 157, 181–190 (2009).
Crome, S. Q., Wang, A. Y. & Levings, M. K. Translational mini-review series on Th17 cells: function and regulation of human T helper 17 cells in health and disease. Clin. Exp. Immunol. 11 Nov 2009 (doi:10.1111/j.1365-2249.2009.04037.x).
Koenders, M. I. & van den Berg, W. B. Translational mini-review series on Th17 cells: are T helper 17 cells really pathogenic in autoimmunity? Clin. Exp. Immunol. 11 Nov 2009 (doi:10.1111/j.1365-2249.2009.04039.x).
Pinkse, G. G. et al. Autoreactive CD8 T cells associated with β cell destruction in type 1 diabetes. Proc. Natl Acad. Sci. USA 102, 18425–18430 (2005).
Skowera, A. et al. CTLs are targeted to kill β cells in patients with type 1 diabetes through recognition of a glucose-regulated preproinsulin epitope. J. Clin. Invest. 118, 3390–3402 (2008).
Monti, P. et al. Islet transplantation in patients with autoimmune diabetes induces homeostatic cytokines that expand autoreactive memory T cells. J. Clin. Invest. 118, 1806–1814 (2008).
Willcox, A., Richardson, S. J., Bone, A. J., Foulis, A. K. & Morgan, N. G. Analysis of islet inflammation in human type 1 diabetes. Clin. Exp. Immunol. 155, 173–181 (2009).
Bougneres, P. F. et al. Factors associated with early remission of type I diabetes in children treated with cyclosporine. N. Engl. J. Med. 318, 663–670 (1988).
Martin, S. et al. Development of type 1 diabetes despite severe hereditary B-lymphocyte deficiency. N. Engl. J. Med. 345, 1036–1040 (2001).
Huurman, V. A. et al. Cellular islet autoimmunity associates with clinical outcome of islet cell transplantation. PLoS ONE 3, e2435 (2008).
Hilbrands, R. et al. Differences in baseline lymphocyte counts and autoreactivity are associated with differences in outcome of islet cell transplantation in type 1 diabetic patients. Diabetes 58, 2267–2276 (2009).
Bingley, P. J. et al. Combined analysis of autoantibodies improves prediction of IDDM in islet cell antibody-positive relatives. Diabetes 43, 1304–1310 (1994).
Verge, C. F. et al. Combined use of autoantibodies (IA-2 autoantibody, GAD autoantibody, insulin autoantibody, cytoplasmic islet cell antibodies) in type 1 diabetes: Combinatorial Islet Autoantibody Workshop. Diabetes 47, 1857–1866 (1998).
Verge, C. F. et al. Prediction of type I diabetes in first-degree relatives using a combination of insulin, GAD, and ICA512bdc/IA-2 autoantibodies. Diabetes 45, 926–933 (1996).
Brusko, T. M., Putnam, A. L. & Bluestone, J. A. Human regulatory T cells: role in autoimmune disease and therapeutic opportunities. Immunol. Rev. 223, 371–390 (2008).
Gregori, S., Battaglia, M. & Roncarolo, M. G. Re-establishing immune tolerance in type 1 diabetes via regulatory T cells. Novartis Found. Symp. 292, 174–183 (2008).
Sanda, S., Roep, B. O. & von Herrath, M. Islet antigen specific IL-10+ immune responses but not CD4+CD25+FoxP3+ cells at diagnosis predict glycaemic control in type 1 diabetes. Clin. Immunol. 127, 138–143 (2008).
Effects of insulin in relatives of patients with type 1 diabetes mellitus. N. Engl. J. Med. 346, 1685–1691 (2002).
Skyler, J. S. et al. Effects of oral insulin in relatives of patients with type 1 diabetes: The Diabetes Prevention Trial — Type 1. Diabetes Care 28, 1068–1076 (2005).
Harrison, L. C. et al. Pancreatic β-cell function and immune responses to insulin after administration of intranasal insulin to humans at risk for type 1 diabetes. Diabetes Care 27, 2348–2355 (2004).
Pfleger, C., Meierhoff, G., Kolb, H. & Schloot, N. C. Association of T-cell reactivity with β-cell function in recent onset type 1 diabetes patients. J. Autoimmun. 9 Sep 2009 (doi:10.1016/j.jaut.2009.08.004).
Acknowledgements
B.O.R. is a recipient of a Vici award from The Netherlands Organization for Health Research and Development (ZonMW) and is supported by the Dutch Diabetes Research Foundation. M.P. acknowledges financial support from the Department of Health through the National Institute for Health Research comprehensive Biomedical Research Centre award to Guy's & St Thomas' National Health Service Foundation Trust in partnership with King's College London and from the Juvenile Diabetes Research Foundation. Both B.O.R. and M.P. receive funding from the European Union 7th Framework Programme Large-scale Focused Collaborative Research Project on Natural Immunomodulators as Novel Immunotherapies for Type 1 Diabetes.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
M.P. has no competing financial interests. B.O.R. is a member of the Medical Advisory Boards of Diamyd Medical and Andromeda Biotech.
Related links
Glossary
- Biomarker
-
A substance or objective measurement that relates to a biological status. It indicates a specific disease state, risk of the disease, amelioration of disease on treatment or susceptibility to disease amelioration by a specific treatment. A biomarker can be an analyte measured in the blood, a cellular activity, a gene polymorphism or a radiographic feature. For example, multiple islet cell-specific autoantibodies are biomarkers that predict type 1 diabetes risk.
- Humanized mouse
-
A mouse carrying functioning human genes, cells, tissues or organs that are introduced by transgenesis, injection or transplantation. For example, an immune-deficient mouse transgenically expressing susceptibility genes for type 1 diabetes and reconstituted with T cells from a patient with the disease and human islets of Langerhans can be used to study relevant autoimmune processes.
- Surrogate end point
-
A biomarker that, when altered by treatment, indicates a clinical benefit. It is typically an analyte in the blood, such as could be represented by reduction in relevant effector T cell activation or the appearance of regulatory T cells.
Rights and permissions
About this article
Cite this article
Roep, B., Peakman, M. Surrogate end points in the design of immunotherapy trials: emerging lessons from type 1 diabetes. Nat Rev Immunol 10, 145–152 (2010). https://doi.org/10.1038/nri2705
Issue Date:
DOI: https://doi.org/10.1038/nri2705
This article is cited by
-
First-in-human, double-blind, randomized phase 1b study of peptide immunotherapy IMCY-0098 in new-onset type 1 diabetes
BMC Medicine (2023)
-
Prioritizing Early Disease Intervention
Therapeutic Innovation & Regulatory Science (2023)
-
The parasite-derived peptide FhHDM-1 activates the PI3K/Akt pathway to prevent cytokine-induced apoptosis of β-cells
Journal of Molecular Medicine (2021)
-
Immunological biomarkers for the development and progression of type 1 diabetes
Diabetologia (2018)
-
Combinatorial detection of autoreactive CD8+ T cells with HLA-A2 multimers: a multi-centre study by the Immunology of Diabetes Society T Cell Workshop
Diabetologia (2018)