Abstract
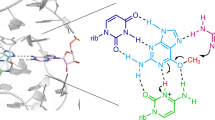
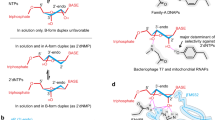
We used mutants of RNase T1 and the Rp isomer of a thiosubstituted substrate to determine stereospecific thioeffects on catalysis. The analysis reveals subtle structural and functional changes in the Lntermolecular transition state interactions. Tyr 38 contributes to catalysis through a hydrogen bond with the pro-Rp oxygen. Y38F RNase T1 prefers the Rp thiosubstituted analog over the natural phosphodiester substrate that is favored by the wild type enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fersht, A.R. Enzyme Structure and Mechanism (W.H. Freeman and Company, New York; 1985).
Eckstein, F. Ann. Rev. Biochem. 54, 367–402 (1985).
Milligan, J.F. & Uhlenbeck, O.C. Biochemistry 28, 2849–2855 (1989).
Dahm, S.C. & Uhlenbeck, O.C. Biochemistry 30, 9464–9469 (1991).
Eckstein, F., Schulz, H.H., Rüterjans, H., Haar, W. & Maurer, W. Biochemistry 11, 3507–3512 (1972).
Heinemann, U. & Saenger, W. Nature 299, 27–31 (1982).
Walz, F.G., Osterman, H.L. & Libertin, C. Arch. Biochem. Biophys. 195, 95–102 (1979).
Steyaert, J. Eur. J. Biochem. 247, 1–11 (1997).
Heinemann, U. & Hahn, U. In Protein–Nucleic Acid Interactions, 111–141 (MacMillan, London; 1989).
Zegers, I. et al. Nature Struct. Biol. 5, 280–283 (1998).
Steyaert, J., Hallenga, K., Wyns, L. & Stanssens, P. Biochemistry 29, 9064–9072 (1990).
Heydenreich, A. et al. Eur. J. Biochem. 218, 1005–1012 (1993).
Steyaert, J. & Wyns, L. J. Mol. Biol. 229, 770–781 (1993).
Steyaert, J., Haikal, A.F., Wyns, L. & Stanssens, P. Biochemistry 30, 8666–8670 (1991).
Doumen, J. et al. Protein Sci. 5, 1523–1530 (1996).
Steyaert, J., Wyns, L. & Stanssens, P. Biochemistry 30, 8661–8665 (1991).
Steyaert, J., Opsomer, C., Wyns, L. & Stanssens, P. Biochemistry 30, 494–499 (1991).
Horovitz, A. Folding & Design 1, 121–126 (1996).
Lienhard, G.E. Science 180, 149–154 (1973).
Herschlag, D., Piccirilli, J.A. & Cech, T.R. Biochemistry 30, 4844–4854 (1991).
Fersht, A.R. Biochemistry 27, 1577–1580 (1988).
Saenger, W. Principles of Nucleic Acid Structure (Springer-Verlag, New York; 1984).
Fersht, A.R. et al. Nature 314, 235–238 (1985).
Wilkinson, A.J., Fersht, A.R., Blow, D.M. & Winter, G. Biochemistry 22, 3581–3586 (1983).
Gregoret, L.M., Rader, S.D., Fletterick, R.J. & Cohen, F.E. Protein: Struct. Funct. Genet. 9, 99–107 (1991).
Herschlag, D. & Jencks, W.P. J. Am. Chem. Soc. 111, 7587–7596 (1989).
Hengge, A.C. & Cleland, W.W. J. Am. Chem. Soc. 112, 7421–7422 (1990).
Burgers, P.M.J. & Eckstein, F. Biochemistry 18, 592–596 (1979).
Almer, H. & Strömberg, R. Tetrahedron Lett. 294, 3723–3726 (1991).
Oivanen, M., Ora, M., Almer, H., Strömberg, R. & Lönnberg, H.J. Org. Chem. 60, 5620–5627 (1995).
Almer, H. & Strömberg, R. J. Am. Chem. Soc. 118, 7921–7928 (1996).
Almer, H., Stawinski, J., Strömberg, R. & Thelin, M. J. Org. Chem. 57, 6163–6169 (1992).
Pace, C.N., Vajdos, F., Fee, L., Grimsley, G. & Gray, T. Prot. Sci. 4, 2411–2423 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Loverix, S., Winquist, A., Strömberg, R. et al. An engineered ribonuclease preferring phosphorothioate RNA. Nat Struct Mol Biol 5, 365–368 (1998). https://doi.org/10.1038/nsb0598-365
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0598-365
This article is cited by
-
Structural and functional basis for RNA cleavage by Ire1
BMC Biology (2011)
-
A stepwise 2′-hydroxyl activation mechanism for the bacterial transcription termination factor Rho helicase
Nature Structural & Molecular Biology (2009)