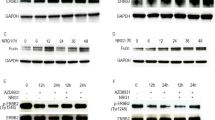
Abstract
Cancer progression is an abnormal form of tissue repair characterized by chronic inflammation. IκB kinase-β (IKKβ) required for nuclear factor-κB (NF-κB) activation plays a critical role in this process. Using EOC cells isolated from malignant ovarian cancer ascites and solid tumors, we identified IKKβ as a major factor promoting a functional TLR–MyD88–NF-κB pathway that confers to EOC cell the capacity to constitutively secrete proinflammatory/protumor cytokines and therefore promoting tumor progression and chemoresistance. Furthermore, we describe for the first time the identification of the microRNA hsa-miR-199a as a regulator of IKKβ expression. Our study describes the property of ovarian cancer cells to enhance the inflammatory microenvironment as a result of the expression of an active IKKβ pathway. Identification of these markers in patients' tumor samples may facilitate the adequate selection of treatment and open new venues for the development of effective therapy for chemoresistant ovarian cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Aggarwal BB . (2003). Signalling pathways of the TNF superfamily: a double-edged sword. Nat Rev Immunol 3: 745–756.
Balkwill F . (2004). Cancer and the chemokine network. Nat Rev Cancer 4: 540–550.
Balkwill F, Coussens LM . (2004). Cancer: an inflammatory link. Nature 431: 405–406.
Barnhart BC, Peter ME . (2003). The TNF receptor 1: a split personality complex. Cell 114: 148–150.
Baumgartner B, Weber M, Quirling M, Fischer C, Page S, Adam M et al. (2002). Increased IkappaB kinase activity is associated with activated NF-kappaB in acute myeloid blasts. Leukemia 16: 2062–2071.
Chen R, Alvero AB, Silasi DA, Mor G . (2007). Inflammation, cancer and chemoresistance: taking advantage of the Toll-like receptor signaling pathway. Am J Reprod Immunol 57: 93–107.
Coussens LM, Werb Z . (2002). Inflammation and cancer. Nature 420: 860–867.
Duan Z, Lamendola DE, Penson RT, Kronish KM, Seiden MV . (2002). Overexpression of IL-6 but not IL-8 increases paclitaxel resistance of U-2OS human osteosarcoma cells. Cytokine 17: 234–242.
Flick MB, O'Malley D, Rutherford T, Rodov S, Kamsteeg M, Hao XY et al. (2004). Apoptosis-based evaluation of chemosensitivity in ovarian cancer patients. J Soc Gynecol Investig 11: 252–259.
Fukata M, Michelsen KS, Eri R, Thomas LS, Hu B, Lukasek K et al. (2005). Toll-like receptor-4 is required for intestinal response to epithelial injury and limiting bacterial translocation in a murine model of acute colitis. Am J Physiol Gastrointest Liver Physiol 288: G1055–G1065.
Greten FR, Eckmann L, Greten TF, Park JM, Li ZW, Egan LJ et al. (2004). IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118: 285–296.
Greten FR, Karin M . (2004). The IKK/NF-kappaB activation pathway-a target for prevention and treatment of cancer. Cancer Lett 206: 193–199.
Hacker H, Karin M . (2006). Regulation and function of IKK and IKK-related kinases. Sci STKE 2006: re13.
Harroch S, Gothelf Y, Revel M, Chebath J . (1995). 5′ upstream sequences of MyD88, an IL-6 primary response gene in M1 cells: detection of functional IRF-1 and Stat factors binding sites. Nucleic Acids Res 23: 3539–3546.
Hu MC, Lee DF, Xia W, Golfman LS, Ou-Yang F, Yang JY et al. (2004). IkappaB kinase promotes tumorigenesis through inhibition of forkhead FOXO3a. Cell 117: 225–237.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C et al. (2006). Cancer statistics, 2006. CA Cancer J Clin 56: 106–130.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ . (2007). Cancer statistics, 2007. CA Cancer J Clin 57: 43–66.
Jiang D, Liang J, Fan J, Yu S, Chen S, Luo Y et al. (2005). Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med 11: 1173–1179.
Kamsteeg M, Rutherford T, Sapi E, Hanczaruk B, Shahabi S, Flick M et al. (2003). Phenoxodiol—an isoflavone analog—induces apoptosis in chemoresistant ovarian cancer cells. Oncogene 22: 2611–2620.
Karin M, Cao Y, Greten FR, Li ZW . (2002). NF-kappaB in cancer: from innocent bystander to major culprit. Nat Rev Cancer 2: 301–310.
Kelly MG, Alvero AB, Chen R, Silasi DA, Abrahams VM, Chan S et al. (2006). TLR-4 signaling promotes tumor growth and paclitaxel chemoresistance in ovarian cancer. Cancer Res 66: 3859–3868.
Leung CH, Grill SP, Lam W, Gao W, Sun HD, Cheng YC . (2006). Eriocalyxin B inhibits nuclear factor-kappaB activation by interfering with the binding of both p65 and p50 to the response element in a noncompetitive manner. Mol Pharmacol 70: 1946–1955.
Li L, Aggarwal BB, Shishodia S, Abbruzzese J, Kurzrock R . (2004). Nuclear factor-kappaB and IkappaB kinase are constitutively active in human pancreatic cells, and their down-regulation by curcumin (diferuloylmethane) is associated with the suppression of proliferation and the induction of apoptosis. Cancer 101: 2351–2362.
Li Q, Van Antwerp D, Mercurio F, Lee KF, Verma IM . (1999). Severe liver degeneration in mice lacking the IkappaB kinase 2 gene. Science 284: 321–325.
Li Q, Verma IM . (2002). NF-kappaB regulation in the immune system. Nat Rev Immunol 2: 725–734.
Liu B, Park E, Zhu F, Bustos T, Liu J, Shen J et al. (2006). A critical role for I kappaB kinase alpha in the development of human and mouse squamous cell carcinomas. Proc Natl Acad Sci USA 103: 17202–17207.
Ludwig L, Kessler H, Wagner M, Hoang-Vu C, Dralle H, Adler G et al. (2001). Nuclear factor-kappaB is constitutively active in C-cell carcinoma and required for RET-induced transformation. Cancer Res 61: 4526–4535.
Luo JL, Kamata H, Karin M . (2005). IKK/NF-kappaB signaling: balancing life and death—a new approach to cancer therapy. J Clin Invest 115: 2625–2632.
Maeda S, Chang L, Li ZW, Luo JL, Leffert H, Karin M . (2003). IKKbeta is required for prevention of apoptosis mediated by cell-bound but not by circulating TNFalpha. Immunity 19: 725–737.
Maeda S, Kamata H, Luo JL, Leffert H, Karin M . (2005). IKKbeta couples hepatocyte death to cytokine-driven compensatory proliferation that promotes chemical hepatocarcinogenesis. Cell 121: 977–990.
Mercurio F, Murray BW, Shevchenko A, Bennett BL, Young DB, Li JW et al. (1999). IkappaB kinase (IKK)-associated protein 1, a common component of the heterogeneous IKK complex. Mol Cell Biol 19: 1526–1538.
Nakanishi C, Toi M . (2005). Nuclear factor-kappaB inhibitors as sensitizers to anticancer drugs. Nat Rev Cancer 5: 297–309.
O'Dwyer PJ, Moyer JD, Suffness M, Harrison Jr SD, Cysyk R, Hamilton TC et al. (1994). Antitumor activity and biochemical effects of aphidicolin glycinate (NSC 303812) alone and in combination with cisplatin in vivo. Cancer Res 54: 724–729.
Pikarsky E, Porat RM, Stein I, Abramovitch R, Amit S, Kasem S et al. (2004). NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature 431: 461–466.
Pull SL, Doherty JM, Mills JC, Gordon JI, Stappenbeck TS . (2005). Activated macrophages are an adaptive element of the colonic epithelial progenitor niche necessary for regenerative responses to injury. Proc Natl Acad Sci USA 102: 99–104.
Rakoff-Nahoum S, Medzhitov R . (2007). Regulation of spontaneous intestinal tumorigenesis through the adaptor protein MyD88. Science 317: 124–127.
Rakoff-Nahoum S, Paglino J, Eslami-Varzaneh F, Edberg S, Medzhitov R . (2004). Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118: 229–241.
Romieu-Mourez R, Landesman-Bollag E, Seldin DC, Traish AM, Mercurio F, Sonenshein GE . (2001). Roles of IKK kinases and protein kinase CK2 in activation of nuclear factor-kappaB in breast cancer. Cancer Res 61: 3810–3818.
Rothwarf DM, Karin M . (1999). The NF-kappa B activation pathway: a paradigm in information transfer from membrane to nucleus. Sci STKE 1999: RE1.
Schwartz PE . (2002). Current diagnosis and treatment modalities for ovarian cancer. Cancer Treat Res 107: 99–118.
Shishodia S, Aggarwal BB . (2004). Nuclear factor-kappaB: a friend or a foe in cancer? Biochem Pharmacol 68: 1071–1080.
Tamatani T, Azuma M, Aota K, Yamashita T, Bando T, Sato M . (2001). Enhanced IkappaB kinase activity is responsible for the augmented activity of NF-kappaB in human head and neck carcinoma cells. Cancer Lett 171: 165–172.
Yang J, Richmond A . (2001). Constitutive IkappaB kinase activity correlates with nuclear factor-kappaB activation in human melanoma cells. Cancer Res 61: 4901–4909.
Acknowledgements
This study was supported in part by funds from the Discovery to Cure Program, Nicholas Brady and Sands Foundation. We thank the support of Drs Mark Landers, Amy Cumeo and Heather White from Invitrogen on the micro-RNA studies. We thank Dr Yung-Chi Cheng from Pharmacology Department, Yale University for providing Eriocalyxin B. We also thank Dr Sankar Ghosh from Immunology Department, Yale University for sharing pBIIX-LUC.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, R., Alvero, A., Silasi, D. et al. Regulation of IKKβ by miR-199a affects NF-κB activity in ovarian cancer cells. Oncogene 27, 4712–4723 (2008). https://doi.org/10.1038/onc.2008.112
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.112
Keywords
This article is cited by
-
TNFα-Induced Altered miRNA Expression Links to NF-κB Signaling Pathway in Endometriosis
Inflammation (2023)
-
Analysis of microRNA-199a-3p expression in CD4+ T cells of systemic lupus erythematosus
Clinical Rheumatology (2023)
-
TWIST1 induces proteasomal degradation of β-catenin during the differentiation of ovarian cancer stem-like cells
Scientific Reports (2022)
-
High expression level of miR-1260 family in the peripheral blood of patients with ovarian carcinoma
Journal of Ovarian Research (2021)
-
miR-155 and miR-146a collectively regulate meningitic Escherichia coli infection-mediated neuroinflammatory responses
Journal of Neuroinflammation (2021)