Abstract
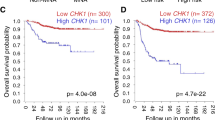
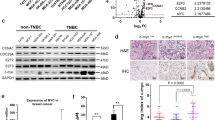
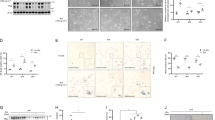
MYCN amplification is a major biomarker of poor prognosis, occurring in 25–30% of neuroblastomas. MYCN has contradictory roles in promoting cell growth and sensitizing cells to apoptosis. We have recently shown that p53 is a direct transcriptional target of MYCN in neuroblastoma and that p53-mediated apoptosis may be an important mechanism of MYCN-induced apoptosis. Although p53 mutations are rare in neuroblastoma at diagnosis, the p53/MDM2/p14ARF pathway is often inactivated through MDM2 amplification or p14ARF inactivation. We hypothesized that reactivation of p53 by inhibition of its negative regulator MDM2, using the MDM2-p53 antagonists Nutlin-3 and MI-63, will result in p53-mediated growth arrest and apoptosis especially in MYCN-amplified cells. Using the SHEP Tet21N MYCN-regulatable system, MYCN(−) cells were more resistant to both Nutlin-3 and MI-63 mediated growth inhibition and apoptosis compared with MYCN(+) cells and siRNA-mediated knockdown of MYCN in four MYCN-amplified cell lines resulted in decreased p53 expression and activation, as well as decreased levels of apoptosis following treatment with MDM2-p53 antagonists. In a panel of 18 neuroblastoma cell lines treated with Nutlin-3 and MI-63, the subset amplified for MYCN had a significantly lower mean GI50 value (50% growth inhibition) and increased caspase 3/7 activity compared with the non-MYCN-amplified group of cell lines, but p53 mutant cell lines were resistant to the antagonists regardless of MYCN status. We conclude that amplification or overexpression of MYCN sensitizes neuroblastoma cell lines with wild-type p53 to MDM2-p53 antagonists and that these compounds may therefore be particularly effective in treating high-risk MYCN-amplified disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Alt JR, Greiner TC, Cleveland JL, Eischen CM . (2003). Mdm2 haplo-insufficiency profoundly inhibits Myc-induced lymphomagenesis. Embo J 22: 1442–1450.
Amente S, Gargano B, Diolaiti D, Della Valle G, Lania L, Majello B . (2007). p14ARF interacts with N-Myc and inhibits its transcriptional activity. FEBS Lett 581: 821–825.
Barbieri E, Mehta P, Chen Z, Zhang L, Slack A, Berg S et al. (2006). MDM2 inhibition sensitizes neuroblastoma to chemotherapy-induced apoptotic cell death. Mol Cancer Ther 5: 2358–2365.
Bell E, Premkumar R, Carr J, Lu X, Lovat PE, Kees UR et al. (2006). The role of MYCN in the failure of MYCN amplified neuroblastoma cell lines to G1 arrest after DNA damage. Cell Cycle 5: 2639–2647.
Canner JA, Sobo M, Ball S, Hutzen B, DeAngelis S, Willis W et al. (2009). MI-63: a novel small-molecule inhibitor targets MDM2 and induces apoptosis in embryonal and alveolar rhabdomyosarcoma cells with wild-type p53. Br J Cancer 101: 774–781.
Carr-Wilkinson J, O'Toole K, Wood KM, Challen CC, Baker AG, Board JR et al. (2010). High frequency of p53/MDM2/p14ARF pathway abnormalities in relapsed neuroblastoma. Clin Cancer Res 16: 1108–1118.
Carr J, Bell E, Pearson AD, Kees UR, Beris H, Lunec J et al. (2006). Increased frequency of aberrations in the p53/MDM2/p14(ARF) pathway in neuroblastoma cell lines established at relapse. Cancer Res 66: 2138–2145.
Chen L, Iraci N, Gherardi S, Gamble LD, Wood KM, Perini G et al. (2010). p53 is a direct transcriptional target of MYCN in neuroblastoma. Cancer Res 70: 1377–1388.
Chen Z, Lin Y, Barbieri E, Burlingame S, Hicks J, Ludwig A et al. (2009). Mdm2 deficiency suppresses MYCN-Driven neuroblastoma tumorigenesis in vivo. Neoplasia 11: 753–762.
Chipuk JE, Green DR . (2006). Dissecting p53-dependent apoptosis. Cell Death Differ 13: 994–1002.
Cohn SL, Pearson AD, London WB, Monclair T, Ambros PF, Brodeur GM et al. (2009). The International Neuroblastoma Risk Group (INRG) classification system: an INRG Task Force report. J Clin Oncol 27: 289–297.
Cohn SL, Tweddle DA . (2004). MYCN amplification remains prognostically strong 20 years after its ‘clinical debut’. Eur J Cancer 40: 2639–2642.
Corvi R, Savelyeva L, Breit S, Wenzel A, Handgretinger R, Barak J et al. (1995). Non-syntenic amplification of MDM2 and MYCN in human neuroblastoma. Oncogene 10: 1081–1086.
Demidenko ZN, Korotchkina LG, Gudkov AV, Blagosklonny MV . (2010). Paradoxical suppression of cellular senescence by p53. Proc Natl Acad Sci USA 107: 9660–9664.
Efeyan A, Ortega-Molina A, Velasco-Miguel S, Herranz D, Vassilev LT, Serrano M . (2007). Induction of p53-dependent senescence by the MDM2 antagonist nutlin-3a in mouse cells of fibroblast origin. Cancer Res 67: 7350–7357.
Fulda S, Lutz W, Schwab M, Debatin KM . (2000). MycN sensitizes neuroblastoma cells for drug-triggered apoptosis. Med Pediatr Oncol 35: 582–584.
Goldman SC, Chen CY, Lansing TJ, Gilmer TM, Kastan MB . (1996). The p53 signal transduction pathway is intact in human neuroblastoma despite cytoplasmic localization. Am J Pathol 148: 1381–1385.
Hogarty MD . (2003). The requirement for evasion of programmed cell death in neuroblastomas with MYCN amplification. Cancer Lett 197: 173–179.
Hosoi G, Hara J, Okamura T, Osugi Y, Ishihara S, Fukuzawa M et al. (1994). Low frequency of the p53 gene mutations in neuroblastoma. Cancer 73: 3087–3093.
Kang JH, Rychahou PG, Ishola TA, Qiao J, Evers BM, Chung DH . (2006). MYCN silencing induces differentiation and apoptosis in human neuroblastoma cells. Biochem Biophys Res Commun 351: 192–197.
Kojima K, Konopleva M, Samudio IJ, Shikami M, Cabreira-Hansen M, McQueen T et al. (2005). MDM2 antagonists induce p53-dependent apoptosis in AML: implications for leukemia therapy. Blood 106: 3150–3159.
Korotchkina LG, Leontieva OV, Bukreeva EI, Demidenko ZN, Gudkov AV, Blagosklonny MV . (2010). The choice between p53-induced senescence and quiescence is determined in part by the mTOR pathway. Aging (Albany NY) 2: 344–352.
Lutz W, Stohr M, Schurmann J, Wenzel A, Lohr A, Schwab M . (1996). Conditional expression of N-myc in human neuroblastoma cells increases expression of alpha-prothymosin and ornithine decarboxylase and accelerates progression into S-phase early after mitogenic stimulation of quiescent cells. Oncogene 13: 803–812.
Maris JM, Hogarty MD, Bagatell R, Cohn SL . (2007). Neuroblastoma. Lancet 369: 2106–2120.
Maris JM, Matthay KK . (1999). Molecular biology of neuroblastoma. J Clin Oncol 17: 2264–2279.
Matthay KK, Villablanca JG, Seeger RC, Stram DO, Harris RE, Ramsay NK et al. (1999). Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. Children's Cancer Group. N Engl J Med 341: 1165–1173.
Michalak E, Villunger A, Erlacher M, Strasser A . (2005). Death squads enlisted by the tumour suppressor p53. Biochem Biophys Res Commun 331: 786–798.
Muller CR, Paulsen EB, Noordhuis P, Pedeutour F, Saeter G, Myklebost O . (2007). Potential for treatment of liposarcomas with the MDM2 antagonist Nutlin-3A. Int J Cancer 121: 199–205.
Petroni M, Veschi V, Prodosmo A, Rinaldo C, Massimi I, Carbonari M et al. (2011). MYCN sensitizes human neuroblastoma to apoptosis by HIPK2 activation through a DNA damage response. Mol Cancer Res 9: 67–77.
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D et al. (1990). New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82: 1107–1112.
Slack A, Chen Z, Tonelli R, Pule M, Hunt L, Pession A et al. (2005). The p53 regulatory gene MDM2 is a direct transcriptional target of MYCN in neuroblastoma. Proc Natl Acad Sci USA 102: 731–736.
Slack A, Shohet JM . (2005). MDM2 as a critical effector of the MYCN oncogene in tumorigenesis. Cell Cycle 4: 857–860.
Tweddle DA, Malcolm AJ, Cole M, Pearson AD, Lunec J . (2001). p53 cellular localization and function in neuroblastoma: evidence for defective G(1) arrest despite WAF1 induction in MYCN-amplified cells. Am J Pathol 158: 2067–2077.
Tweddle DA, Pearson AD, Haber M, Norris MD, Xue C, Flemming C et al. (2003). The p53 pathway and its inactivation in neuroblastoma. Cancer Lett 197: 93–98.
Van Maerken T, Rihani A, Dreidax D, De Clercq S, Yigit N, Marine JC et al. (2011). Functional analysis of the p53 pathway in neuroblastoma cells using the small-molecule MDM2 antagonist nutlin-3. Mol Cancer Ther 10: 983–993.
Van Maerken T, Speleman F, Vermeulen J, Lambertz I, De Clercq S, De Smet E et al. (2006). Small-molecule MDM2 antagonists as a new therapy concept for neuroblastoma. Cancer Res 66: 9646–9655.
van Noesel MM, Pieters R, Voute PA, Versteeg R . (2003). The N-myc paradox: N-myc overexpression in neuroblastomas is associated with sensitivity as well as resistance to apoptosis. Cancer Lett 197: 165–172.
van Noesel MM, Versteeg R . (2004). Pediatric neuroblastomas: genetic and epigenetic ‘danse macabre’. Gene 325: 1–15.
Vassilev LT, Vu BT, Graves B, Carvajal D, Podlaski F, Filipovic Z et al. (2004). In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303: 844–848.
Vogan K, Bernstein M, Leclerc JM, Brisson L, Brossard J, Brodeur GM et al. (1993). Absence of p53 gene mutations in primary neuroblastomas. Cancer Res 53: 5269–5273.
Wang P, Greiner TC, Lushnikova T, Eischen CM . (2006). Decreased Mdm2 expression inhibits tumor development induced by loss of ARF. Oncogene 25: 3708–3718.
Weiss WA, Aldape K, Mohapatra G, Feuerstein BG, Bishop JM . (1997). Targeted expression of MYCN causes neuroblastoma in transgenic mice. Embo J 16: 2985–2995.
Wenzel A, Cziepluch C, Hamann U, Schurmann J, Schwab M . (1991). The N-Myc oncoprotein is associated in vivo with the phosphoprotein Max(p20/22) in human neuroblastoma cells. Embo J 10: 3703–3712.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ et al. (1998). Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev 12: 2424–2433.
Acknowledgements
We thank the following for cell lines: Sue Cohn (NBLW and NBLS), Linda Harris (SJNB1), Penny Lovat (SHSY5Y, SHEP and IMR32), John Maris (NB69), Patrick Reynolds (SKNRA, SMSKCNR, LAN5, LAN6 and CHLA136), Manfred Schwab (LS and SHEP Tet21N), Rogier Versteeg (NGP), Barbara Spengler (SKNBe2C), Micro Ponzoni (GIMEN), Ursula Kees (PER108), Maria Lastowska (TR14), Clinton Stewart (NB1691) and Jean Bénard (SKNAS, IGRN91). We are grateful to Mike Cole (Newcastle Cancer Centre, Newcastle University) for his statistical advice and to Cancer Research UK for funding this work. This study is supported by Cancer Research UK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website ()
Rights and permissions
About this article
Cite this article
Gamble, L., Kees, U., Tweddle, D. et al. MYCN sensitizes neuroblastoma to the MDM2-p53 antagonists Nutlin-3 and MI-63. Oncogene 31, 752–763 (2012). https://doi.org/10.1038/onc.2011.270
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.270
Keywords
This article is cited by
-
LIN28B inhibition sensitizes cells to p53-restoring PPI therapy through unleashed translational suppression
Oncogenesis (2022)
-
Cell death-based treatment of neuroblastoma
Cell Death & Disease (2018)
-
Neuroblastoma treatment in the post-genomic era
Journal of Biomedical Science (2017)
-
RLIP76 Inhibition: A Promising Developmental Therapy for Neuroblastoma
Pharmaceutical Research (2017)
-
Effect of low doses of actinomycin D on neuroblastoma cell lines
Molecular Cancer (2016)