Abstract
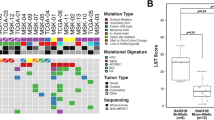
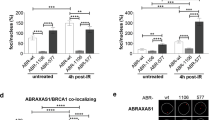
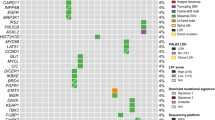
Germline mutations in two major susceptibility genes, BRCA1 and BRCA2, account for nearly 20% of familial breast cancers. A majority of the remaining genetic factors involved in heritable breast cancer susceptibility are, however, unknown. Recently, a new BRCA1-interacting protein, receptor associated protein 80 (RAP80), was identified. RAP80 plays an important role in BRCA1-mediated DNA damage responses (DDRs) by recruiting BRCA1 to DNA double-strand breaks (DSBs). A comprehensive screening of DNA from affected index cases of 112 BRCA1/BRCA2 mutation-negative Finnish breast cancer families revealed altogether 10 alterations in RAP80, one of which, c.241-243delGAA, resulted in a single glutamic acid deletion at residue 81 in a highly conserved region of ubiquitin interaction motif 1. The resultant delE81 protein product displayed significantly reduced ubiquitin binding and DSB localization. Expression of the RAP80 delE81 allele impaired both BRCA1 and ABRA1 DSB recruitment, thus compromising BRCA1-mediated DDR signaling. Compared with wild-type RAP80, expression of the delE81 allele was associated with a significant increase in cytogenetically detectable chromosomal aberrations, particularly chromatid breaks. Although evidently quite rare, these results suggest that critical constitutional mutations in RAP80 abrogate DDR function and may be involved in genetic predisposition to cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Akbari MR, Ghadirian P, Robidoux A, Foumani M, Sun Y, Royer R et al. (2009). Germline RAP80 mutations and susceptibility to breast cancer. Breast Cancer Res Treat 113: 377–381.
Allinen M, Huusko P, Mantyniemi S, Launonen V, Winqvist R . (2001). Mutation analysis of the CHK2 gene in families with hereditary breast cancer. Br J Cancer 85: 209–212.
Baer R, Ludwig T . (2002). The BRCA1/BARD1 heterodimer, a tumor suppressor complex with ubiquitin E3 ligase activity. Curr Opin Genet Dev 12: 86–91.
Cantor SB, Bell DW, Ganesan S, Kass EM, Drapkin R, Grossman S et al. (2001). BACH1, a novel helicase-like protein, interacts directly with BRCA1 and contributes to its DNA repair function. Cell 105: 149–160.
Celeste A, Petersen S, Romanienko PJ, Fernandez-Capetillo O, Chen HT, Sedelnikova OA et al. (2002). Genomic instability in mice lacking histone H2AX. Science 296: 922–927.
Chen J, Silver DP, Walpita D, Cantor SB, Gazdar AF, Tomlinson G et al. (1998). Stable interaction between the products of the BRCA1 and BRCA2 tumor suppressor genes in mitotic and meiotic cells. Mol Cell 2: 317–328.
Erkko H, Xia B, Nikkil J, Schleutker J, Syrj K, Mannermaa A et al. (2007). A recurrent mutation in PALB2 in Finnish cancer families. Nature 446: 316–319.
Greenberg RA . (2008). Recognition of DNA double strand breaks by the BRCA1 tumor suppressor network. Chromosoma 117: 305–317.
Greenberg RA, Sobhian B, Pathania S, Cantor SB, Nakatani Y, Livingston DM . (2006). Multifactorial contributions to an acute DNA damage response by BRCA1/BARD1-containing complexes. Genes Dev 20: 34–46.
Harper JW, Elledge SJ . (2007). The DNA damage response: ten years after. Mol Cell 28: 739–745.
Hashizume R, Fukuda M, Maeda I, Nishikawa H, Oyake D, Yabuki Y et al. (2001). The RING heterodimer BRCA1-BARD1 is a ubiquitin ligase inactivated by a breast cancer-derived mutation. J Biol Chem 276: 14537–14540.
Heikkinen K, Karppinen SM, Soini Y, Mäkinen M, Winqvist R . (2003). Mutation screening of Mre11 complex genes: Indication of RAD50 involvement in breast and ovarian cancer susceptibility. J Med Genet 40: e131.
Heikkinen K, Rapakko K, Karppinen SM, Erkko H, Knuutila S, Lundan T et al. (2006). RAD50 and NBS1 are breast cancer susceptibility genes associated with genomic instability. Carcinogenesis 27: 1593–1599.
Huang TT, D’Andrea AD . (2006). Regulation of DNA repair by ubiquitylation. Nat Rev Mol Cell Biol 7: 323–334.
Huen MSY, Grant R, Manke I, Minn K, Yu X, Yaffe MB et al. (2007). RNF8 transduces the DNA-damage signal via histone ubiquitylation and checkpoint protein assembly. Cell 131: 901–914.
Huusko P, Pääkkönen K, Launonen V, Pöyhönen M, Blanco G, Kauppila A et al. (1998). Evidence of founder mutations in Finnish BRCA1 and BRCA2 families. Am J Hum Genet 62: 1544–1548.
Isola J, DeVries S, Chu L, Ghazvini S, Waldman F . (1994). Analysis of changes in DNA sequence copy number by comparative genomic hybridization in archival paraffin-embedded tumor samples. Am J Pathol 145: 1301–1308.
Karppinen SM, Erkko H, Reini K, Pospiech H, Heikkinen K, Rapakko K et al. (2006). Identification of a common polymorphism in the TopBP1 gene associated with hereditary susceptibility to breast and ovarian cancer. Eur J Cancer 42: 2647–2652.
Kim H, Chen J, Yu X . (2007a). Ubiquitin-binding protein RAP80 mediates BRCA1-dependent DNA damage response. Science 316: 1202–1205.
Kim H, Huang J, Chen J . (2007b). CCDC98 is a BRCA1-BRCT domain-binding protein involved in the DNA damage response. Nat Struct Mol Biol 14: 710–715.
King MC, Marks JH, Mandell JB . (2003). Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 302: 643–646.
Kolas NK, Chapman JR, Nakada S, Ylanko J, Chahwan R, Sweeney FD et al. (2007). Orchestration of the DNA-damage response by the RNF8 ubiquitin ligase. Science 318: 1637–1640.
Körkkö J, Annunen S, Pihlajamaa T, Prockop DJ, Ala-Kokko L . (1998). Conformation sensitive gel electrophoresis for simple and accurate detection of mutations: Comparison with denaturing gradient gel electrophoresis and nucleotide sequencing. Proc Natl Acad Sci USA 95: 1681–1685.
Liu Z, Wu J, Yu X . (2007). CCDC98 targets BRCA1 to DNA damage sites. Nat Struct Mol Biol 14: 716–720.
Lou Z, Minter-Dykhouse K, Franco S, Gostissa M, Rivera MA, Celeste A et al. (2006). MDC1 maintains genomic stability by participating in the amplification of ATM-dependent DNA damage signals. Mol Cell 21: 187–200.
Mailand N, Bekker-Jensen S, Faustrup H, Melander F, Bartek J, Lukas C et al. (2007). RNF8 ubiquitylates histones at DNA double-strand breaks and promotes assembly of repair proteins. Cell 131: 887–900.
Moynahan ME, Chiu JW, Koller BH, Jasin M . (1999). Brca1 controls homology-directed DNA repair. Mol Cell 4: 511–518.
Novak DJ, Sabbaghian N, Maillet P, Chappuis PO, Foulkes WD, Tischkowitz M . (2008). Analysis of the genes coding for the BRCA1-interacting proteins, RAP80 and abraxas (CCDC98), in high-risk, non-BRCA1/2, multiethnic breast cancer cases. Breast Cancer Res Treat. 10.1007/s10549-008-0134-y.
Osorio A, Barroso A, Garcia MJ, Martinez-Delgado B, Urioste M, Benitez J . (2009). Evaluation of the BRCA1 interacting genes RAP80 and CCDC98 in familial breast cancer susceptibility. Breast Cancer Res Treat 113: 371–376.
Patel KJ, Yu VPCC, Lee H, Corcoran A, Thistlethwaite FC, Evans MJ et al. (1998). Involvement of Brca2 in DNA repair. Mol Cell 1: 347–357.
Rahman N, Stratton MR . (1998). The genetics of breast cancer susceptibility. Annu Rev Genet 32: 95–121.
Rapakko K, Allinen M, Syrjäkoski K, Vahteristo P, Huusko P, Vähäkangas K et al. (2001). Germline TP53 alterations in Finnish breast cancer families are rare and occur at conserved mutation-prone sites. Br J Cancer 84: 116–119.
Rogakou EP, Boon C, Redon C, Bonner WM . (1999). Megabase chromatin domains involved in DNA double-strand breaks in vivo. J Cell Biol 146: 905–916.
Ruffner H, Joazeiro CAP, Hemmati D, Hunter T, Verma IM . (2001). Cancer-predisposing mutations within the RING domain of BRCA1: loss of ubiquitin protein ligase activity and protection from radiation hypersensitivity. Proc Natl Acad Sci USA 98: 5134–5139.
Schwab M, Claas A, Savelyeva L . (2002). BRCA2: a genetic risk factor for breast cancer. Cancer Lett 175: 1–8.
Scully R, Ganesan S, Vlasakova K, Chen J, Socolovsky M, Livingston DM . (1999). Genetic analysis of BRCA1 function in a defined tumor cell line. Mol Cell 4: 1093–1099.
Seal S, Thompson D, Renwick A, Elliott A, Kelly P, Barfoot R et al. (2006). Truncating mutations in the Fanconi anemia J gene BRIP1 are low-penetrance breast cancer susceptibility alleles. Nat Genet 38: 1239–1241.
Sobhian B, Shao G, Lilli DR, Culhane AC, Moreau LA, Xia B et al. (2007). RAP80 targets BRCA1 to specific ubiquitin structures at DNA damage sites. Science 316: 1198–1202.
Stratton MR, Rahman N . (2008). The emerging landscape of breast cancer susceptibility. Nat Genet 40: 17–22.
Swanson KA, Kang RS, Stamenova SD, Hicke L, Radhakrishnan I . (2003). Solution structure of Vps27 UIM–ubiquitin complex important for endosomal sorting and receptor downregulation. EMBO J 22: 4597–4606.
Tuhkanen H, Anttila M, Kosma VM, Ylä-Herttuala S, Heinonen S, Kuronen A et al. (2004). Genetic alterations in the peritumoral stromal cells of malignant and borderline epithelial ovarian tumors as indicated by allelic imbalance on chromosome 3P. Int J Cancer 109: 247–252.
Wang B, Elledge SJ . (2007). Ubc13/Rnf8 ubiquitin ligases control foci formation of the Rap80/Abraxas/Brca1/Brcc36 complex in response to DNA damage. Proc Natl Acad Sci USA 104: 20759–20763.
Wang B, Matsuoka S, Ballif BA, Zhang D, Smogorzewska A, Gygi SP et al. (2007). Abraxas and RAP80 form a BRCA1 protein complex required for the DNA damage response. Science 316: 1194–1198.
Wong AKC, Ormonde PA, Pero R, Chen Y, Lian L, Salada G et al. (1998). Characterization of a carboxy-terminal BRCA1 interacting protein. Oncogene 17: 2279–2285.
Wooster R, Weber BL . (2003). Breast and ovarian cancer. N Engl J Med 348: 2339–2347.
Xu B, Kim S, Kastan MB . (2001). Involvement of Brca1 in S-phase and G2-phase checkpoints after ionizing irradiation. Mol Cell Biol 21: 3445–3450.
Xu X, Weaver Z, Linke SP, Li C, Gotay J, Wang XW et al. (1999). Centrosome amplification and a defective G2–M cell cycle checkpoint induce genetic instability in BRCA1 exon 11 isoform–deficient cells. Mol Cell 3: 389–395.
Yan J, Jetten AM . (2008). RAP80 and RNF8, key players in the recruitment of repair proteins to DNA damage sites. Cancer Lett 271: 179–190.
Yan J, Kim YS, Yang XP, Li LP, Liao G, Xia F et al. (2007). The ubiquitin-interacting motif containing protein RAP80 interacts with BRCA1 and functions in DNA damage repair response. Cancer Res 67: 6647–6656.
Yu X, Wu LC, Bowcock AM, Aronheim A, Baer R . (1998). The C-terminal (BRCT) domains of BRCA1 interact in vivo with CtIP, a protein implicated in the CtBP pathway of transcriptional repression. J Biol Chem 273: 25388–25392.
Zhao GY, Sonoda E, Barber LJ, Oka H, Murakawa Y, Yamada K et al. (2007). A critical role for the ubiquitin-conjugating enzyme Ubc13 in initiating homologous recombination. Mol Cell 25: 663–675.
Acknowledgements
We would like to thank Dr Aki Mustonen, Dr Jaakko Ignatius, Dr Ylermi Soini, Dr Mervi Grip and Dr Arja Jukkola-Vuorinen for their contribution, and nurses Kari Mononen and Outi Kajula for their help in patient contacts and Ms Helmi Konola for technical assistance. We also gratefully acknowledge Dr Junjie Chen for the antibodies to ABRA1, and Dr David Livingston for critical discussion of the manuscript before submission. The Finnish Breast Cancer Group, the Cancer Foundation of Northern Finland, the Orion-Farmos Research Foundation, the Academy of Finland, the Finnish Cancer Society, the Foundation for the Finnish Cancer Institute and the Sigrid Juselius Foundation to RW, the NIH training grant T32-GM-007229 to KAC and the National Cancer Institute K08 award 1K08CA106597-01, the Sidney Kimmel Foundation Scholar Award, the Mary Kay Ash Charitable Foundation Cancer Research Grant and funds from the Abramson Family Cancer Research Institute to RAG financially supported this study.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Nikkilä, J., Coleman, K., Morrissey, D. et al. Familial breast cancer screening reveals an alteration in the RAP80 UIM domain that impairs DNA damage response function. Oncogene 28, 1843–1852 (2009). https://doi.org/10.1038/onc.2009.33
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.33
Keywords
This article is cited by
-
Active Site Gate Dynamics Modulate the Catalytic Activity of the Ubiquitination Enzyme E2-25K
Scientific Reports (2018)
-
RAP80, ubiquitin and SUMO in the DNA damage response
Journal of Molecular Medicine (2017)
-
Assessment of changes in the brca2 and p53 genes in breast invasive ductal carcinoma in northeast Brazil
Biological Research (2014)
-
BRCA1 tumor suppressor network: focusing on its tail
Cell & Bioscience (2012)
-
Expression of the BRCA1 complex member BRE predicts disease free survival in breast cancer
Breast Cancer Research and Treatment (2012)