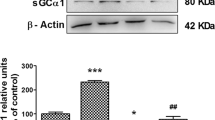
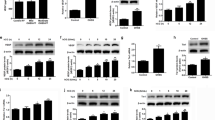
Abstract
Gonadotropin-releasing hormone (GnRH) receptor expression is often elevated in ovarian cancer, but its potential role in ovarian cancer metastasis has just begun to be revealed. Cadherin switching is a crucial step during tumorigenesis, particularly in metastasis. Here, we showed that GnRH is an inducer of E- to P-cadherin switching, which is reminiscent of that seen during ovarian tumor progression. Overexpression of P-cadherin significantly enhanced, whereas knockdown of P-cadherin reduced migration and invasion regardless of E-cadherin expression, suggesting that inappropriate expression of P-cadherin contributes to the invasive phenotype. These effects of P-cadherin were mediated by activation of the Rho GTPases, Rac1, and Cdc42, through accumulation of p120 catenin (p120ctn) in the cytoplasm. The use of p120ctn small interfering RNA or chimeric cadherin construct to inhibit p120ctn expression and cytoplasmic localization, respectively, resulted in significant inhibition of cell migration and invasion, with a concomitant reduction in Rac1 and Cdc42 activation, confirming that the effect was p120ctn specific. Similarly, the migratory/invasive phenotype could be reversed by expression of dominant-negative Rac1 and Cdc42. These results identify for the first time cadherin switching and p120ctn signaling as important targets of GnRH function and as novel mediators of invasiveness and tumor progression in ovarian cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout











Similar content being viewed by others
References
Cavallaro U, Christofori G . (2004). Multitasking in tumor progression: signaling functions of cell adhesion molecules. Ann N Y Acad Sci 1014: 58–66.
Chen CL, Cheung LW, Lau MT, Choi JH, Auersperg N, Wang HS et al. (2007). Differential role of gonadotropin-releasing hormone on human ovarian epithelial cancer cell invasion. Endocrine 31: 311–320.
Chen GT, Tai CT, Yeh LS, Yang TC, Tsai HD . (2002). Identification of the cadherin subtypes present in the human peritoneum and endometriotic lesions: potential role for P-cadherin in the development of endometriosis. Mol Reprod Dev 62: 289–294.
Cheng CK, Leung PC . (2005). Molecular biology of gonadotropin-releasing hormone (GnRH)-I, GnRH-II, and their receptors in humans. Endocr Rev 26: 283–306.
Cheung LW, Leung PC, Wong AS . (2006). Gonadotropin-releasing hormone promotes ovarian cancer cell invasiveness through c-Jun NH2-terminal kinase-mediated activation of matrix metalloproteinase (MMP)-2 and MMP-9. Cancer Res 66: 10902–10910.
Cheung LW, Wong AS . (2008). Gonadotropin-releasing hormone: GnRH receptor signaling in extrapituitary tissues. FEBS J 275: 5479–5495.
Choi JH, Park JT, Davidson B, Morin PJ, Shih Ie M, Wang TL . (2008). Jagged-1 and Notch3 juxtacrine loop regulates ovarian tumor growth and adhesion. Cancer Res 68: 5716–5723.
Clevers H . (2006). Wnt/beta-catenin signaling in development and disease. Cell 127: 469–480.
Emons G, Pahwa GS, Brack C, Sturm R, Oberheuser F, Knuppen R . (1989). Gonadotropin releasing hormone binding sites in human epithelial ovarian carcinomata. Eur J Cancer Clin Oncol 25: 215–221.
Fagotto F, Gumbiner BM . (1996). Cell contact-dependent signaling. Dev Biol 180: 445–454.
Feltes CM, Kudo A, Blaschuk O, Byers SW . (2002). An alternatively spliced cadherin-11 enhances human breast cancer cell invasion. Cancer Res 62: 6688–6697.
Irmer G, Burger C, Muller R, Ortmann O, Peter U, Kakar SS et al. (1995). Expression of the messenger RNAs for luteinizing hormone-releasing hormone (LHRH) and its receptor in human ovarian epithelial carcinoma. Cancer Res 55: 817–822.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T et al. (2008). Cancer statistics, 2008. CA Cancer J Clin 58: 71–96.
Kang SK, Cheng KW, Nathwani PS, Choi KC, Leung PC . (2000). Autocrine role of gonadotropin-releasing hormone and its receptor in ovarian cancer cell growth. Endocrine 13: 297–304.
Kim KY, Choi KC, Auersperg N, Leung PC . (2006). Mechanism of gonadotropin-releasing hormone (GnRH)-I and -II-induced cell growth inhibition in ovarian cancer cells: role of the GnRH-I receptor and protein kinase C pathway. Endocr Relat Cancer 13: 211–220.
Li SL, Vuagnat B, Gruaz NM, Eshkol A, Sizonenko PC, Aubert ML . (1994). Binding kinetics of the long-acting gonadotropin-releasing hormone (GnRH) antagonist antide to rat pituitary GnRH receptors. Endocrinology 135: 45–52.
Maeda M, Johnson E, Mandal SH, Lawson KR, Keim SA, Svoboda RA et al. (2006). Expression of inappropriate cadherins by epithelial tumor cells promotes endocytosis and degradation of E-cadherin via competition for p120(ctn). Oncogene 25: 4595–4604.
Miranda KC, Khromykh T, Christy P, Le TL, Gottardi CJ, Yap AS et al. (2001). A dileucine motif targets E-cadherin to the basolateral cell surface in Madin-Darby canine kidney and LLC-PK1 epithelial cells. J Biol Chem 276: 22565–22572.
Nieman MT, Prudoff RS, Johnson KR, Wheelock MJ . (1999). N-cadherin promotes motility in human breast cancer cells regardless of their E-cadherin expression. J Cell Biol 147: 631–644.
Nobes CD, Hall A . (1999). Rho GTPases control polarity, protrusion, and adhesion during cell movement. J Cell Biol 144: 1235–1244.
Noren NK, Liu BP, Burridge K, Kreft B . (2000). p120 catenin regulates the actin cytoskeleton via Rho family GTPases. J Cell Biol 150: 567–580.
Olson MF, Ashworth A, Hall A . (1995). An essential role for Rho, Rac, and Cdc42 GTPases in cell cycle progression through G1. Science 269: 1270–1272.
Paredes J, Correia AL, Ribeiro AS, Albergaria A, Milanezi F, Schmitt FC . (2007). P-cadherin expression in breast cancer: a review. Breast Cancer Res 9: 214.
Patel IS, Madan P, Getsios S, Bertrand MA, MacCalman CD . (2003). Cadherin switching in ovarian cancer progression. Int J Cancer 106: 172–177.
Pece S, Gutkind JS . (2000). Signaling from E-cadherins to the MAPK pathway by the recruitment and activation of epidermal growth factor receptors upon cell-cell contact formation. J Biol Chem 275: 41227–41233.
Perez-Moreno M, Fuchs E . (2006). Catenins: keeping cells from getting their signals crossed. Dev Cell 11: 601–612.
Qian X, Karpova T, Sheppard AM, McNally J, Lowy DR . (2004). E-cadherin-mediated adhesion inhibits ligand-dependent activation of diverse receptor tyrosine kinases. EMBO J 23: 1739–1748.
Sommers CL, Gelmann EP, Kemler R, Cowin P, Byers SW . (1994). Alterations in beta-catenin phosphorylation and plakoglobin expression in human breast cancer cells. Cancer Res 54: 3544–3552.
Soubry A, van Hengel J, Parthoens E, Colpaert C, Van Marck E, Waltregny D et al. (2005). Expression and nuclear location of the transcriptional repressor Kaiso is regulated by the tumor microenvironment. Cancer Res 65: 2224–2233.
Spentzos D, Cannistra SA, Grall F, Levine DA, Pillay K, Libermann TA et al. (2007). IGF axis gene expression patterns are prognostic of survival in epithelial ovarian cancer. Endocr Relat Cancer 14: 781–790.
Sundfeldt K . (2003). Cell-cell adhesion in the normal ovary and ovarian tumors of epithelial origin; an exception to the rule. Mol Cell Endocrinol 202: 89–96.
Suyama K, Shapiro I, Guttman M, Hazan RB . (2002). A signaling pathway leading to metastasis is controlled by N-cadherin and the FGF receptor. Cancer Cell 2: 301–314.
Taniuchi K, Nakagawa H, Hosokawa M, Nakamura T, Eguchi H, Ohigashi H et al. (2005). Overexpressed P-cadherin/CDH3 promotes motility of pancreatic cancer cells by interacting with p120ctn and activating rho-family GTPases. Cancer Res 65: 3092–3099.
Wheelock MJ, Shintani Y, Maeda M, Fukumoto Y, Johnson KR . (2008). Cadherin switching. J Cell Sci 121: 727–735.
Wong AS, Maines-Bandiera SL, Rosen B, Wheelock MJ, Johnson KR, Leung PC et al. (1999). Constitutive and conditional cadherin expression in cultured human ovarian surface epithelium: influence of family history of ovarian cancer. Int J Cancer 81: 180–188.
Acknowledgements
We thank Drs N Auersperg, A Hall, and C Gottardi for cell lines and plasmids. This work was supported by Canadian Institutes of Health Research grant (PCK Leung), and by Hong Kong Research Grant Council Grant 778108 and HKU Outstanding Young Researcher Award (AST Wong).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Cheung, L., Leung, P. & Wong, A. Cadherin switching and activation of p120 catenin signaling are mediators of gonadotropin-releasing hormone to promote tumor cell migration and invasion in ovarian cancer. Oncogene 29, 2427–2440 (2010). https://doi.org/10.1038/onc.2009.523
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.523
Keywords
This article is cited by
-
Increased expression of P-cadherin is an indicator of poor prognosis in breast cancer: a systematic review and meta-analysis
Breast Cancer Research and Treatment (2020)
-
MUC16 impacts tumor proliferation and migration through cytoplasmic translocation of P120-catenin in epithelial ovarian cancer cells: an original research
BMC Cancer (2019)
-
Cadherin-1 and cadherin-3 cooperation determines the aggressiveness of pancreatic ductal adenocarcinoma
British Journal of Cancer (2018)
-
Overexpression of CTNND1 in hepatocellular carcinoma promotes carcinous characters through activation of Wnt/β-catenin signaling
Journal of Experimental & Clinical Cancer Research (2016)
-
P-cadherin and the journey to cancer metastasis
Molecular Cancer (2015)