Abstract
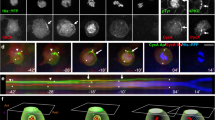
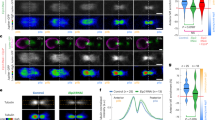
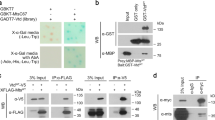
The orientation of the mitotic spindle relative to the cell axis determines whether polarized cells undergo symmetric or asymmetric divisions. Drosophila epithelial cells and neuroblasts provide an ideal pair of cells to study the regulatory mechanisms involved. Epithelial cells divide symmetrically, perpendicular to the apical–basal axis. In the asymmetric divisions of neuroblasts, by contrast, the spindle reorients parallel to that axis, leading to the unequal distribution of cell-fate determinants to one daughter cell1. Receptor-independent G-protein signalling involving the GoLoco protein Pins is essential for spindle orientation in both cell types2. Here, we identify Mushroom body defect (Mud)3 as a downstream effector in this pathway. Mud directly associates and colocalizes with Pins at the cell cortex overlying the spindle pole(s) in both neuroblasts and epithelial cells. The cortical Mud protein is essential for proper spindle orientation in the two different division modes. Moreover, Mud localizes to centrosomes during mitosis independently of Pins to regulate centrosomal organization. We propose that Drosophila Mud, vertebrate NuMA4 and Caenorhabditis elegans Lin-5 (refs 5, 6) have conserved roles in the mechanism by which G-proteins regulate the mitotic spindle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Betschinger, J. & Knoblich, J.A. Dare to be different: asymmetric cell division in Drosophila, C. elegans and vertebrates. Curr. Biol. 14, R674–R685 (2004).
Izumi, Y., Ohta, N., Itoh-Furuya, A., Fuse, N. & Matsuzaki, F. Differential functions of G protein and Baz-aPKC signaling pathways in Drosophila neuroblast asymmetric division. J. Cell Biol. 164, 729–738 (2004).
Guan, Z. et al. Mushroom body defect, a gene involved in the control of neuroblast proliferation in Drosophila, encodes a coiled-coil protein. Proc. Natl Acad. Sci. USA 97, 8122–8127 (2000).
Du, Q., Stukenberg, P.T. & Macara, I.G. A mammalian Partner of inscuteable binds NuMA and regulates mitotic spindle organization. Nature Cell Biol. 3, 1069–1075 (2001).
Colombo, K. et al. Translation of polarity cues into asymmetric spindle positioning in Caenorhabditis elegans embryos. Science 300, 1957–1961 (2003).
Srinivasan, D.G., Fisk, R.M., Xu, H. & van den Heuvel, S. A complex of LIN-5 and GPR proteins regulates G protein signaling and spindle function in C. elegans. Genes Dev. 17, 1225–1239 (2003).
Schaefer, M., Petronczki, M., Dorner, D., Forte, M. & Knoblich, J.A. Heterotrimeric G proteins direct two modes of asymmetric cell division in the Drosophila nervous system. Cell 107, 183–194 (2001).
Wilkie, T.M. & Kinch, L. New roles for Gα and RGS proteins: communication continues despite pulling sisters apart. Curr. Biol. 15, R843–R854 (2005).
Yu, F. et al. Locomotion defects, together with Pins, regulates heterotrimeric G-protein signaling during Drosophila neuroblast asymmetric divisions. Genes Dev. 19, 1341–1353 (2005).
Bellaiche, Y. & Gotta, M. Heterotrimeric G proteins and regulation of size asymmetry during cell division. Curr. Opin. Cell Biol. 17, 658–663 (2005).
Schober, M., Schaefer, M. & Knoblich, J.A. Bazooka recruits Inscuteable to orient asymmetric cell divisions in Drosophila neuroblasts. Nature 402, 548–551 (1999).
Yu, F., Morin, X., Cai, Y., Yang, X. & Chia, W. Analysis of partner of inscuteable, a novel player of Drosophila asymmetric divisions, reveals two distinct steps in inscuteable apical localization. Cell 100, 399–409 (2000).
Wodarz, A., Ramrath, A., Kuchinke, U. & Knust, E. Bazooka provides an apical cue for Inscuteable localization in Drosophila neuroblasts. Nature 402, 544–547 (1999).
Yu, F., Cai, Y., Kaushik, R., Yang, X. & Chia, W. Distinct roles of Gαi and Gβ13F subunits of the heterotrimeric G protein complex in the mediation of Drosophila neuroblast asymmetric divisions. J. Cell Biol. 162, 623–633 (2003).
Gotta, M. & Ahringer, J. Distinct roles for Gα and Gβγ in regulating spindle position and orientation in Caenorhabditis elegans embryos. Nature Cell Biol. 3, 297–300 (2001).
Du, Q. & Macara, I.G. Mammalian Pins is a conformational switch that links NuMA to heterotrimeric G proteins. Cell 119, 503–516 (2004).
Prokop, A. & Technau, G.M. Normal function of the mushroom body defect gene of Drosophila is required for the regulation of the number and proliferation of neuroblasts. Dev. Biol. 161, 321–337 (1994).
Cai, Y., Yu, F., Lin, S., Chia, W. & Yang, X. Apical complex genes control mitotic spindle geometry and relative size of daughter cells in Drosophila neuroblast and pI asymmetric divisions. Cell 112, 51–62 (2003).
Kraut, R., Chia, W., Jan, L.Y., Jan, Y. N & Knoblich, J.A. Role of inscuteable in orienting asymmetric cell divisions in Drosophila. Nature 383, 50–55 (1996).
Merdes, A., Ramyar, K., Vechio, J.D. & Cleveland, D.W. A complex of NuMA and cytoplasmic dynein is essential for mitotic spindle assembly. Cell 87, 447–458 (1996).
Ahringer, J. Control of cell polarity and mitotic spindle positioning in animal cells. Curr. Opin. Cell Biol. 15, 73–81 (2003).
Siegrist, S.E. & Doe, C.Q. Microtubule-induced Pins/Gαi cortical polarity in Drosophila neuroblasts. Cell 123, 1323–1335 (2005).
Gaglio, T., Saredi, A. & Compton, D.A. NuMA is required for the organization of microtubules into aster-like mitotic arrays. J. Cell Biol. 131, 693–708 (1995).
Merdes, A., Heald, R., Samejima, K., Earnshaw, W.C. & Cleveland, D.W. Formation of spindle poles by dynein/dynactin-dependent transport of NuMA. J. Cell Biol. 149, 851–862 (2000).
Siller, K.H., Serr, M., Steward, R., Hays, T. S. & Doe, C.Q. Live imaging of Drosophila brain neuroblasts reveals a role for lis1/dynactin in spindle assembly and mitotic checkpoint control. Mol. Biol. Cell 16, 5127–5140 (2005).
Giot, L. et al. A protein interaction map of Drosophila melanogaster. Science 302, 1727–1736 (2003).
Lu, B., Roegiers, F., Jan, L.Y. & Jan, Y.N. Adherens junctions inhibit asymmetric division in the Drosophila epithelium. Nature 409, 522–525 (2001).
Berrueta, L., Tirnauer, J.S., Schuyler, S.C., Pellman, D . & Bierer, B. E. The APC-associated protein EB1 associates with components of the dynactin complex and cytoplasmic dynein intermediate chain. Curr. Biol. 9, 425–428 (1999).
Askham, J.M., Vaughan, K.T., Goodson, H.V. & Morrison, E.E. Evidence that an interaction between EB1 and p150(Glued) is required for the formation and maintenance of a radial microtubule array anchored at the centrosome. Mol. Biol. Cell 13, 3627–3645 (2002).
Ligon, L.A., Shelly, S.S., Tokito, M. & Holzbaur, E.L. The microtubule plus-end proteins EB1 and dynactin have differential effects on microtubule polymerization. Mol. Biol. Cell 14, 1405–1417 (2003).
Chou, T.B. & Perrimon, N. Use of a yeast site-specific recombinase to produce female germline chimeras in Drosophila. Genetics 131, 643–653 (1992).
Acknowledgements
We thank F. Yu, W. Chia, J. Knoblich and Developmental Studies Hybridoma Bank and the Bloomington Stock Center for providing flies and antibodies. We also thank N. Fuse, W. Moon, H. Ogawa, A. Nakamura, T. Issiki, F. Yu and W. Chia for discussions and comments on the manuscript. This work was supported by CREST (Core Research for Evolution Science and Technology) for the Japan Science and Technology Corporation and a grant from the Deutsche Forschungsgemeinschaft (Ra561/5-2). Y. I. is supported by a Special Postdoctoral Research Program of RIKEN.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3 and S4 (PDF 750 kb)
Rights and permissions
About this article
Cite this article
Izumi, Y., Ohta, N., Hisata, K. et al. Drosophila Pins-binding protein Mud regulates spindle-polarity coupling and centrosome organization. Nat Cell Biol 8, 586–593 (2006). https://doi.org/10.1038/ncb1409
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1409
This article is cited by
-
Polarized branched Actin modulates cortical mechanics to produce unequal-size daughters during asymmetric division
Nature Cell Biology (2023)
-
Cytoskeletal control of early mammalian development
Nature Reviews Molecular Cell Biology (2021)
-
Centrosome instability: when good centrosomes go bad
Cellular and Molecular Life Sciences (2021)
-
Liquid-liquid phase separation in biology: mechanisms, physiological functions and human diseases
Science China Life Sciences (2020)
-
The last-born daughter cell contributes to division orientation of Drosophila larval neuroblasts
Nature Communications (2018)