Abstract
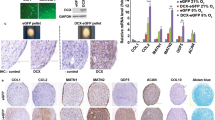
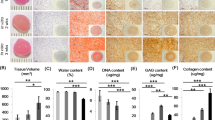
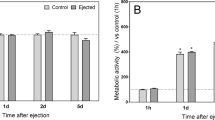
The long-term goal of the present study is to develop a clinically applicable approach to enhance natural repair mechanisms within cartilage lesions by targeting bone marrow-derived cells for genetic modification. To determine if bone marrow-derived cells infiltrating osteochondral defects could be transduced in situ, we implanted collagen–glycosaminoglycan (CG) matrices preloaded with adenoviral vectors containing various marker genes into lesions surgically generated in rabbit femoral condyles. Analysis of the recovered implants showed transgenic expression up to 21 days; however, a considerable portion was found in the synovial lining, indicating leakage of the vector and/or transduced cells from the matrix. As an alternative medium for gene delivery, we investigated the feasibility of using coagulated bone marrow aspirates. Mixture of an adenoviral suspension with the fluid phase of freshly aspirated bone marrow resulted in uniform dispersion of the vector throughout, and levels of transgenic expression in direct proportion to the density of nucleated cells in the ensuing clot. Furthermore, cultures of mesenchymal progenitor cells, previously transduced ex vivo with recombinant adenovirus, were readily incorporated into the coagulate when mixed with fresh aspirate. These vector-seeded and cell-seeded bone marrow clots were found to maintain their structural integrity following extensive culture and maintained transgenic expression in this manner for several weeks. When used in place of the CG matrix as a gene delivery vehicle in vivo, genetically modified bone marrow clots were able to generate similarly high levels of transgenic expression in osteochondral defects with better containment of the vector within the defect. Our results suggest that coagulates formed from aspirated bone marrow may be useful as a means of gene delivery to cartilage and perhaps other musculoskeletal tissues. Cells within the fluid can be readily modified with an adenoviral vector, and the matrix formed from the clot is completely natural, native to the host and is the fundamental platform on which healing and repair of mesenchymal tissues is based.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Buckwalter JA . Articular cartilage: injuries and potential for healing. J Orthop Sports Phys Ther 1998; 28: 192–202.
Rodrigo JJ, Steadman JR, Silliman JF, Fulstone HA . Improvement of full-thickness chondral defect healing in the human knee after debridement and microfracture using continuous passive motion. Am J Knee Surg 1994; 7: 109–116.
Minas T, Nehrer S . Current concepts in the treatment of articular cartilage defects. Orthopedics 1997; 20: 525–538.
Convery FR, Meyers MH, Akeson WH . Fresh osteochondral allografting of the femoral condyle. Clin Orthop 1991; 273: 139–145.
Amiel D et al. Rib perichondral grafts for the repair of full-thickness articular-cartilage defects. A morphological and biochemical study in rabbits. J Bone Joint Surg Am 1985; 67: 911–920.
Grande DA, Singh IJ, Pugh J . Healing of experimentally produced lesions in articular cartilage following chondrocyte transplantation. Anat Rec 1987; 218: 142–148.
Buckwalter JA, Mankin HJ . Articular cartilage repair and transplantation. Arthritis Rheum 1998; 41: 1331–1342.
Hubbard MJ . Articular debridement versus washout for degeneration of the medial femoral condyle. A five-year study. J Bone Joint Surg Br 1996; 78: 217–219; (see comments).
Kim HK, Moran ME, Salter RB . The potential for regeneration of articular cartilage in defects created by chondral shaving and subchondral abrasion. An experimental investigation in rabbits. J Bone Joint Surg Am 1991; 73: 1301–1315.
Newman AP . Articular cartilage repair. Am J Sports Med 1998; 26: 309–324.
Dandy DJ . Abrasion chondroplasty. Arthroscopy 1986; 2: 51–53.
Kaps C et al. Bone morphogenetic proteins promote cartilage differentiation and protect engineered artificial cartilage from fibroblast invasion and destruction. Arthritis Rheum 2002; 46: 149–162.
Majumdar MK, Wang E, Morris EA . BMP-2 and BMP-9 promotes chondrogenic differentiation of human multipotential mesenchymal cells and overcomes the inhibitory effect of IL-1. J Cell Physiol 2001; 189: 275–284.
Sellers RS, Peluso D, Morris EA . The effect of recombinant human bone morphogenetic protein-2 (rhBMP-2) on the healing of full-thickness defects of articular cartilage. J Bone Joint Surg Am 1997; 79: 1452–1463.
Mi Z et al. Adenovirus-mediated gene transfer of insulin-like growth factor 1 stimulates proteoglycan synthesis in rabbit joints. Arthritis Rheum 2000; 43: 2563–2570.
Fortier LA, Mohammed HO, Lust G, Nixon AJ . Insulin-like growth factor-I enhances cell-based repair of articular cartilage. J Bone Joint Surg Br 2002; 84: 276–288.
Denker AE, Nicoll SB, Tuan RS . Formation of cartilage-like spheroids by micromass cultures of murine C3H10T1/2 cells upon treatment with transforming growth factor-beta 1. Differentiation 1995; 59: 25–34.
Glansbeek HL et al. Stimulation of articular cartilage repair in established arthritis by local administration of transforming growth factor-beta into murine knee joints. Lab Invest 1998; 78: 133–142.
Izumi T, Scully SP, Heydemann A, Bolander ME . Transforming growth factor beta 1 stimulates type II collagen expression in cultured periosteum-derived cells. J Bone Miner Res 1992; 7: 115–121.
Trippel SB . Growth factor actions on articular cartilage. J Rheumatol Suppl 1995; 43: 129–132.
Nixon AJ, Fortier LA, Williams J, Mohammed H . Enhanced repair of extensive articular defects by insulin-like growth factor-I-laden fibrin composites. J Orthop Res 1999; 17: 475–487.
Evans CH, Ghivizzani SC, Robbins PD . Potential applications of gene therapy in sports medicine. Phys Med Rehabil Clin N Am 2000; 11: 405–416.
Kang R et al. Ex vivo gene transfer to chondrocytes in full-thickness articular cartilage defects: a feasibility study. Osteoarthritis Cartilage 1997; 5: 139–143.
Mason JM et al. Expression of human bone morphogenic protein 7 in primary rabbit periosteal cells: potential utility in gene therapy for osteochondral repair. Gene Therapy 1998; 5: 1098–1104.
Mason JM et al. Cartilage and bone regeneration using gene-enhanced tissue engineering. Clin Orthop 2000; 379: S171–S178; (in process citation).
Baragi VM et al. Transplantation of transduced chondrocytes protects articular cartilage from interleukin 1-induced extracellular matrix degradation. J Clin Invest 1995; 96: 2454–2460.
Madry H, Zurakowski D, Trippel SB . Overexpression of human insulin-like growth factor-I promotes new tissue formation in an ex vivo model of articular chondrocyte transplantation. Gene Therapy 2001; 8: 1443–1449.
Evans CH et al. Clinical trial to assess the safety, feasibility, and efficacy of transferring a potentially anti-arthritic cytokine gene to human joints with rheumatoid arthritis. Hum Gene Ther 1996; 7: 1261–1280.
Nehrer S et al. Canine chondrocytes seeded in type I and type II collagen implants investigated in vitro. J Biomed Mater Res 1997; 38: 95–104; (published erratum appears in J Biomed Mater Res 1997; 38 (4): 288).
Nehrer S et al. Matrix collagen type and pore size influence behaviour of seeded canine chondrocytes. Biomaterials 1997; 18: 769–776.
Bakker AC et al. Overexpression of active TGF-beta-1 in the murine knee joint: evidence for synovial-layer-dependent chondro-osteophyte formation. Osteoarthritis Cartilage 2001; 9: 128–136.
Mi Z et al. Adverse effects of adenovirus-mediated gene transfer of human transforming growth factor beta 1 into rabbit knees. Arthritis Res 2003; 5: 132–139.
Johnstone B, Yoo JU . Autologous mesenchymal progenitor cells in articular cartilage repair. Clin Orthop 1999; 367: S156–S162.
Hardy S et al. Construction of adenovirus vectors through Cre-lox recombination. J Virol 1997; 71: 1842–1849.
Nehrer S et al. Chondrocyte-seeded collagen matrices implanted in a chondral defect in a canine model. Biomaterials 1998; 19: 2313–2328.
Johnstone B et al. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res 1998; 238: 265–272.
Yoo JU et al. The chondrogenic potential of human bone-marrow-derived mesenchymal progenitor cells. J Bone Joint Surg Am 1998; 80: 1745–1757.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pascher, A., Palmer, G., Steinert, A. et al. Gene delivery to cartilage defects using coagulated bone marrow aspirate. Gene Ther 11, 133–141 (2004). https://doi.org/10.1038/sj.gt.3302155
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302155
Keywords
This article is cited by
-
Where is human-based cellular pharmaceutical R&D taking us in cartilage regeneration?
3 Biotech (2020)
-
Effects of combined rAAV-mediated TGF-β and sox9 gene transfer and overexpression on the metabolic and chondrogenic activities in human bone marrow aspirates
Journal of Experimental Orthopaedics (2017)
-
Basic science of osteoarthritis
Journal of Experimental Orthopaedics (2016)
-
Effects of rAAV-mediated FGF-2 gene transfer and overexpression upon the chondrogenic differentiation processes in human bone marrow aspirates
Journal of Experimental Orthopaedics (2016)
-
rAAV-mediated overexpression of sox9, TGF-β and IGF-I in minipig bone marrow aspirates to enhance the chondrogenic processes for cartilage repair
Gene Therapy (2016)