Abstract
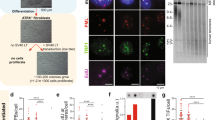
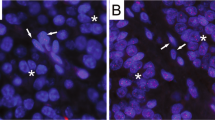
We have used cultured human mammary epithelial cells (HMEC) and breast tumor-derived lines to gain information on defects that occur during breast cancer progression. HMEC immortalized by a variety of agents (the chemical carcinogen benzo(a)pyrene, oncogenes c-myc and ZNF217, and/or dominant negative p53 genetic suppressor element GSE22) displayed marked upregulation (10–15 fold) of the telomere-binding protein, TRF2. Upregulation of TRF2 protein was apparently due to differences in post-transcriptional regulation, as mRNA levels remained comparable in finite lifespan and immortal HMEC. TRF2 protein was not upregulated by the oncogenic agents alone in the absence of immortalization, nor by expression of exogenously introduced hTERT genes. We found TRF2 levels to be at least twofold higher than in control cells in 11/15 breast tumor cell lines, suggesting that elevated TRF2 levels are a frequent occurrence during the transformation of breast tumor cells in vivo. The dispersed distribution of TRF2 throughout the nuclei in some immortalized and tumor-derived cells indicated that not all the TRF2 was associated with telomeres in these cells. The process responsible for accumulation of TRF2 in immortalized HMEC and breast tumor-derived cell lines may promote tumorigenesis by contributing to the cells’ ability to maintain an indefinite lifespan.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bilaud T, Brun C, Ancelin K, Koering CE, Laroche T and Gilson E . (1997). Nat. Genet., 17, 236–239.
Blackburn EH . (2000). Nature, 408, 53–56.
Brenner AJ, Stampfer MR and Aldaz CM . (1998). Oncogene, 17, 199–205.
Broccoli D, Smogorzewska A, Chong L and de Lange T . (1997). Nat. Genet., 17, 231–235.
Feldser DM, Hackett JA and Greider CW . (2003). Nat. Rev. Cancer, 3, 623–627.
Griffith JD, Comeau L, Rosenfield S, Stansel RM, Bianchi A, Moss H and de Lange T . (1999). Cell, 97, 503–514.
Karlseder J, Broccoli D, Dai Y, Hardy S and de Lange T . (1999). Science, 283, 1321–1325.
Karlseder J, Hoke K, Mirzoeva OK, Bakkenist C, Kastan MB, Petrini JH and de Lange T . (2004). PLoS Biol., 2, E240.
Karlseder J, Smogorzewska A and de Lange T . (2002). Science, 295, 2446–2449.
Kim SH, Kaminker P and Campisi J . (2002). Oncogene, 21, 503–511.
Klapper W, Qian W, Schulte C and Parwaresch R . (2003). Leukemia, 17, 2007–2015.
Maser RS and DePinho RA . (2002). Science, 297, 565–569.
Morgenstern JP and Land H . (1990). Nucleic Acids Res., 18, 3587–3596.
Nakagami Y, Ito M, Hara T and Inoue T . (2002). Radiat. Med., 20, 121–129.
Nijjar T, Wigington D, Garbe JC, Waha A, Stampfer MR and Yaswen P . (1999). Cancer Res., 59, 5112–5118.
Nonet G, Stampfer MR, Chin K, Gray JW, Collins CC and Yaswen P . (2001). Cancer Res., 61, 1250–1254.
Opresko PL, von Kobbe C, Laine JP, Harrigan J, Hickson ID and Bohr VA . (2002). J. Biol. Chem., 277, 41110–41119.
Ossovskaya VS, Mazo IA, Chernov MV, Chernova OB, Strezoska Z, Kondratov R, Stark GR, Chumakov PM and Gudkov AV . (1996). Proc. Natl. Acad. Sci. USA, 93, 10309–10314.
Romanov S, Kozakiewicz K, Holst C, Stampfer MR, Haupt LM and Tlsty T . (2001). Nature, 409, 633–637.
Smogorzewska A, Karlseder J, Holtgreve-Grez H, Jauch A and de Lange T . (2002). Curr. Biol., 12, 1635–1644.
Stampfer M, Garbe J, Levine G, Lichsteiner S, Vasserot A and Yaswen P . (2001). Proc. Natl. Acad. Sci, USA, 98, 4498–4503.
Stampfer MR and Bartley JC . (1985). Proc. Natl. Acad. Sci. USA, 82, 2394–2398.
Stampfer MR, Bodnar A, Garbe J, Wong M, Pan A, Villeponteau B and Yaswen P . (1997). Mol. Biol. Cell, 8, 2391–2405.
Stampfer MR, Garbe J, Nijjar T, Wigington D, Swisshelm K and Yaswen P . (2003). Oncogene, 22, 5238–5251.
Stampfer MR, Pan CH, Hosoda J, Bartholomew J, Mendelsohn J and Yaswen P . (1993). Exp. Cell Res., 208, 175–188.
Stampfer MR and Yaswen P . (2003). Cancer Lett., 194, 199–208.
Walen K and Stampfer MR . (1989). Cancer Genet. Cytogenet., 37, 249–261.
Yaswen P and Stampfer MR . (2002). Int. J. Biochem. Cell Biol., 34, 1382–1394.
Zhu XD, Kuster B, Mann M, Petrini JH and de Lange T . (2000). Nat. Genet., 25, 347–352.
Acknowledgements
We thank T de Lange for the TRF2 cDNA expression vector, and J Campisi for Tin2 antibodies as well as helpful discussions. This work was supported by grants from the National Institutes of Health (CA-24844), the California Breast Cancer Research Program (6JB-0133 and 8KB-0119), and the Department of Defense (DAMD17-02-1-0443), as well as by the Office of Energy Research, Office of Health and Biological Research, US Department of Energy under Contract No. DE-AC03-76SF00098. EB was supported by an NRSA fellowship (CA-108480) from the NIH. DG received additional support from the Indiana University Cancer Center, the American Cancer Society, an Indiana University School of Medicine Biomedical Research grant, and the Indiana Genomics Initiative (INGEN). INGEN of Indiana University is supported in part by Lilly Endowment, Inc.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Nijjar, T., Bassett, E., Garbe, J. et al. Accumulation and altered localization of telomere-associated protein TRF2 in immortally transformed and tumor-derived human breast cells. Oncogene 24, 3369–3376 (2005). https://doi.org/10.1038/sj.onc.1208482
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208482
Keywords
This article is cited by
-
Inhibition of TRF2 accelerates telomere attrition and DNA damage in naïve CD4 T cells during HCV infection
Cell Death & Disease (2018)
-
Elevated levels of TRF2 induce telomeric ultrafine anaphase bridges and rapid telomere deletions
Nature Communications (2015)
-
Chalcone-imidazolone conjugates induce apoptosis through DNA damage pathway by affecting telomeres
Cancer Cell International (2011)
-
Elevated TRF2 in advanced breast cancers with short telomeres
Breast Cancer Research and Treatment (2011)
-
The 3D nuclear organization of telomeres marks the transition from Hodgkin to Reed–Sternberg cells
Leukemia (2009)