Abstract
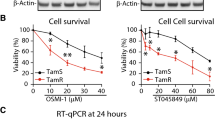
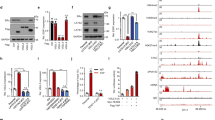
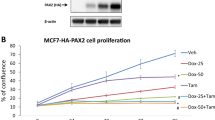
Tamoxifen resistance is a major problem in the treatment of estrogen receptor (ER)-positive patients. We have previously reported that hexamethylene bis-acetamide-inducible protein 1 (HEXIM1) inhibits ERα activity by competing with ERα for binding to cyclin T1, a subunit of positive transcription elongation b (P-TEFb). This results in the inhibition of the phosphorylation of RNA polymerase II (RNAPII) at serine 2 and the inhibition of transcription elongation of ERα target genes. As HEXIM1 can inhibit ER activity, we examined whether it has a critical role in the inhibitory effects of tamoxifen on ER. We observed that tamoxifen-induced HEXIM1 recruitment to the promoter region of ER target genes and decreased the recruitment of cyclin T1 and serine 2 phosphorylated RNAPII to the coding regions of these genes. Conversely, in cells wherein HEXIM1 expression has been downregulated we observed attenuation of the inhibitory effects of tamoxifen on estrogen-induced cyclin T1 recruitment to coding regions of ER target genes. As a consequence, downregulation of HEXIM1 resulted in the attenuation of the repressive effects of tamoxifen on estrogen-induced gene expression and proliferation. Conferring clinical relevance to our studies is our analysis of human breast cancer tissue samples that indicated association of lower expression of HEXIM1 with tumor recurrence in patients who received tamoxifen. Our studies provide a better understanding of the mechanistic basis for the inhibitory effect of tamoxifen on ER activity and may suggest new therapeutic targets for the treatment of tamoxifen-resistant breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- CCND1:
-
cyclin D1
- ERα:
-
estrogen receptor alpha
- E2:
-
17-beta estradiol
- HEXIM1:
-
hexamethylene-inducible gene-1
- P-TEFb:
-
positive transcription elongation factor b
- RNAP II:
-
RNA polymerase II
- TOT:
-
trans-hydroxy-tamoxifen
References
Anderson H, Bulun S, Smith I, Dowsett M . (2007). Predictors of response to aromatase inhibitors. J Steroid Biochem Mol Biol 106: 49–54.
Cleator SJ, Ahamed E, Coombes RC, Palmieri C . (2009). A 2009 update on the treatment of patients with hormone receptor-positive breast cancer. Clin Breast Cancer 9 (Suppl 1): S6–S17.
Deroo BJ, Korach KS . (2006). Estrogen receptors and human disease. J Clin Invest 116: 561–570.
Gruber CJ, Tschugguel W, Schneeberger C, Huber JC . (2002). Production and actions of estrogens. N Engl J Med 346: 340–352.
Hurvitz SA, Pietras RJ . (2008). Rational management of endocrine resistance in breast cancer. Cancer 9: 2385–2397.
Kusuhara M, Nagasaki K, Kimura K, Maass N, Manabe T, Ishikawa S et al. (1999). Cloning of hexamethylene-bis-acetamide-inducible transcript, HEXIM1, in human vascular smooth muscle cells. Biomed Res 20: 273–279.
Lahusen T, Henke RT, Kagan BL, Wellstein A, Riegel AT . (2009). The role and regulation of the nuclear receptor co-activator AIB1 in breast cancer. Breast Cancer Res Treat 116: 225–237.
Lavinsky RM, Jepsen K, Heinzel T, Torchia J, Mullen TM, Schiff R et al. (1998). Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes. Proc Natl Acad Sci USA 95: 2920–2925.
Lewis JS, Jordan VC . (2005). Selective estrogen receptor modulators (SERMs): mechanisms of anticarcinogenesis and drug resistance. Mutat Res 591: 247–263.
Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schutz G, Umesono K et al. (1995). The nuclear receptor superfamily: the second decade. Cell 83: 835–839.
Musgrove EA, Sutherland RL . (1994). Cell cycle control by steroid hormones. Semin Cancer Biol 5: 381–389.
Ogba N, Chaplin LJ, Doughman YQ, Fujinaga K, Montano MM . (2008). HEXIM1 regulates 17beta-estradiol/estrogen receptor-alpha-mediated expression of cyclin D1 in mammary cells via modulation of P-TEFb. Cancer Res 68: 7015–7024.
Osborne CK . (1998). Tamoxifen in the treatment of breast cancer. N Engl J Med 339: 1609–1618.
Riggins RB, Schrecengost RS, Guerrero MS, Bouton AH . (2007). Pathways to tamoxifen resistance. Cancer Lett 256: 1–24.
Ring A, Dowsett M . (2004). Mechanisms of tamoxifen resistance. Endocr Relat Cancer 11: 643–658.
Scott DJ, Parkes AT, Ponchel F, Cummings M, Poola I, Speirs V . (2007). Changes in expression of steroid receptors, their downstream target genes and their associated co-regulators during the sequential acquisition of tamoxifen resistance in vitro. Int J Oncol 31: 557–565.
Shiau AK, Barstad D, Loria PM, Cheng L, Kushner PJ, Agard DA et al. (1998). The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell 95: 927–937.
Webb P, Nguyen P, Kushner PJ . (2003). Differential SERM effects on corepressor binding dictate ERalpha activity in vivo. J Biol Chem 278: 6912–6920.
Wittmann BM, Fujinaga K, Deng H, Ogba N, Montano MM . (2005). The breast cell growth inhibitor, estrogen down regulated gene 1, modulates a novel functional interaction between estrogen receptor alpha and transcriptional elongation factor cyclin T1. Oncogene 24: 5576–5588.
Wittmann BM, Wang N, Montano MM . (2003). Identification of a novel inhibitor of breast cell growth that is down-regulated by estrogens and decreased in breast tumors. Cancer Res 63: 5151–5158.
Yager JD, Davidson NE . (2006). Estrogen carcinogenesis in breast cancer. N Engl J Med 354: 270–282.
Yik JH, Chen R, Nishimura R, Jennings JL, Link AJ, Zhou Q . (2003). Inhibition of P-TEFb (CDK9/Cyclin T) kinase and RNA polymerase II transcription by the coordinated actions of HEXIM1 and 7SK snRNA. Mol Cell 12: 971–982.
Zwart W, Rondaij M, Jalink K, Sharp ZD, Mancini MA, Neefjes J et al. (2009). Resistance to antiestrogen arzoxifene is mediated by overexpression of cyclin D1. Mol Endocrinol 23: 1335–1345.
Acknowledgements
We thank Wei Wang (CWRU Department of Epidemiology and Biostatistics) for help with construction of graphs representing the relationship between disease recurrence and expressions levels of HEXIM1 in breast tissue. This work was supported by National Institute of Health grant CA92440 to MMM and American Cancer Society grant RSG CCE-110689 to J.J.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Ketchart, W., Ogba, N., Kresak, A. et al. HEXIM1 is a critical determinant of the response to tamoxifen. Oncogene 30, 3563–3569 (2011). https://doi.org/10.1038/onc.2011.76
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.76
Keywords
This article is cited by
-
Non-epigenetic induction of HEXIM1 by DNMT1 inhibitors and functional relevance
Scientific Reports (2020)
-
SUPT6H controls estrogen receptor activity and cellular differentiation by multiple epigenomic mechanisms
Oncogene (2015)
-
Inhibition of metastasis by HEXIM1 through effects on cell invasion and angiogenesis
Oncogene (2013)
-
Complex effects of flavopiridol on the expression of primary response genes
Cell Division (2012)