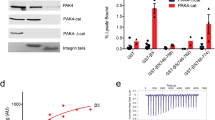
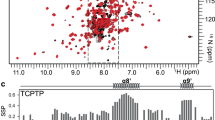
Abstract
Integrin-linked kinase (ILK) is a highly evolutionarily conserved intracellular protein that was originally identified as an integrin-interacting protein, and extensive genetic and biochemical studies have shown that ILK expression is vital during both embryonic development and tissue homeostasis. At the cellular and tissue levels, ILK regulates signaling pathways for cell adhesion-mediated cell survival (anoikis), apoptosis, proliferation and mitosis, migration, invasion, and vascularization and tumor angiogenesis. ILK also has central roles in cardiac and smooth-muscle contractility, and ILK dysregulation causes cardiomyopathies in humans. ILK protein levels are increased in several human cancers and often the expression level predicts poor patient outcome. Abundant evidence has accumulated suggesting that, of the diverse functions of ILK, some may require kinase activity whereas others depend on protein–protein interactions and are, therefore, independent of kinase activity. However, the past several years have seen an ongoing debate about whether ILK indeed functions as a protein serine/threonine kinase. This debate centers on the atypical protein kinase domain of ILK, which lacks some amino-acid residues thought to be essential for phosphotransferase activity. However, similar deficiencies are present in the catalytic domains of other kinases now known to possess protein kinase activity. Numerous studies have shown that ILK phosphorylates peptide substrates in vitro, corresponding to ILK-mediated phosphorylations in intact cells, and a recent report characterizing in vitro phosphotransferase activity of highly purified, full-length ILK, accompanied by detailed enzyme kinetic analyses, shows that, at least in vitro, ILK is a bona fide protein kinase. However, several genetic studies suggest that, not all biological functions of ILK require kinase activity, and that it can function as an adaptor/scaffold protein. Here, we review evidence for and against ILK being an active kinase, and provide a framework for strategies to further analyze the kinase and adaptor functions of ILK in different cellular contexts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Acconcia F, Barnes CJ, Singh RR, Talukder AH, Kumar R . (2007). Phosphorylation-dependent regulation of nuclear localization and functions of integrin-linked kinase. Proc Natl Acad Sci USA 104: 6782–6787.
Akhtar N, Marlow R, Lambert E, Schatzmann F, Lowe ET, Cheung J et al. (2009). Molecular dissection of integrin signalling proteins in the control of mammary epithelial development and differentiation. Development 136: 1019–1027.
Allen BG, Walsh MP . (1994). The biochemical basis of the regulation of smooth-muscle contraction. Trends Biochem Sci 19: 362–368.
Amano M, Ito M, Kimura K, Fukata Y, Chihara K, Nakano T et al. (1996). Phosphorylation and activation of myosin by Rho-associated kinase (Rho-kinase). J Biol Chem 271: 20246–20249.
Attwell S, Mills J, Troussard A, Wu C, Dedhar S . (2003). Integration of cell attachment, cytoskeletal localization, and signaling by integrin-linked kinase (ILK), CH-ILKBP, and the tumor suppressor PTEN. Mol Biol Cell 14: 4813–4825.
Balendran A, Casamayor A, Deak M, Paterson A, Gaffney P, Currie R et al. (1999). PDK1 acquires PDK2 activity in the presence of a synthetic peptide derived from the carboxyl terminus of PRK2. Curr Biol 9: 393–404.
Bendig G, Grimmler M, Huttner IG, Wessels G, Dahme T, Just S et al. (2006). Integrin-linked kinase, a novel component of the cardiac mechanical stretch sensor, controls contractility in the zebrafish heart. Genes Dev 20: 2361–2372.
Boppart MD, Burkin DJ, Kaufman SJ . (2011). Activation of AKT signaling promotes cell growth and survival in alpha7beta1 integrin-mediated alleviation of muscular dystrophy. Biochim Biophys Acta 1812: 439–446.
Boudeau J, Miranda-Saavedra D, Barton GJ, Alessi DR . (2006). Emerging roles of pseudokinases. Trends Cell Biol 16: 443–452.
Boulbes D, Chen CH, Shaikenov T, Agarwal NK, Peterson TR, Addona TA et al. (2010). Rictor phosphorylation on the Thr-1135 site does not require mammalian target of rapamycin complex 2. Mol Cancer Res 8: 896–906.
Cabodi S, del Pilar Camacho-Leal M, Di Stefano P, Defilippi P . (2010). Integrin signalling adaptors: not only figurants in the cancer story. Nat Rev Cancer 10: 858–870.
Chiswell BP, Zhang R, Murphy JW, Boggon TJ, Calderwood DA . (2008). The structural basis of integrin-linked kinase–PINCH interactions. Proc Natl Acad Sci USA 105: 20677–20682.
Delcommenne M, Tan C, Gray V, Rue L, Woodgett J, Dedhar S . (1998). Phosphoinositide-3-OH kinase-dependent regulation of glycogen synthase kinase 3 and protein kinase B/AKT by the integrin-linked kinase. Proc Natl Acad Sci USA 95: 11211–11216.
Deng JT, Van Lierop JE, Sutherland C, Walsh MP . (2001). Ca2+-independent smooth muscle contraction. A novel function for integrin-linked kinase. J Biol Chem 276: 16365–16373.
Deng JT, Sutherland C, Brautigan DL, Eto M, Walsh MP . (2002). Phosphorylation of the myosin phosphatase inhibitors, CPI-17 and PHI-1, by integrin-linked kinase. Biochem J 367: 517–524.
Dobreva I, Fielding A, Foster LJ, Dedhar S . (2008). Mapping the integrin-linked kinase interactome using SILAC. J Proteome Res 7: 1740–1749.
Erdődi F, Kiss E, Walsh MP, Stefansson B, Deng JT, Eto M et al. (2003). Phosphorylation of protein phosphatase type-1 inhibitory proteins by integrin-linked kinase and cyclic nucleotide-dependent protein kinases. Biochem Biophys Res Commun 306: 382–387.
Eswaran J, Patnaik D, Filippakopoulos P, Wang FW, Stein RL, Murray JW et al. (2009). Structure and functional characterization of the atypical human kinase haspin. Proc Natl Acad Sci USA 106: 20198–20203.
Eto M . (2009). Regulation of cellular protein phosphatase-1 (PP1) by phosphorylation of the CPI-17 family, C-kinase-activated PP1 inhibitors. J Biol Chem 284: 35273–35277.
Fielding AB, Dobreva I, McDonald PC, Foster LJ, Dedhar S . (2008). Integrin-linked kinase localizes to the centrosome and regulates mitotic spindle organization. J Cell Biol 180: 681–689.
Fielding AB, Lim S, Montgomery K, Dobreva I, Dedhar S . (2011). A critical role of integrin-linked kinase, ch-TOG and TACC3 in centrosome clustering in cancer cells. Oncogene 30: 521–534.
Filipenko NR, Attwell S, Roskelley C, Dedhar S . (2005). Integrin-linked kinase activity regulates Rac- and Cdc42-mediated actin cytoskeleton reorganization via alpha-PIX. Oncogene 38: 5837–5849.
Fukuda K, Gupta S, Chen K, Wu C, Qin J . (2009). The pseudoactive site of ILK is essential for its binding to α-parvin and localization to focal adhesions. Mol Cell 36: 819–830.
Grashoff C, Aszodi A, Sakai T, Hunziker EB, Fassler R . (2003). Integrin-linked kinase regulates chondrocyte shape and proliferation. EMBO Rep 4: 432–438.
Hanks SK, Quinn AM, Hunter T . (1988). The protein kinase family: conserved features and deduced phylogeny of the catalytic domains. Science 241: 42–52.
Hanks SK, Hunter T . (1995). Protein kinases 6. The eukaryotic protein kinase superfamily: kinase (catalytic) domain structure and classification. FASEB J 9: 576–596.
Hannigan GE, Leung-Hagesteijn C, Fitz-Gibbon L, Coppolino MG, Radeva G, Filmus J et al. (1996). Regulation of cell adhesion and anchorage-dependent growth by a new beta 1-integrin-linked protein kinase. Nature 379: 91–96.
Hannigan G, Troussard AA, Dedhar S . (2005). Integrin-linked kinase: a cancer therapeutic target unique among its ILK. Nat Rev Cancer 5: 51–63.
Hannigan GE, Coles JG, Dedhar S . (2007). Integrin-linked kinase at the heart of cardiac contractility, repair, and disease. Circ Res 100: 1408–1414.
Herman PK, Stack JH, DeModena JA, Emr SD . (1991). A novel protein kinase homolog essential for protein sorting to the yeast lysosome-like vacuole. Cell 64: 425–437.
Huang J, Mahavadi S, Sriwai W, Hu W, Murthy KS . (2006). Gi-coupled receptors mediate phosphorylation of CPI-17 and MLC20 via preferential activation of the PI3K/ILK pathway. Biochem J 396: 193–200.
Jin H, Sperka T, Herrlich P, Morrison H . (2006). Tumorigenic transformation by CPI-17 through inhibition of a merlin phosphatase. Nature 442: 576–579.
Knight ZA, Shokat KM . (2007). Chemical genetics: where genetics and pharmacology meet. Cell 128: 425–430.
Knoll R, Postel R, Wang J, Kratzner R, Hennecke G, Vacaru AM et al. (2007). Laminin-alpha4 and integrin-linked kinase mutations cause human cardiomyopathy via simultaneous defects in cardiomyocytes and endothelial cells. Circulation 116: 515–525.
Kogata N, Tribe RM, Fassler R, Way M, Adams RH . (2009). Integrin-linked kinase controls vascular wall formation by negatively regulating Rho/ROCK-mediated vascular smooth muscle cell contraction. Genes Dev 23: 2278–2283.
Kumar AS, Naruszewicz I, Wang P, Leung-Hagesteijn C, Hannigan GE . (2004). ILKAP regulates ILK signaling and inhibits anchorage-independent growth. Oncogene 23: 3454–3461.
Lange A, Wickstrom SA, Jakobson M, Zent R, Sainio K, Fassler R . (2009). Integrin-linked kinase is an adaptor with essential functions during mouse development. Nature 461: 1002–1006.
Legate KR, Montanez E, Kudlacek O, Fassler R . (2006). ILK, PINCH and parvin: the tIPP of integrin signalling. Nat Rev Mol Cell Biol 7: 20–31.
Leung-Hagesteijn C, Mahendra A, Naruszewicz I, Hannigan GE . (2001). Modulation of integrin signal transduction by ILKAP, a protein phosphatase 2C associating with the integrin-linked kinase, ILK1. EMBO J 20: 2160–2170.
Liu J, Costello PC, Pham NA, Pintillie M, Jabali M, Sanghera J et al. (2006). Integrin-linked kinase inhibitor KP-392 demonstrates clinical benefits in an orthotopic human non-small cell lung cancer model. J Thorac Oncol 1: 771–779.
Lorenz K, Grashoff C, Torka R, Sakai T, Langbein L, Bloch W et al. (2007). Integrin-linked kinase is required for epidermal and hair follicle morphogenesis. J Cell Biol 177: 501–513.
Lundgren K, Walworth N, Booher R, Dembski M, Kirschner M, Beach D . (1991). mik1 and wee1 cooperate in the inhibitory tyrosine phosphorylation of cdc2. Cell 64: 1111–1122.
Lynch DK, Ellis CA, Edwards PA, Hiles ID . (1999). Integrin-linked kinase regulates phosphorylation of serine 473 of protein kinase B by an indirect mechanism. Oncogene 18: 8024–8032.
Mackinnon AC, Qadota H, Norman KR, Moerman DG, Williams BD . (2002). C. elegans PAT-4/ILK functions as an adaptor protein within integrin adhesion complexes. Curr Biol 12: 787–797.
Maydan M, McDonald PC, Sanghera J, Yan J, Rallis C, Pinchin S et al. (2010). Integrin-linked kinase is a functional Mn2+-dependent protein kinase that regulates glycogen synthase kinase-3beta (GSK-3beta) phosphorylation. PLoS One 5: e12356.
McDonald PC, Fielding AB, Dedhar S . (2008a). Integrin-linked kinase—essential roles in physiology and cancer biology. J Cell Sci 121: 3121–3132.
McDonald PC, Oloumi A, Mills J, Dobreva I, Maidan M, Gray V et al. (2008b). Rictor and integrin-linked kinase interact and regulate Akt phosphorylation and cancer cell survival. Cancer Res 68: 1618–1624.
Montanez E, Wickstrom SA, Altstatter J, Chu H, Fassler R . (2009). Alpha-parvin controls vascular mural cell recruitment to vessel wall by regulating RhoA/ROCK signalling. EMBO J 28: 3132–3144.
Mukherjee K, Sharma M, Urlaub H, Bourenkov GP, Jahn R, Südhof TC et al. (2008). CASK functions as a Mg2+-independent neurexin kinase. Cell 133: 328–339.
Murányi A, MacDonald JA, Deng JT, Wilson DP, Haystead TA, Walsh MP et al. (2002). Phosphorylation of the myosin phosphatase target subunit by integrin-linked kinase. Biochem J 366: 211–216.
Muranyi AL, Dedhar S, Hogge DE . (2010). Targeting integrin linked kinase and FMS-like tyrosine kinase-3 is cytotoxic to acute myeloid leukemia stem cells but spares normal progenitors. Leuk Res 34: 1358–1365.
Murata-Hori M, Suizu F, Iwasaki T, Kikuchi A, Hosoya H . (1999). ZIP kinase identified as a novel myosin regulatory light chain kinase in HeLa cells. FEBS Lett 451: 81–84.
Niiro N, Ikebe M . (2001). Zipper-interacting protein kinase induces Ca(2+)-free smooth muscle contraction via myosin light chain phosphorylation. J Biol Chem 276: 29567–29574.
Nikolopoulos SN, Turner CE . (2001). Integrin-linked kinase (ILK) binding to paxillin LD1 motif regulates ILK localization to focal adhesions. J Biol Chem 276: 23499–23505.
Nikolopoulos SN, Turner CE . (2002). Molecular dissection of actopaxin–integrin-linked kinase–paxillin interactions and their role in subcellular localization. J Biol Chem 277: 1568–1575.
Oloumi A, Syam S, Dedhar S . (2006). Modulation of Wnt3a-mediated nuclear (beta)-catenin accumulation and activation by integrin-linked kinase in mammalian cells. Oncogene 25: 7747–7757.
Oloumi A, Maidan M, Lock FE, Tearle H, McKinney S, Muller WJ et al. (2010). Cooperative signaling between Wnt1 and integrin-linked kinase induces accelerated breast tumor development. Breast Cancer Res 12: R38.
Pasquali C, Bertschy-Meier D, Chabert C, Curchod ML, Arod C, Booth R et al. (2007). A chemical proteomics approach to phosphatidylinositol 3-kinase signaling in macrophages. Mol Cell Proteomics 6: 1829–1841.
Pereira JA, Benninger Y, Baumann R, Goncalves AF, Ozcelik M, Thurnherr T et al. (2009). Integrin-linked kinase is required for radial sorting of axons and Schwann cell remyelination in the peripheral nervous system. J Cell Biol 185: 147–161.
Persad S, Attwell S, Gray V, Delcommenne M, Troussard A, Sanghera J et al. (2000). Inhibition of integrin-linked kinase (ILK) suppresses activation of protein kinase B/Akt and induces cell cycle arrest and apoptosis of PTEN-mutant prostate cancer cells. Proc Natl Acad Sci USA 97: 3207–3212.
Persad S, Attwell S, Gray V, Mawji N, Deng JT, Leung D et al. (2001). Regulation of protein kinase B/Akt-serine 473 phosphorylation by integrin-linked kinase: critical roles for kinase activity and amino acids arginine 211 and serine 343. J Biol Chem 276: 27462–27469.
Pontier SM, Huck L, White DE, Rayment J, Sanguin-Gendreau V, Hennessy B et al. (2010). Integrin-linked kinase has a critical role in ErbB2 mammary tumor progression: implications for human breast cancer. Oncogene 29: 3374–3385.
Postel R, Vakeel P, Topczewski J, Knoll R, Bakkers J . (2008). Zebrafish integrin-linked kinase is required in skeletal muscles for strengthening the integrin–ECM adhesion complex. Dev Biol 318: 92–101.
Quelo I, Gauthier C, Hannigan GE, Dedhar S, St-Arnaud R . (2004). Integrin-linked kinase regulates the nuclear entry of the c-Jun coactivator α-NAC and its coactivation potency. J Biol Chem 279: 43893–43899.
Rallis C, Pinchin SM, Ish-Horowicz D . (2010). Cell-autonomous integrin control of Wnt and Notch signalling during somitogenesis. Development 137: 3591–3601.
Sakai T, Li S, Docheva D, Grashoff C, Sakai K, Kostka G et al. (2003). Integrin-linked kinase (ILK) is required for polarizing the epiblast, cell adhesion, and controlling actin accumulation. Genes Dev 17: 926–940.
Suh HN, Han HJ . (2011). Collagen I regulates the self-renewal of mouse embryonic stem cells through α2β1 integrin- and DDR1-dependent Bmi-1. J Cell Physiol (e-pub ahead of print 22 February 2011; doi: 10.1002/jcp.22697).
Tan C, Cruet-Hennequart S, Troussard A, Fazli L, Costello P, Sutton K et al. (2004). Regulation of tumor angiogenesis by integrin-linked kinase (ILK). Cancer Cell 5: 79–90.
Taylor SS, Kornev AP . (2010). Yet another ‘active’ pseudokinase, Erb3. Proc Natl Acad Sci USA 107: 8047–8048.
Troussard AA, Mawji NM, Ong C, Mui A, St -Arnaud R, Dedhar S . (2003). Conditional knock-out of integrin-linked kinase demonstrates an essential role in protein kinase B/Akt activation. J Biol Chem 278: 22374–22378.
Troussard AA, McDonald PC, Wederell ED, Mawji NM, Filipenko NR, Gelmon KA et al. (2006). Preferential dependence of breast cancer cells versus normal cells on integrin-linked kinase for protein kinase B/Akt activation and cell survival. Cancer Res 66: 393–403.
Wang HV, Chang LW, Brixius K, Wickstrom SA, Montanez E, Thievessen I et al. (2008). Integrin-linked kinase stabilizes myotendinous junctions and protects muscle from stress-induced damage. J Cell Biol 180: 1037–1049.
Weber LP, Van Lierop JE, Walsh MP . (1999). Ca2+-independent phosphorylation of myosin in rat caudal artery and chicken gizzard myofilaments. J Physiol 516: 805–824.
White DE, Cardiff RD, Dedhar S, Muller WJ . (2001). Mammary epithelial-specific expression of the integrin-linked kinase (ILK) results in the induction of mammary gland hyperplasias and tumors in transgenic mice. Oncogene 20: 7064–7072.
White DE, Coutu P, Shi YF, Tardif JC, Nattel S, St Arnaud R et al. (2006). Targeted ablation of ILK from the murine heart results in dilated cardiomyopathy and spontaneous heart failure. Genes Dev 20: 2355–2360.
Wickstrom SA, Lange A, Hess MW, Polleux J, Spatz JP, Kruger M et al. (2010a). Integrin-linked kinase controls microtubule dynamics required for plasma membrane targeting of caveolae. Dev Cell 19: 574–588.
Wickstrom SA, Lange A, Montanez E, Fassler R . (2010b). The ILK/PINCH/parvin complex: the kinase is dead, long live the pseudokinase!. EMBO J 29: 281–291.
Wilson DP, Sutherland C, Borman MA, Deng JT, MacDonald JA, Walsh MP . (2005). Integrin-linked kinase is responsible for Ca2+-independent myosin diphosphorylation and contraction of vascular smooth muscle. Biochem J 392: 641–648.
Yamaji S, Suzuki A, Sugiyama Y, Koide Y, Yoshida M, Kanamori H et al. (2001). A novel integrin-linked kinase-binding protein, affixin, is involved in the early stage of cell–substrate interaction. J Cell Biol 153: 1251–1264.
Yau CY, Wheeler JJ, Sutton KL, Hedley DW . (2005). Inhibition of integrin-linked kinase by a selective small molecule inhibitor, QLT0254, inhibits the PI3K/PKB/mTOR, Stat3, and FKHR pathways and tumor growth, and enhances gemcitabine-induced apoptosis in human orthotopic primary pancreatic cancer xenografts. Cancer Res 65: 1497–1504.
Younes MN, Kim S, Yigitbasi OG, Mandal M, Jasser SA, Dakak Yazici Y et al. (2005). Integrin-linked kinase is a potential therapeutic target for anaplastic thyroid cancer. Mol Cancer Ther 4: 1146–1156.
Zervas CG, Gregory SL, Brown NH . (2001). Drosophila integrin-linked kinase is required at sites of integrin adhesion to link the cytoskeleton to the plasma membrane. J Cell Biol 152: 1007–1018.
Acknowledgements
This work was supported by funding from the Canadian Institutes of Health Research (to SD and MPW), the Canadian Cancer Society Research Institute (to SD) and the National Health and Medical Research Council (NHMRC) of Australia (to GEH).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hannigan, G., McDonald, P., Walsh, M. et al. Integrin-linked kinase: Not so ‘pseudo’ after all. Oncogene 30, 4375–4385 (2011). https://doi.org/10.1038/onc.2011.177
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.177
Keywords
This article is cited by
-
Indoxyl sulfate- and P-cresol-induced monocyte adhesion and migration is mediated by integrin-linked kinase-dependent podosome formation
Experimental & Molecular Medicine (2022)
-
The kinase activity of integrin-linked kinase regulates cellular senescence in gastric cancer
Cell Death & Disease (2022)
-
Insight into the mechanism of molecular recognition between human Integrin-Linked Kinase and Cpd22 and its implication at atomic level
Journal of Computer-Aided Molecular Design (2022)
-
Novel protein pathways in development and progression of pulmonary sarcoidosis
Scientific Reports (2020)
-
A Deregulated PI3K-AKT Signaling Pathway in Patients with Colorectal Cancer
Journal of Gastrointestinal Cancer (2019)