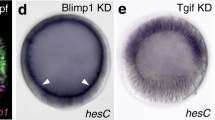
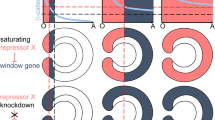
Abstract
Cnidarians are the simplest metazoans with a nervous system. They are well known for their regeneration capacity, which is based on the restoration of a signalling centre (organizer). Recent work has identified the canonical Wnt pathway in the freshwater polyp Hydra, where it acts in organizer formation and regeneration. Wnt signalling is also essential for cnidarian embryogenesis. In the sea anemone Nematostella vectensis 11 of the 12 known wnt gene subfamilies were identified. Different wnt genes exhibit serial and overlapping expression domains along the oral–aboral axis of the embryo (the ‘wnt code’). This is reminiscent of the hox code (cluster) in bilaterian embryogenesis that is, however, absent in cnidarians. It is proposed that the common ancestor of cnidarians and bilaterians invented a set of wnt genes that patterned the ancient main body axis. Major antagonists of Wnt ligands (e.g. Dkk 1/2/4) that were previously known only from chordates, are also present in cnidarians and exhibit a similar conserved function. The unexpectedly high level of genetic complexity of wnt genes evolved in early multi-cellular animals about 650 Myr ago and suggests a radical expansion of the genetic repertoire, concurrent with the evolution of multi-cellularity and the diversification of eumetazoan body plans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aguilera O, Fraga MF, Ballestar E, Paz MF, Herranz M, Espada J et al. (2006). Epigenetic inactivation of the Wnt antagonist Dickkopf-1 (Dkk-1) gene in human colorectal cancer. Oncogene 25: 4116–4121.
Augustin R, Franke A, Khalturin K, Kiko R, Siebert S, Hemmrich G et al. (2006). Dickkopf related genes are components of the positional value gradient in hydra. Dev Biol 296: 62–70.
Banziger C, Soldini D, Schutt C, Zipperlen P, Hausmann G, Basler K . (2006). Wntless, a conserved membrane protein dedicated to the secretion of Wnt proteins from signaling cells. Cell 125: 509–522.
Bartscherer K, Pelte N, Ingelfinger D, Boutros M . (2006). Secretion of Wnt ligands requires Evi, a conserved transmembrane protein. Cell 125: 523–533.
Brooke NM, Garcia-Fernandez J, Holland PW . (1998). The Parahox gene cluster is an evolutionary sister of the Hox gene cluster. Nature 392: 920–922.
Brooke NM, Holland PW . (2003). The evolution of multicellularity and early animal genomes. Curr Opin Genet Dev 13: 599–603.
Broun M, Bode HR . (2002). Characterization of the head organizer in hydra. Development 129: 875–884.
Broun M, Gee L, Reinhardt B, Bode HR . (2005). Formation of the head organizer in hydra involves the canonical Wnt pathway. Development 132: 2907–2916.
Browne EN . (1909). The production of new hydranths in hydra by the insertion of small grafts. J Exp Zool 7: 1–37.
Chen JY, Oliveri P, Gao F, Dornbos SQ, Li CW, Bottjer DJ et al. (2002). Precambrian animal life: probable developmental and adult cnidarian forms from Southwest China. Dev Biol 248: 182–196.
Chen JY, Oliveri P, Li CW, Zhou GQ, Gao F, Hagadorn JW et al. (2000). Precambrian animal diversity: putative phosphatized embryos from the Doushantuo formation of China. Proc Natl Acad Sci USA 97: 4457–4462.
Chourrout D, Delsuc F, Chourrout P, Edvardsen RB, Rentzsch F, Renfer E et al. (2006). Minimal Protohox cluster inferred from comparing bilaterian and cnidarian Hox complements. Nature 442: 684–687.
Conway Morris S . (2000). The Cambrian ‘explosion’: slow-fuse or megatonnage? Proc Natl Acad Sci USA 97: 4426–4429.
Cramer von Laue C . (2003). Untersuchungen zur dualen Funktion von beta-Catenin im Wnt-Signalweg und der Cadherin-vermittelten Zelladhäsion bei Hydra; PhD. Department of Biology, Darmstadt University of Technololgy. Darmstadt, pp 97.
Darling JA, Reitzel AR, Burton PM, Mazza ME, Ryan JF, Sullivan JC et al. (2005). Rising starlet: the starlet sea anemone, Nematostella vectensis. Bioessays 27: 211–221.
Davidson G, Mao B, del Barco Barrantes I, Niehrs C . (2002). Kremen proteins interact with Dickkopf1 to regulate anteroposterior Cns patterning. Development 129: 5587–5596.
Extavour CG, Pang K, Matus DQ, Martindale MQ . (2005). Vasa and Nanos expression patterns in a sea anemone and the evolution of bilaterian germ cell specification mechanisms. Evol Dev 7: 201–215.
Fedders H, Augustin R, Bosch TC . (2004). A Dickkopf-3-related gene is expressed in differentiating nematocytes in the basal metazoan hydra. Dev Genes Evol 214: 72–80.
Ferrier DE, Holland PW . (2001). Ancient origin of the Hox Gene cluster. Nat Rev Genet 2: 33–38.
Finnerty JR, Pang K, Burton P, Paulson D, Martindale MQ . (2004). Origins of bilateral symmetry: Hox and Dpp expression in a sea anemone. Science 304: 1335–1337.
Fritzenwanker JH, Saina M, Technau U . (2004). Analysis of forkhead and snail expression reveals epithelial-mesenchymal transitions during embryonic and larval development of Nematostella vectensis. Dev Biol 275: 389–402.
Frobius AC, Genikhovich G, Kurn U, Anton-Erxleben F, Bosch TC . (2003). Expression of developmental genes during early embryogenesis of hydra. Dev Genes Evol 213: 445–455.
Garcia-Fernandez J . (2005a). The genesis and evolution of homeobox gene clusters. Nat Rev Genet 6: 881–892.
Garcia-Fernandez J . (2005b). Hox, parahox, protohox: facts and guesses. Heredity 94: 145–152.
Gonzalez-Sancho JM, Aguilera O, Garcia JM, Pendas-Franco N, Pena C, Cal S et al. (2005). The Wnt antagonist Dickkopf-1 gene is a downstream target of beta-catenin/Tcf and is downregulated in human colon cancer. Oncogene 24: 1098–1103.
Guder C, Pinho S, Nacak TG, Schmidt HA, Hobmayer B, Niehrs C et al. (2006). An ancient Wnt-Dickkopf antagonism in hydra. Development 133: 901–911.
Hand C, Uhlinger KR . (1992). The culture, sexual, and asexual reproduction and growth of the sea anemone Nematostella vectensis. Biol Bull 182: 169–176.
Hassel M, Albert K, Hofheinz S . (1993). Pattern formation in Hydra vulgaris is controlled by lithium-sensitive processes. Dev Biol 156: 362–371.
Hayward DC, Miller DJ, Ball EE . (2004). Snail expression during embryonic development of the coral acropora: blurring the diploblast/triploblast divide? Dev Genes Evol 214: 257–260.
He X, Axelrod JD . (2006). A Wnter wonderland in snowbird. Development 133: 2597–2603.
Hobmayer B, Rentzsch F, Kuhn K, Happel CM, von Laue CC, Snyder P et al. (2000). Wnt signalling molecules act in axis formation in the diploblastic metazoan hydra. Nature 407: 186–189.
Hobmayer E, Hatta M, Fischer R, Fujisawa T, Holstein TW, Sugiyama T . (1996). Identification of a hydra homologue of the Beta-Catenin/Plakoglobin/Armadillo gene family. Gene 172: 155–159.
Holland LZ . (2002). Heads or tails? Amphioxus and the evolution of anterior-posterior patterning in deuterostomes. Dev Biol 241: 209–228.
Holland LZ, Rached LA, Tamme R, Holland ND, Kortschak D, Inoko H et al. (2001). Characterization and developmental expression of the amphioxus homolog of notch (amphinotch): evolutionary conservation of multiple expression domains in amphioxus and vertebrates. Dev Biol 232: 493–507.
Holland P . (2004). Developmental biology. The ups and downs of a sea anemone. Science 304: 1255–1256.
Holstein TW, Hobmayer E, Technau U . (2003). Cnidarians: an evolutionarily conserved model system for regeneration? Dev Dyn 226: 257–267.
Jessen JR, Solnica-Krezel L . (2005). Axis formation – beta-catenin catches a Wnt. Cell 120: 736–737.
Jones SE, Jomary C . (2002). Secreted frizzled-related proteins: searching for relationships and patterns. Bioessays 24: 811–820.
Kamm K, Schierwater B, Jakob W, Dellaporta SL, Miller DJ . (2006). Axial patterning and diversification in the cnidaria predate the Hox system. Curr Biol 16: 920–926.
Kawano Y, Kypta R . (2003). Secreted antagonists of the Wnt signalling pathway. J Cell Sci 116: 2627–2634.
King N, Hittinger CT, Carroll SB . (2003). Evolution of key cell signaling and adhesion protein families predates animal origins. Science 301: 361–363.
Kirschner MW, Gerhart JC . (2005). The Plausibility of Life. Yale University Press: New Haven, London.
Klein RD, Gu Q, Goddard A, Rosenthal A . (1996). Selection for genes encoding secreted proteins and receptors. Proc Natl Acad Sci USA 93: 7108–7113.
Kusserow A, Pang K, Sturm C, Hrouda M, Lentfer J, Schmidt HA et al. (2005). Unexpected complexity of the Wnt gene family in a sea anemone. Nature 433: 156–160.
Lee PN, Pang K, Matus DQ, Martindale MQ . (2006). A Wnt of things to come: evolution of Wnt signaling and polarity in cnidarians. Semin Cell Dev Biol 17: 157–167.
Lie DC, Colamarino SA, Song HJ, Desire L, Mira H, Consiglio A et al. (2005). Wnt signalling regulates adult Hippocampal neurogenesis. Nature 437: 1370–1375.
Logan CY, Nusse R . (2004). The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20: 781–810.
Magie CR, Pang K, Martindale MQ . (2005). Genomic inventory and expression of Sox and Fox genes in the cnidarian Nematostella vectensis. Dev Genes Evol 215: 618–630.
Mao B, Niehrs C . (2003). Kremen2 modulates Dickkopf2 activity during Wnt/Lrp6 signaling. Gene 302: 179–183.
Martindale MQ, Pang K, Finnerty JR . (2004). Investigating the origins of triploblasty: ‘mesodermal’ gene expression ina diploblastic animal, the sea anemone Nematostella vectensis (phylum, cnidaria; class, anthozoa). Development 131: 2463–2474.
Matus DQ, Thomsen GH, Martindale MQ . (2006). Dorso/ventral genes are asymmetrically expressed and involved in germ-layer demarcation during cnidarian gastrulation. Curr Biol 16: 499–505.
Miller DJ, Ball EE, Technau U . (2005). Cnidarians and Ancestral genetic complexity in the animal kingdom. Trends Genet 21: 536–539.
Miller JR . (2002). The Wnts. Genome Biol 3: reviews3001.1–3001.15.
Minobe S, Fei K, Yan L, Sarras Jr M, Werle M . (2000). Identification and characterization of the epithelial polarity receptor ‘frizzled’ in Hydra vulgaris. Dev Genes Evol 210: 258–262.
Muller WA, Teo R, Frank U . (2004a). Totipotent migratory stem cells in a hydroid. Dev Biol 275: 215–224.
Muller WA, Teo R, Mohrlen F . (2004b). Patterning a multi-headed mutant in hydractinia: enhancement of head formation and its phenotypic normalization. Int J Dev Biol 48: 9–15.
Onai T, Sasai N, Matsui M, Sasai Y . (2004). Xenopus Xsalf: anterior neuroectodermal specification by attenuating cellular responsiveness to Wnt signaling. Dev Cell 7: 95–106.
Plickert G, Jacoby V, Frank U, Muller W, Mokady O . (2006). Wnt signaling in hydroid development: formation of the primary body axis in embryogenesis and its subsequent Patterning. Dev Biol 298: 368–378.
Price MA . (2006). Cki, there's more than one: casein kinase I family members in Wnt and hedgehog signaling. Genes Dev 20: 399–410.
Prud'homme B, Lartillot N, Balavoine G, Adoutte A, Vervoort M . (2002). Phylogenetic analysis of the Wnt gene family. Insights from lophotrochozoan members. Curr Biol 12: 1395–1400.
Rentzsch F, Anton R, Saina M, Hammerschmidt M, Holstein TW, Technau U . (2006). Asymmetric expression of the bmp antagonists chordin and gremlin in the sea anemone Nematostella vectensis: implications for the evolution of axial patterning. Dev Biol.
Rentzsch F, Hobmayer B, Holstein TW . (2005). Glycogen synthase kinase 3 has a proapoptotic function in hydra gametogenesis. Dev Biol 278: 1–12.
Scholz CB, Technau U . (2003). The ancestral role of brachyury: expression of nembra1 in the basal cnidarian Nematostella vectensis (anthozoa). Dev Genes Evol 212: 563–570.
Spring J, Yanze N, Josch C, Middel AM, Winninger B, Schmid V . (2002). Conservation of brachyury, mef2, and snail in the myogenic lineage of jellyfish: a connection to the mesoderm of bilateria. Dev Biol 244: 372–384.
Spring J, Yanze N, Middel AM, Stierwald M, Groger H, Schmid V . (2000). The mesoderm specification factor twist in the life cycle of jellyfish. Dev Biol 228: 363–375.
Steele RE . (2006). Trembley's polyps go transgenic. Proc Natl Acad Sci USA 103: 6415–6416.
Strutt H, Price MA, Strutt D . (2006). Planar polarity is positively regulated by casein kinase iepsilon in Drosophila. Curr Biol 16: 1329–1336.
Tao Q, Yokota C, Puck H, Kofron M, Birsoy B, Yan D et al. (2005). Maternal Wnt11 activates the canonical Wnt signaling pathway required for axis formation in Xenopus embryos. Cell 120: 857–871.
Technau U, Bode HR . (1999). Hybra1, a brachyury homologue, acts during head formation in hydra. Development 126: 999–1010.
Technau U, Cramer von Laue C, Rentzsch F, Luft S, Hobmayer B, Bode HR et al. (2000). Parameters of self-organization in hydra aggregates. Proc Natl Acad Sci USA 97: 12127–12131.
Technau U, Rudd S, Maxwell P, Gordon PM, Saina M, Grasso LC et al. (2005). Maintenance of ancestral complexity and non-metazoan genes in two basal cnidarians. Trends Genet 21: 633–639.
Technau U, Scholz CB . (2003). Origin and evolution of endoderm and mesoderm. Int J Dev Biol 47: 531–539.
Teo R, Mohrlen F, Plickert G, Muller WA, Frank U . (2006). An Evolutionary conserved role of wnt signaling in stem cell fate decision. Dev Biol 289: 91–99.
Torras R, Gonzalez-Crespo S . (2005). Posterior expression of nanos orthologs during embryonic and larval development of the anthozoan Nematostella vectensis. Int J Dev Biol 49: 895–899.
Torras R, Yanze N, Schmid V, Gonzalez-Crespo S . (2004). Nanos expression at the embryonic posterior pole and the medusa phase in the hydrozoan podocoryne carnea. Evol Dev 6: 362–371.
Tree DR, Ma D, Axelrod JD . (2002). A three-tiered mechanism for regulation of planar cell polarity. Semin Cell Dev Biol 13: 217–224.
Veeman MT, Axelrod JD, Moon RT . (2003). A second canon. Functions and mechanisms of beta-catenin-independent Wnt Signaling. Dev Cell 5: 367–377.
Wikramanayake AH, Hong M, Lee PN, Pang K, Byrum CA, Bince JM et al. (2003). An ancient role for nuclear beta-catenin in the evolution of axial polarity and germ layer segregation. Nature 426: 446–450.
Wittlieb JK, Khalturin K, Lohmann JU, Anton-Erxleben F, Bosch TC . (2006). Transgenic Hydra allow in vivo tracking of individual stem cells during morphogenesis. Proc Natl Acad Sci USA 103: 6208–6211.
Yang Y . (2003). Wnts and wing: Wnt signaling in vertebrate limb development and musculoskeletal morphogenesis. Birth Defects Res Part C Embryo Today 69: 305–317.
Acknowledgements
This work was supported by the FWF (FP16685) and DFG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guder, C., Philipp, I., Lengfeld, T. et al. The Wnt code: cnidarians signal the way. Oncogene 25, 7450–7460 (2006). https://doi.org/10.1038/sj.onc.1210052
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210052
Keywords
This article is cited by
-
Hox genes regulate asexual reproductive behavior and tissue segmentation in adult animals
Nature Communications (2021)
-
Medusozoan genomes inform the evolution of the jellyfish body plan
Nature Ecology & Evolution (2019)
-
Temperature and insulin signaling regulate body size in Hydra by the Wnt and TGF-beta pathways
Nature Communications (2019)
-
Wnt evolution and function shuffling in liberal and conservative chordate genomes
Genome Biology (2018)
-
Hox and Wnt pattern the primary body axis of an anthozoan cnidarian before gastrulation
Nature Communications (2018)