Abstract
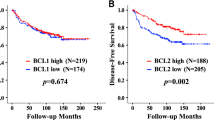
Traditional clinicopathological features do not predict which patients will develop chemotherapy resistance. The TP53 gene is frequently altered in ovarian cancer but its prognostic implications are controversial. Little is known on the impact of TP53-downstream genes on prognosis. Using molecular and immunohistochemical analyses we examined TP53 and its downstream genes p21 BAX and BCL-2 in ovarian tumour tissues and have evaluated the results in relation to clinico-pathological parameters, clinical outcome and response to platinum-based chemotherapy. Associations of tested factors and patient and tumour characteristics were studied by Spearman rank correlation and Pearsons χ2 test. The Cox proportional hazard model was used for univariate and multivariate analysis. The associations of tested factors with response was tested using logistic regression analysis. TP53 mutation, p21 and BCL-2 expression were not associated with increased rates of progression and death. Expression of TP53 was associated with a shorter overall survival only (relative hazard rate [RHR] 2.01 P= 0.03). Interestingly, when combining TP53 mutation and expression data, this resulted in an increased association with overall survival (P = 0.008). BAX expression was found to be associated with both progression-free (RHR 0.44 P = 0.05) and overall survival (RHR 0.42 P = 0.03). Those patients who simultaneously expressed BAX and BCL-2 had a longer progression-free and overall survival compared to patients whose tumours did not express BCL-2 (P = 0.05 and 0.015 respectively). No relations were observed between tested factors and response to platinum-based chemotherapy. We conclude that BAX expression may represent a prognostic indicator for patients with ovarian cancer and that the combined evaluation of BAX and BCL-2 may provide additional prognostic significance. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Anttila MA, Kosma VM, Hongxiu J, Puolakka J, Juhola M, Saarikoski S and Syrjanen K (1999) p21/WAF1 expression as related to p53, cell proliferation and prognosis in epithelial ovarian cancer. Br J Cancer 79: 1870–1878
Baekelandt M, Holm R, Trope CG, Nesland JM and Kristensen GB (1999a) Lack of independent prognostic significance of p21 and p27 expression in advanced ovarian cancer: an immunohistochemical study. Clin Cancer Res 5: 2848–2853
Baekelandt M, Kristensen G, Nesland J, Trope C and Holm R (1999b) Clinical significance of apoptosis-related factors p53, Mdm2, and Bcl-2 in advanced ovarian cancer. J Clin Oncol 17: 2061–2068
Baekelandt M, Holm R, Nesland JM, Trope CG and Kristensen GB (2000) Expression of apoptosis-related proteins is an independent determinant of patient prognosis in advanced ovarian cancer. J Clin Oncol 22: 3775–3781
Barlow R, Bartholomew D and Bremner J (1993) Statistical interference under order restrictions. John Wiley & Sons: London
Berns EM, Klijn JG, Smid M, van Staveren IL, Look MP, van Putten WL and Foekens JA (1996) TP53 and MYC gene alterations independently predict poor prognosis in breast cancer patients. Genes Chromosomes Cancer 16: 170–179
Bonetti A, Zaninelli M, Leone R, Cetto GL, Pelosi G, Biolo S, Menghi A, Manfrin E, Bonetti F and Piubello Q (1998) bcl-2 but not p53 expression is associated with resistance to chemotherapy in advanced breast cancer. Clin Cancer Res 4: 2331–2336
Buttitta F, Marchetti A, Gadducci A, Pellegrini S, Morganti M, Carnicelli V, Cosio S, Gagetti O, Genazzani AR and Bevilacqua G (1997) p53 alterations are predictive of chemoresistance and aggressiveness in ovarian carcinomas: a molecular and immunohistochemical study. Br J Cancer 75: 230–235
Casey G, Lopez ME, Ramos JC, Plummer SJ, Arboleda MJ, Shaughnessy M, Karlan B and Slamon DJ (1996) DNA sequence analysis of exons 2 through 11 and immunohistochemical staining are required to detect all known p53 alterations in human malignancies. Oncogene 13: 1971–1981
Diebold J, Baretton G, Felchner M, Meier W, Dopfer K, Schmidt M and Lohrs U (1996) bcl-2 expression, p53 accumulation, and apoptosis in ovarian carcinomas. Am J Clin Pathol 105: 341–349
Dive C and Hickman JA (1991) Drug-target interactions: only the first step in the commitment to a programmed cell death?. Br J Cancer 64: 192–196
Dong Y, Walsh MD, McGuckin MA, Cummings MC, Gabrielli BG, Wright GR, Hurst T, Khoo SK and Parsons PG (1997) Reduced expression of retinoblastoma gene product (pRB) and high expression of p53 are associated with poor prognosis in ovarian cancer. Int J Cancer 74: 407–415
Eastman A (1987) The formation, isolation and characterization of DNA adducts produced by anticancer platinum complexes. Pharmacol Ther 34: 155–166
Eastman A (1990) Activation of programmed cell death by anticancer agents: cisplatin as a model system. Cancer Cells 2: 275–280
el-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW and Vogelstein B (1993) WAF1, a potential mediator of p53 tumor suppression. Cell 75: 817–825
el-Deiry WS, Harper JW, O'Connor PM, Velculescu VE, Canman CE, Jackman J, Pietenpol JA, Burrell M, Hill DE and Wang Y (1994) WAF1/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res 54: 1169–1174
EORTC Breast Cancer Cooperative Group (1980) Revision of the standards for the assessment of hormone receptors in human breast cancer; report of the second E.O.R.T.C. Workshop, held on 16–17 March, 1979, in the Netherlands Cancer Institute. Eur J Cancer 16: 1513–1515
Ferrandina G, Fagotti A, Salerno M, Natali P, Mottolese M, Maneschi F, Pasqua AD, Benedetti-Panici P, Mancuso S and Scambia G (1999) p53 overexpression is associated with cytoreduction and response to chemotherapy in ovarian cancer. Br J Cancer 81: 733–740
Gasparini G, Barbareschi M, Doglioni C, Palma PD, Mauri FA, Boracchi P, Bevilacqua P, Caffo O, Morelli L and Verderio P (1995) Expression of bcl-2 protein predicts efficacy of adjuvant treatments in operable node-positive breast cancer. Clin Cancer Res 1: 189–198
Hartmann LC, Podratz KC, Keeney GL, Kamel NA, Edmonson JH, Grill JP, Su JQ, Katzmann JA and Roche PC (1994) Prognostic significance of p53 immunostaining in epithelial ovarian cancer. J Clin Oncol 12: 64–69
Henriksen R, Wilander E and Oberg K (1995) Expression and prognostic significance of Bcl-2 in ovarian tumours. Br J Cancer 72: 1324–1329
Herod JJ, Eliopoulos AG, Warwick J, Niedobitek G, Young LS and Kerr DJ (1996) The prognostic significance of Bcl-2 and p53 expression in ovarian carcinoma. Cancer Res 56: 2178–2184
Hockenbery D, Nunez G, Milliman C, Schreiber RD and Korsmeyer SJ (1990) Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death. Nature 348: 334–336
Kaplan E and Meier P (1958) Non-parametric estimation from incomplete observations. J Am Stat Assoc 53: 457–481
Kassim SK, Ali HS, Sallam MM, Fayed ST, Seada LS, abd-Elkawy E, Seada MA and Khalifa A (1999) Increased bcl-2 expression is associated with primary resistance to chemotherapy in human epithelial ovarian cancer. Clin Biochem 32: 333–338
Kohler MF, Kerns BJ, Humphrey PA, Marks JR, Bast RC Jr and Berchuck A (1993) Mutation and overexpression of p53 in early-stage epithelial ovarian cancer. Obstet Gynecol 81: 643–650
Kottmeier HL (1976) Presentation of therapeutic results in carcinoma of the female pelvis: experience of the Annual Report on the Results of Treatment in Carcinoma of the Uterus, Vagina, and Ovary. Gynecol Oncol 4: 13–19
Koukourakis MI, Giatromanolaki A, O'Byrne KJ, Whitehouse RM, Talbot DC, Gatter KC and Harris AL (1997) Potential role of bcl-2 as a suppressor of tumour angiogenesis in non-small-cell lung cancer. Int J Cancer 74: 565–570
Mano Y, Kikuchi Y, Yamamoto K, Kita T, Hirata J, Tode T, Ishii K and Nagata I (1999) Bcl-2 as a predictor of chemosensitivity and prognosis in primary epithelial ovarian cancer. Eur J Cancer 35: 1214–1219
Marks JR, Davidoff AM, Kerns BJ, Humphrey PA, Pence JC, Dodge RK, Clarke-Pearson DL, Iglehart JD, Bast RC Jr and Berchuck A (1991) Overexpression and mutation of p53 in epithelial ovarian cancer. Cancer Res 51: 2979–2984
Marone M, Scambia G, Mozzetti S, Ferrandina G, Iacovella S, De Pasqua A, Benedetti-Panici P and Mancuso S (1998) bcl-2, bax, bcl-XL, and bcl-XS expression in normal and neoplastic ovarian tissues. Clin Cancer Res 4: 517–524
Marx D, Binder C, Meden H, Lenthe T, Ziemek T, Hiddemann T, Kuhn W and Schauer A (1997) Differential expression of apoptosis associated genes bax and bcl-2 in ovarian cancer. Anticancer Res 17: 2233–2240
Miyashita T, Kitada S, Krajewski S, Horne WA, Delia D and Reed JC (1995) Overexpression of the Bcl-2 protein increases the half-life of p21 Bax. J Biol Chem 270: 26049–26052
Miyashita T and Reed JC (1993) Bcl-2 oncoprotein blocks chemotherapy-induced apoptosis in a human leukemia cell line. Blood 81: 151–157
Miyashita T, Krajewski S, Krajewska M, Wang HG, Lin HK, Liebermann DA, Hoffman B and Reed JC (1994) Tumor suppressor p53 is a regulator of bcl-2 and bax gene expression in vitro and in vivo. Oncogene 9: 1799–1805
Miyashita T and Reed JC (1995) Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 80: 293–299
Neijt JP, ten Bokkel Huinink WW, van der Burg ME, yan Oosterom AT, Willemse PH, Vermorken JB, van Lindert AC, Heintz AP, Aartsen E and van Lent M (1991) Long-term survival in ovarian cancer. Mature data from The Netherlands Joint Study Group for Ovarian Cancer. Eur J Cancer 27: 1367–1372
Niwa K, Itoh M, Murase T, Morishita S, Itoh N, Mori H and Tamaya T (1994) Alteration of p53 gene in ovarian carcinoma: clinicopathological correlation and prognostic significance. Br J Cancer 70: 1191–1197
Oltvai ZN, Milliman CL and Korsmeyer SJ (1993) Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74: 609–619
Oltvai ZN and Korsmeyer SJ (1994) Checkpoints of dueling dimers foil death wishes. Cell 79: 189–192
Ozols R, Rubin S and Dembo A (1992) Principles and practice of gynecologic oncology. In: Epithelial Ovarian Cancer, Hoskins W, Perez C and Young R (eds) pp 731–781. Lippincott: Philadelphia
Perego P, Giarola M, Righetti SC, Supino R, Caserini C, Delia D, Pierotti MA, Miyashita T, Reed JC and Zunino F (1996) Association between cisplatin resistance and mutation of p53 gene and reduced bax expression in ovarian carcinoma cell systems. Cancer Res 56: 556–562
Pietenpol JA, Papadopoulos N, Markowitz S, Willson JK, Kinzler KW and Vogelstein B (1994) Paradoxical inhibition of solid tumor cell growth by bcl2. Cancer Res 54: 3714–3717
Righetti SC, Della Torre G, Pilotti S, Menard S, Ottone F, Colnaghi MI, Pierotti MA, Lavarino C, Cornarotti M, Oriana S, Bohm S, Bresciani GL, Spatti G and Zunino F (1996) A comparative study of p53 gene mutations, protein accumulation, and response to cisplatin-based chemotherapy in advanced ovarian carcinoma. Cancer Res 56: 689–693
Rohlke P, Milde-Langosch K, Weyland C, Pichlmeier U, Jonat W and Loning T (1997) p53 is a persistent and predictive marker in advanced ovarian carcinomas: multivariate analysis including comparison with Ki67 immunoreactivity. J Cancer Res Clin Oncol 123: 496–501
Schuyer M, Henzen-Logmans SC, van der Burg ME, Fieret EJ, Klijn JG, Foekens JA and Berns EM (1998) High prevalence of codon 213 ArgStop mutations of the TP53 gene in human ovarian cancer in the southwestern part of the Netherlands. Int J Cancer 76: 299–303
Shelling AN, Cooke IE and Ganesan TS (1995) The genetic analysis of ovarian cancer. Br J Cancer 72: 521–527
Skilling JS, Sood A, Niemann T, Lager DJ and Buller RE (1996) An abundance of p53 null mutations in ovarian carcinoma. Oncogene 13: 117–123
Strobel T, Swanson L, Korsmeyer S and Cannistra SA (1996) BAX enhances paclitaxel-induced apoptosis through a p53-independent pathway. Proc Natl Acad Sci USA 93: 14094–14099
Strobel T, Swanson L, Korsmeyer S and Cannistra SA (1997) Radiation-induced apoptosis is not enhanced by expression of either p53 or BAX in SW626 ovarian cancer cells. Oncogene 14: 2753–2758
Strobel T, Kraeft SK, Chen LB and Cannistra SA (1998) BAX expression is associated with enhanced intracellular accumulation of paclitaxel: a novel role for BAX during chemotherapy-induced cell death. Cancer Res 58: 4776–4781
Tai YT, Lee S, Niloff E, Weisman C, Strobel T and Cannistra SA (1998) BAX protein expression and clinical outcome in epithelial ovarian cancer. J Clin Oncol 16: 2583–2590
van der Burg ME, Henzen-Logmans SC, Berns EM, van Putten WL, Klijn JG and Foekens JA (1996) Expression of urokinase-type plasminogen activator (uPA) and its inhibitor PAI-1 in benign, borderline, malignant primary and metastatic ovarian tumors. Int J Cancer 69: 475–479
van der Zee AG, Hollema H, Suurmeijer AJ, Krans M, Sluiter WJ, Willemse PH, Aalders JG and de Vries EG (1995) Value of P-glycoprotein, glutathione S-transferase pi, c-erbB-2, and p53 as prognostic factors in ovarian carcinomas. J Clin Oncol 13: 70–78
Wen WH, Reles A, Runnebaum IB, Sullivan-Halley J, Bernstein L, Jones LA, Felix JC, Kreienberg R, el-Naggar A and Press MF (1999) p53 mutations and expression in ovarian cancers: correlation with overall survival. Int J Gynecol Pathol 18: 29–41
World Health Organization (1979) WHO handbook for reporting results of cancer treatments. Vol. no. 49. World Health Organization offset publication: Geneva
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Schuyer, M., Burg, M., Henzen-Logmans, S. et al. Reduced expression of BAX is associated with poor prognosis in patients with epithelial ovarian cancer: a multifactorial analysis of TP53, p21, BAX and BCL-2 . Br J Cancer 85, 1359–1367 (2001). https://doi.org/10.1054/bjoc.2001.2101
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2101
Keywords
This article is cited by
-
Lower expression of Bax predicts poor clinical outcome in patients with glioma after curative resection and radiotherapy/chemotherapy
Journal of Neuro-Oncology (2019)
-
Oncogenes associated with drug resistance in ovarian cancer
Journal of Cancer Research and Clinical Oncology (2015)
-
Prognostic significance of several biomarkers in epithelial ovarian cancer: a meta-analysis of published studies
Journal of Cancer Research and Clinical Oncology (2013)
-
Immunohistochemistry with apoptotic-antiapoptotic proteins (p53, p21, bax, bcl-2), c-kit, telomerase, and metallothionein as a diagnostic aid in benign, borderline, and malignant serous and mucinous ovarian tumors
Diagnostic Pathology (2012)
-
Multifunctional Tumor-Targeted Polymer-Peptide-Drug Delivery System for Treatment of Primary and Metastatic Cancers
Pharmaceutical Research (2010)