Abstract
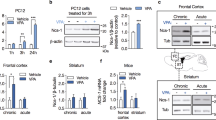
The extracellular signal-regulated kinase (ERK) pathway mediates neuronal plasticity in the CNS. The mood stabilizers lithium and valproate activate the ERK pathway in prefrontal cortex and hippocampus and potentiate ERK pathway-mediated neurite growth, neuronal survival and hippocampal neurogenesis. Here, we examined the role of the ERK pathway in behavioral plasticity related to facets of bipolar disorder. Mice with ERK1 ablation acquired reduced phosphorylation of RSK1, an ERK substrate, in prefrontal cortex and striatum, but not in hippocampus or cerebellum, indicating the ablation-induced brain region-specific ERK signaling deficits. ERK1 ablation produced a behavioral excitement profile similar to that induced by psychostimulants. The profile is characterized by hyperactivity, enhanced goal-directed activity and increased pleasure-related activity with potential harmful consequence. ERK1-ablated mice were hyperactive in multiple tests and resistant to behavioral despair in the forced swim test. These mice displayed more home-cage voluntary wheel running activities, rearings in a large arena and open-arm visits in an elevated plus maze. Treatments with valproate and olanzapine, but not lithium reduced baseline activities in ERK1-ablated mice. All three treatments attenuated amphetamine-induced hyperactivity in ablated mice. These data indicate a profound involvement of ERK1 signaling in behavioral excitement and in the behavioral action of antimanic agents. The extent to which ERK pathway perturbation contributes to the susceptibility, mood switch mechanism(s) and symptom pathophysiology of bipolar disorder requires further investigation. Whether there is a shared mechanism through which mood stabilizers produce their clinical actions on mood, thought and behavioral symptoms of mania also requires further investigation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Kelleher III RJ, Govindarajan A, Tonegawa S . Translational regulatory mechanisms in persistent forms of synaptic plasticity. Neuron 2004; 44: 59–73.
Thomas GM, Huganir RL . MAPK cascade signalling and synaptic plasticity. Nat Rev Neurosci 2004; 5: 173–183.
Sweatt JD . Mitogen-activated protein kinases in synaptic plasticity and memory. Curr Opin Neurobiol 2004; 14: 311–317.
Chen G, Manji HK . The extracellular signal-regulated kinase pathway: an emerging promising target for mood stabilizers. Curr Opin Psychiatry 2006; 19: 313–323.
Huang EJ, Reichardt LF . Trk receptors: roles in neuronal signal transduction. Annu Rev Biochem 2003; 72: 609–642.
Coyle JT, Duman RS . Finding the intracellular signaling pathways affected by mood disorder treatments. Neuron 2003; 38: 157–160.
Berton O, Nestler EJ . New approaches to antidepressant drug discovery: beyond monoamines. Nat Rev Neurosci 2006; 7: 137–151.
Manji HK, Drevets WC, Charney DS . The cellular neurobiology of depression. Nat Med 2001; 7: 541–547.
Selcher JC, Nekrasova T, Paylor R, Landreth GE, Sweatt JD . Mice lacking the ERK1 isoform of MAP kinase are unimpaired in emotional learning. Learn Mem 2001; 8: 11–19.
Mazzucchelli C, Vantaggiato C, Ciamei A, Fasano S, Pakhotin P, Krezel W et al. Knockout of ERK1 MAP kinase enhances synaptic plasticity in the striatum and facilitates striatal-mediated learning and memory. Neuron 2002; 34: 807–820.
Cestari V, Costanzi M, Castellano C, Rossi-Arnaud C . A role for ERK2 in reconsolidation of fear memories in mice. Neurobiol Learn Mem 2006; 86: 133–143.
Goodwin FK, Jamison KR . Manic-Depressive Illness. Oxford University Press: New York, 1990.
Belmaker RH . Bipolar disorder. N Engl J Med 2004; 351: 476–486.
Phiel CJ, Klein PS . Molecular targets of lithium action. Annu Rev Pharmacol Toxicol 2001; 41: 789–813.
Chen G, Creson T, Sharon E, Hao Y, Wang G . Neurotrophic actions of mood-stabilizers: a recent research discovery and its potential clinical applications. Curr Psychiatry Rev 2005; 1: 173–185.
Chuang DM . Neuroprotective and neurotrophic actions of the mood stabilizer lithium: can it be used to treat neurodegenerative diseases? Crit Rev Neurobiol 2004; 16: 83–90.
Chuang DM . The antiapoptotic actions of mood stabilizers: molecular mechanisms and therapeutic potentials. Ann NY Acad Sci 2005; 1053: 195–204.
Yuan PX, Huang LD, Jiang YM, Gutkind JS, Manji HK, Chen G . The mood stabilizer valproic acid activates mitogen-activated protein kinases and promotes neurite growth. J Biol Chem 2001; 276: 31674–31683.
Hao Y, Creson T, Zhang L, Li P, Du F, Yuan P et al. Mood stabilizer valproate promotes ERK pathway-dependent cortical neuronal growth and neurogenesis. J Neurosci 2004; 24: 6590–6599.
Chen G, Rajkowska G, Du F, Seraji-Bozorgzad N, Manji HK . Enhancement of hippocampal neurogenesis by lithium. J neurochem 2000; 75: 1729–1734.
Einat H, Yuan P, Gould TD, Li J, Du J, Zhang L et al. The role of the extracellular signal-regulated kinase signaling pathway in mood modulation. J Neurosci 2003; 23: 7311–7316.
Crawley JN . What's Wrong With My Mouse. Wiley-Liss: New York, 2000.
Cryan JF, Markou A, Lucki I . Assessing antidepressant activity in rodents: recent developments and future needs. Trends Pharmacol Sci 2002; 23: 238–245.
Decker S, Grider G, Cobb M, Li XP, Huff MO, El-Mallakh RS et al. Open field is more sensitive than automated activity monitor in documenting ouabain-induced hyperlocomotion in the development of an animal model for bipolar illness. Prog Neuropsychopharmacol Biol Psychiatry 2000; 24: 455–462.
Porsolt RD, Le Pichon M, Jalfre M . Depression: a new animal model sensitive to antidepressant treatments. Nature 1977; 266: 730–732.
Cox C, Harrison-Read PE, Steinberg H, Tomkiewicz M . Lithium attenuates drug-induced hyperactivity in rats. Nature 1971; 232: 336–338.
Wieland S, Lucki I . Antidepressant-like activity of 5-HT1A agonists measured with the forced swim test. Psychopharmacology 1990; 101: 497–504.
Werme M, Messer C, Olson L, Gilden L, Thoren P, Nestler EJ et al. Delta FosB regulates wheel running. J Neurosci 2002; 22: 8133–8138.
Lett BT, Grant VL, Koh MT, Flynn G . Prior experience with wheel running produces cross-tolerance to the rewarding effect of morphine. Pharmacol Biochem Behav 2002; 72: 101–105.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders DSM-IV-TR. American Psychiatric Association: Washington DC, 2000.
Reeves G, Schweitzer J . Pharmacological management of attention-deficit hyperactivity disorder. Expert Opin Pharmacother 2004; 5: 1313–1320.
Anand A, Verhoeff P, Seneca N, Zoghbi SS, Seibyl JP, Charney DS et al. Brain SPECT imaging of amphetamine-induced dopamine release in euthymic bipolar disorder patients. Am J Psychiatry 2000; 157: 1108–1114.
Peet M, Peters S . Drug-induced mania. Drug Saf 1995; 12: 146–153.
Gainetdinov RR, Caron MG . An animal model of attention deficit hyperactivity disorder. Mol Med Today 2000; 6: 43–44.
Gould TD, O’Donnell K C, Picchini AM, Manji HK . Strain differences in lithium attenuation of D-amphetamine-induced hyperlocomotion: a mouse model for the genetics of clinical response to lithium. Neuropsychopharmacology 2007; 32: 1321–1333.
Tohen M, Jacobs TG, Feldman PD . Onset of action of antipsychotics in the treatment of mania. Bipolar Disord 2000; 2 (Part 2): 261–268.
Gainetdinov RR, Wetsel WC, Jones SR, Levin ED, Jaber M, Caron MG . Role of serotonin in the paradoxical calming effect of psychostimulants on hyperactivity. Science 1999; 283: 397–401.
Einat H . Establishment of a battery of simple models for facets of bipolar disorder: a practical approach to achieve increased validity, better screening and possible insights into endophenotypes of disease. Behav Genet 2007; 37: 244–255.
Einat H, Manji HK, Belmaker RH . New approaches to modeling bipolar disorder. Psychopharmacol Bull 2003; 37: 47–63.
Boyce-Rustay JM, Holmes A . Genetic inactivation of the NMDA receptor NR2A subunit has anxiolytic- and antidepressant-like effects in mice. Neuropsychopharmacology 2006; 31: 2405–2414.
Sakamoto T, Mishina M, Niki H . Mutation of NMDA receptor subunit epsilon 1: effects on audiogenic-like seizures induced by electrical stimulation of the inferior colliculus in mice. Brain Res Mol Brain Res 2002; 102: 113–117.
Filliol D, Ghozland S, Chluba J, Martin M, Matthes HW, Simonin F et al. Mice deficient for delta- and mu-opioid receptors exhibit opposing alterations of emotional responses. Nat Genet 2000; 25: 195–200.
Mombereau C, Kaupmann K, Froestl W, Sansig G, van der Putten H, Cryan JF . Genetic and pharmacological evidence of a role for GABA(B) receptors in the modulation of anxiety- and antidepressant-like behavior. Neuropsychopharmacology 2004; 29: 1050–1062.
Mombereau C, Kaupmann K, Gassmann M, Bettler B, van der Putten H, Cryan JF . Altered anxiety and depression-related behaviour in mice lacking GABAB(2) receptor subunits. Neuroreport 2005; 16: 307–310.
Ferguson SM, Fasano S, Yang P, Brambilla R, Robinson TE . Knockout of ERK1 enhances cocaine-evoked immediate early gene expression and behavioral plasticity. Neuropsychopharmacology 2006; 31: 2660–2668.
Lucki I, O’Leary OF . Distinguishing roles for norepinephrine and serotonin in the behavioral effects of antidepressant drugs. J Clin Psychiatry 2004; 65 (Suppl 4): 11–24.
Cryan JF, Holmes A . The ascent of mouse: advances in modelling human depression and anxiety. Nat Rev Drug Discov 2005; 4: 775–790.
Geyer MA, McIlwain KL, Paylor R . Mouse genetic models for prepulse inhibition: an early review. Mol psychiatry 2002; 7: 1039–1053.
Weeber EJ, Sweatt JD . Molecular neurobiology of human cognition. Neuron 2002; 33: 845–848.
Avale ME, Falzone TL, Gelman DM, Low MJ, Grandy DK, Rubinstein M . The dopamine D4 receptor is essential for hyperactivity and impaired behavioral inhibition in a mouse model of attention deficit/hyperactivity disorder. Mol psychiatry 2004; 9: 718–726.
Avale ME, Nemirovsky SI, Raisman-Vozari R, Rubinstein M . Elevated serotonin is involved in hyperactivity but not in the paradoxical effect of amphetamine in mice neonatally lesioned with 6-hydroxydopamine. J Neurosci Res 2004; 78: 289–296.
Abulaban FS, Dhariwal MA, al-Bekairi AM, Raza M . Antinociceptive activity of sodium valproate in mice after chronic treatment. Gen pharmacol 1997; 29: 463–467.
Li S, Murakami Y, Wang M, Maeda K, Matsumoto K . The effects of chronic valproate and diazepam in a mouse model of post-traumatic stress disorder. Pharmacol Biochem Behav 2006; 85: 324–331.
Gleason SD, Shannon HE . Blockade of phencyclidine-induced hyperlocomotion by olanzapine, clozapine and serotonin receptor subtype selective antagonists in mice. Psychopharmacology 1997; 129: 79–84.
Duncan GE, Moy SS, Lieberman JA, Koller BH . Typical and atypical antipsychotic drug effects on locomotor hyperactivity and deficits in sensorimotor gating in a genetic model of NMDA receptor hypofunction. Pharmacol Biochem Behav 2006; 85: 481–491.
Gurvich N, Klein PS . Lithium and valproic acid: parallels and contrasts in diverse signaling contexts. Pharmacol Ther 2002; 96: 45–66.
De Sarno P, Li X, Jope RS . Regulation of Akt and glycogen synthase kinase-3 beta phosphorylation by sodium valproate and lithium. Neuropharmacology 2002; 43: 1158–1164.
Berridge MJ . The Albert Lasker medical awards. Inositol trisphosphate, calcium, lithium, and cell signaling. JAMA 1989; 262: 1834–1841.
Klein PS, Melton DA . A molecular mechanism for the effect of lithium on development. Proc Natl Acad Sci USA 1996; 93: 8455–8459.
Phiel CJ, Zhang F, Huang EY, Guenther MG, Lazar MA, Klein PS . Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. J Biol Chem 2001; 276: 36734–36741.
Gould TD, Einat H, Bhat R, Manji HK . AR-A014418, a selective GSK-3 inhibitor, produces antidepressant-like effects in the forced swim test. Int J Neuropsychopharmacol 2004; 7: 387–390.
Duman CH, Schlesinger L, Kodama M, Russell DS, Duman RS . A role for MAP kinase signaling in behavioral models of depression and antidepressant treatment. Biol psychiatry 2007; 61: 661–670.
Saarelainen T, Hendolin P, Lucas G, Koponen E, Sairanen M, MacDonald E et al. Activation of the TrkB neurotrophin receptor is induced by antidepressant drugs and is required for antidepressant-induced behavioral effects. J Neurosci 2003; 23: 349–357.
Zorner B, Wolfer DP, Brandis D, Kretz O, Zacher C, Madani R et al. Forebrain-specific trkB-receptor knockout mice: behaviorally more hyperactive than ‘depressive’. Biol psychiatry 2003; 54: 972–982.
Nibuya M, Morinobu S, Duman RS . Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci 1995; 15: 7539–7547.
Shirayama Y, Chen AC, Nakagawa S, Russell DS, Duman RS . Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci 2002; 22: 3251–3261.
Monteggia LM, Barrot M, Powell CM, Berton O, Galanis V, Gemelli T et al. Essential role of brain-derived neurotrophic factor in adult hippocampal function. Proc Natl Acad Sci USA 2004; 101: 10827–10832.
Berton O, McClung CA, Dileone RJ, Krishnan V, Renthal W, Russo SJ et al. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science 2006; 311: 864–868.
Tsankova NM, Berton O, Renthal W, Kumar A, Neve RL, Nestler EJ . Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nat Neurosci 2006; 9: 519–525.
Eisch AJ, Bolanos CA, de Wit J, Simonak RD, Pudiak CM, Barrot M et al. Brain-derived neurotrophic factor in the ventral midbrain-nucleus accumbens pathway: a role in depression. Biol psychiatry 2003; 54: 994–1005.
Nibuya M, Nestler EJ, Duman RS . Chronic antidepressant administration increases the expression of cAMP response element binding protein (CREB) in rat hippocampus. J Neurosci 1996; 16: 2365–2372.
Newton SS, Thome J, Wallace TL, Shirayama Y, Schlesinger L, Sakai N et al. Inhibition of cAMP response element-binding protein or dynorphin in the nucleus accumbens produces an antidepressant-like effect. J Neurosci 2002; 22: 10883–10890.
Conti AC, Cryan JF, Dalvi A, Lucki I, Blendy JA . cAMP response element-binding protein is essential for the upregulation of brain-derived neurotrophic factor transcription, but not the behavioral or endocrine responses to antidepressant drugs. J Neurosci 2002; 22: 3262–3268.
Conti AC, Kuo YC, Valentino RJ, Blendy JA . Inducible cAMP early repressor regulates corticosterone suppression after tricyclic antidepressant treatment. J Neurosci 2004; 24: 1967–1975.
Acknowledgements
This project was supported by funding from NIMH-IRP (Manji and Chen) and NARSAD (Chen). The views expressed in this article do not necessarily represent those of NIMH or NIH or the Federal Government.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website (http://www.nature.com/mp)
Rights and permissions
About this article
Cite this article
Engel, S., Creson, T., Hao, Y. et al. The extracellular signal-regulated kinase pathway contributes to the control of behavioral excitement. Mol Psychiatry 14, 448–461 (2009). https://doi.org/10.1038/sj.mp.4002135
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4002135
Keywords
This article is cited by
-
Dissecting 16p11.2 hemi-deletion to study sex-specific striatal phenotypes of neurodevelopmental disorders
Molecular Psychiatry (2024)
-
adgrl3.1-deficient zebrafish show noradrenaline-mediated externalizing behaviors, and altered expression of externalizing disorder-candidate genes, suggesting functional targets for treatment
Translational Psychiatry (2023)
-
Enlightened: addressing circadian and seasonal changes in photoperiod in animal models of bipolar disorder
Translational Psychiatry (2021)
-
Therapeutic approaches employing natural compounds and derivatives for treating bipolar disorder: emphasis on experimental models of the manic phase
Metabolic Brain Disease (2021)
-
Controlled trial of lovastatin combined with an open-label treatment of a parent-implemented language intervention in youth with fragile X syndrome
Journal of Neurodevelopmental Disorders (2020)