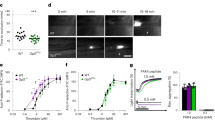
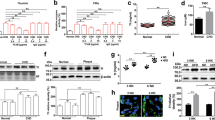
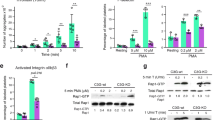
Abstract
Thrombin is a coagulation protease that activates platelets, leukocytes, endothelial and mesenchymal cells at sites of vascular injury, acting partly through an unusual proteolytically activated G-protein-coupled receptor1–3. Knockout of the gene encoding this receptor provided definitive evidence for a second thrombin receptor in mouse platelets and for tissue-specific roles for different thrombin receptors4. We now report the cloning and characterization of a new human thrombin receptor, designated protease-activated receptor 3 (PAR3). PAR3 can mediate throm-bin-triggered phosphoinositide hydrolysis and is expressed in a variety of tissues, including human bone marrow and mouse megakaryocytes, making it a candidate for the sought-after second platelet thrombin receptor. PAR3 provides a new tool for understanding thrombin signalling and a possible target for therapeutics designed selectively to block thrombotic, inflammatory and proliferative responses to thrombin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fenton, J. W. Regulation of thrombin generation and functions. Sem. Hemostas. Thrombos. 14, 234–240 (1988).
Coughlin, S. R. Thrombin receptor function and cardiovascular disease. Trends Cardiovas. Med. 4, 77–83 (1993).
Vu, T.-K. H., Hung, D. T., Wheaton, V. I. & Coughlin, S. R. Molecular cloning of a functional thrombin receptor reveals a novel proteolytic mechanisms of recetor activation. Cell 64, 1057–1068 (1991).
Connolly, A., Ishihara, H., Kahn, M., Farese, R. V. Jr & Coughlin, S. R. Role of the thrombin receptor in development and evidence for a second receptor. Nature 381, 516–519 (1996).
Nystedt, S., Emilsson, K., Wahlestedt, C. & Sundelin, J. Molecular cloning of a potential novel proteinase activated receptor. Proc. Natl Acad. Sci. USA 91, 9208–9212 (1994).
Rydel, T. J. et al. The structure of a complex of recombinant hirudin and human α-thrombin. Sceince 249, 277–280 (1990).
Vu, T.-K. H., Wheaton, V. I., Hung, D. T. & Coughlin, S. R. Domains specifying thrombin-receptor interaction. Nature 353, 674–677 (1991).
Liu, L., Vu, T.-K. H., Esmon, C. T. & Coughlin, S. R. The region of the thrombin receptor resembling hirudin binds to thrombin and alters enzyme specificity. J. Biol. Chem. 266, 16977–16980 (1991).
Mathews, I. I. et al. Crystallographic structures of thrombin complexed with thrombin receptor peptides: existence of expected and novel binding modes. Biochemistry 33, 3266–3279 (1994).
Ishii, K., Gerszten, R., Zheng, Y.-W., Turck, C. W. & Coughlin, S. R. Determinants of thrombin receptor cleavage: Receptor domains involved, specificity, and role of the P3 aspartate. J. Biol. Chem. 270, 16435–16440 (1995).
Chen, J., Ishii, M., Wang, L., Ishii, K. & Coughlin, S. R. Thrombin receptor activation: Confirmation of the intramolecular tethered liganding hypothesis and discovery of an alternative intermolecular liganding mode. J. Biol. Chem. 269 16041–16045 (1994).
Gerszten, R. E. et al. The thrombin receptor's specificity for agonist peptide is defined by its extracellular surface. Nature 368, 648–651 (1994).
Amatruda, T. T., Steele, D. A., Slepak, V. Z. & Simon, M. I. Gαl9, a G protein α subunit specifically expressed in hematopoietic cells. J. Biol. Chem. 88, 5587–5591 (1991).
Kettner, C. & Shaw, E. D-phe-pro-argCH2Cl: a selective affinity label for thrombin. Thrombosis Res. 14, 969–973 (1979).
Rydel, T. J. et al. Crystallographic structure of human γ-thrombin. J. Biol. Chem. 269, 22000–22006 (1994).
Skrzypczak, J. E. et al. Structure of the hirugen and hirulog 1 complexes of α-thrombin. J. Mol. Biol. 221, 1379–1393 (1991).
Scarborough, R. M. et al. Tethered ligand agonist peptides: structural requirements for thrombin receptor activation reveal mechanism of proteolytic unmasking of agonist fucntion. J. Biol. Chem. 267, 13146–13149 (1992).
Hung, D. T., Vu, T.-K. H., Wheaton, V. I., Ishii, K. & Coughlin, S. R. The cloned platelet thrombin receptor is necessary for thrombin-induced platelet activation. Blocking antiserum to the thrombin receptor's hirudin-like domain. J. Clin. Invest. 89, 1350–1353 (1992).
Brass, L. F. et al. Structure and function of the human paltelet thrombin receptor. Studies using monoclonal antibodies directed against a defined domain within the receptor N terminus. J. Biol. Chem. 267, 13795–13798 (1992).
Kinlough Rathbone, R., Perry, D. W., Guccione, M. A., Rand, M. L. & Packham, M. A. Degranulation of human paltelets by the thrombin receptor peptide SFLLRN: omparison with degranulation by thrombin. Thromb. Haemost. 70, 1019–1023 (1993).
Goodwin, C. A. et al. Thrombin receptor activating peptide does not stimulate platelet procoagulant activity. Biochem. Biophys. Res. Commun. 202, 321–327 (1994).
Lau, L. F., Pumiglia, K., Cote, Y. P. & Feinstein, M. B. Thrombin-receptor agonist peptides, in contrast to thrombin itself, are not full agonists for activation and signal transduction in human platelets in the absence of platelet-derived secondary mediators. Biochem. J. 303 391–400 (1994).
Kramer, R. M. et al. Differential activation of cytosolic phospholipase A2 (cPLA2) by thrombin and thrombin receptor agonist peptide in human platelets, evidence for activation of cPLA2 independent of the mitogen-activated protein kinases ERK1/2. J. Biol. Chem. 270, 14816–14823 (1995).
Connolly, T. M. et al. Species variability in platelet and other cellular responsiveness to thrombin receptor-derived peptides. Thromb. Haemost. 72, 627–633 (1994).
Derian, C. K., Santulli, R. J., Tomko, K. A., Haertlein, B. J. & Andrade-Gordon, P. Species differences in paltelet responses to thrombin and SFLLRN. Receptor-mediated calcium mobilization and aggregation and regulation by protein kinases. Thrombosis Res. 6, 505–519 (1995).
Probst, W. C., Snyder, L. A., Schuster, D. I., Brosius, J. & Sealfon, S. C. Sequence alignment of the G-protein coupled receptor superfamily. DNA and Cell 11, 1–20 (1992).
Ishii, K., Hein, L., Kobilka, B. & Coughlin, S. R. Kinetics of thrombin receptor cleavage on intact cells: relation to signaling. J. Biol. Chem. 268, 9780–9786 (1993).
Williams, J. A., McChesney, D. J., Calayag, M. C., Lingappa, V. R. & Logsdon, C. D. Expression of receptors for cholecystokinin and other Ca2+-mobilizing hormones in Xenopus oocytes. Proc. Natl Acad. Sci. USA 85, 4939–4943 (1988).
Nanevicz, T., Wang, L., Chen, M., Ishii, M. & Coughlin, S. R. Activating mutations in the thrombin receptor's agonist recognition domain. Mutations in a G protein-coupled receptor's extracellular domain cause transmembrane signaling. J. Biol. Chem. 271, 702–706 (1996).
Soifer, S. J., Peters, K. G., O'Keefe, J. & Coughlin, S. R. Disparate temporal expression of the prothrombin and thrombin receptor genes during mouse development. Am. J. Pathol. 144, 60–69 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishihara, H., Connolly, A., Zeng, D. et al. Protease-activated receptor 3 is a second thrombin receptor in humans. Nature 386, 502–506 (1997). https://doi.org/10.1038/386502a0
Issue Date:
DOI: https://doi.org/10.1038/386502a0
This article is cited by
-
Diversification of PAR signaling through receptor crosstalk
Cellular & Molecular Biology Letters (2022)
-
Thrombin induces morphological and inflammatory astrocytic responses via activation of PAR1 receptor
Cell Death Discovery (2022)
-
Effects of intermittent hypoxia with thrombin in an in vitro model of human brain endothelial cells and their impact on PAR-1/PAR-3 cleavage
Scientific Reports (2022)
-
PARs in the inflammation-cancer transformation of CRC
Clinical and Translational Oncology (2022)
-
Protease-activated receptors (PARs): mechanisms of action and potential therapeutic modulators in PAR-driven inflammatory diseases
Thrombosis Journal (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.