Abstract
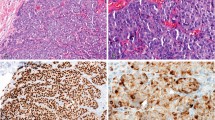
Apoptosis is of important significance in the pathogenesis of cancer. Many methods are available for the measurement of apoptosis but the ‘gold standard’ is to identify apoptotic cells by their morphological features using microscopy. Caspase 3 is a cytosolic enzyme that is activated only in cells committed to undergo apoptosis. The activation of caspase 3 precedes the development of the classical morphological features of apoptosis. Using immunohistochemistry with an antibody against the active form of caspase 3, the apoptotic index (AI) was measured in 116 samples of human breast tissue (22 normal/benign and 94 invasive carcinomas). The AI obtained by measuring caspase activation has a strong correlation with the AI derived by morphological assessment (r = 0.736, P < 0.01). The AI is higher in the invasive group than in the benign group (P = 0.008), and in invasive cancer high AI is associated with high tumour grade (P = 0.013), positive node status (P < 0.001) and negative steroid receptor status (P = 0.001 for ER; P = 0.004 for PR). No significant association is observed between AI and tumour size. Measurement of apoptosis by immunohistochemistry using an antibody against the active form of caspase 3 is therefore reliable and correlates strongly with morphological assessment. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ashkenazi A and Dixit VM (1999) Apoptosis control by death and decoy receptors. Current Opin Cell Biol 11: 255–260
Berardo MD, Elledge RM, de Moor C, Clark GM, Osborne CK and Allred DC (1998) bcl-2 and apoptosis in lymph node positive breast carcinoma. Cancer 82: 1296–1302
Frankfurt OS, Robb JA, Sugarbaker EV and Villa L (1997) Apoptosis in breast carcinomas detected with monoclonal antibody to single-stranded DNA: relation to bcl-2 expression, hormone receptors, and lymph node metastases. Clin Cancer Res 3: 465–471
Gavrieli Y, Sherman Y and Bensasson SA (1992) Identification of programmed cell-death insitu via specific labeling of nuclear-dna fragmentation. Journal of Cell Biology 119: 493–501
Gilmore AP, Metcalfe AM, Romer LH and Streuli CH (2000) Integrin-mediated survival signals regulate the apoptotic function of Bax through its conformation and subcellular localization. J Cell Biol 149: 431–445
Grasl-Kraupp B, Ruttkay-Nedecky B, Koudelka H, Bukowska K, Bursch W and Schulte-Hermann R (1995) In situ detection of fragmented DNA (TUNEL assay) fails to discriminate among apoptosis, necrosis, and autolytic cell death: a cautionary note. Hepatology 21: 1465–1468
Green DR (1998) Apoptotic pathways: the roads to ruin. Cell 94: 695–698
Hall PA (1999) Assessing apoptosis: a critical survey. Endocr Relat Cancer 6: 3–8
Harn HJ, Shen KL, Yueh KC, Ho LI, Yu JC, Chiu SC and Lee WH (1997) Apoptosis occurs more frequently in intraductal carcinoma than in infiltrating duct carcinoma of human breast cancer and correlates with altered p53 expression: detected by terminal-deoxynucleotidyl-transferase-mediated dUTP-FITC nick end labelling (TUNEL). Histopathology 31: 534–539
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407: 770–776
Lipponen P (1999) Apoptosis in breast cancer: relationship with other pathological parameters. Endocr Relat Cancer 6: 13–16
Lipponen P, Aaltomaa S, Kosma VM and Syrjanen K (1994) Apoptosis in breast cancer as related to histopathological characteristics and prognosis. Eur J Cancer 30A: 2068–2073
Mainwaring PN, Ellis PA, Detre S, Smith IE and Dowsett M (1998) Comparison of in situ methods to assess DNA cleavage in apoptotic cells in patients with breast cancer. J Clin Pathol 51: 34–37
Matter A (1979) Microcinematographic and electron microscopic analysis of target cell lysis induced by cytotoxic T lymphocytes. Immunology 36: 179–190
Potten CS (1996) What is an apoptotic index measuring? A commentary. Br J Cancer 74: 1743–1748
Rich T, Allen RL and Wyllie AH (2000) Defying death after DNA damage. Nature 407: 777–783
Rochaix P, Krajewski S, Reed JC, Bonnet F, Voigt JJ and Brousset P (1999) In vivo patterns of Bcl-2 family protein expression in breast carcinomas in relation to apoptosis. J Pathol 187: 410–415
Sanderson CJ (1976) The mechanism of T cell mediated cytotoxicity. II. Morphological studies of cell death by time-lapse microcinematography. Proc R Soc Lond B Biol Sci 192: 241–255
Sarasin A and Stary A (1997) Human cancer and DNA repair-deficient diseases. Cancer Detect Prev 21: 406–411
Savill J and Fadok V (2000) Corpse clearance defines the meaning of cell death. Nature 407: 784–788
Streuli CH and Gilmore AP (1999) Adhesion-mediated signaling in the regulation of mammary epithelial cell survival. Journal of Mammary Gland Biology and Neoplasia 4: 183–191
Thompson CB (1995) Apoptosis In the Pathogenesis and Treatment Of Disease. Science 267, (n5203):1456–1462
Vakkala M, Lahteenmaki K, Raunio H, Paakko P and Soini Y (1999) Apoptosis during breast carcinoma progression. Clin Cancer Res 5: 319–324
Vaux DL and Korsmeyer SJ (1999) Cell death in development. Cell 96: 245–254
Wyllie AH, Kerr JF and Currie AR (1980) Cell death: the significance of apoptosis. Int Rev Cytol 68: 251–306
Zhang GJ, Kimijima I, Abe R, Watanabe T, Kanno M, Hara K and Tsuchiya A (1998) Apoptotic index correlates to bcl-2 and p53 protein expression, histological grade and prognosis in invasive breast cancers. Anticancer Res 18: 1989–98
Zheng WQ and Zhan RZ (1998) Quantitative comparison of apoptosis to cell proliferation and p53 protein in breast carcinomas. Anal Quant Cytol Histol 20: 1–6
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Hadjiloucas, I., Gilmore, A., Bundred, N. et al. Assessment of apoptosis in human breast tissue using an antibody against the active form of caspase 3: relation to tumour histopathological characteristics. Br J Cancer 85, 1522–1526 (2001). https://doi.org/10.1054/bjoc.2001.2115
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2115
Keywords
This article is cited by
-
Evaluation of Human Wharton’s Jelly-Derived Mesenchymal Stem Cells Conditioning Medium (hWJ-MSCs-CM) or Scorpion Venom Breast Cancer Cell Line In Vitro
Journal of Gastrointestinal Cancer (2022)
-
Adeno-associated virus type 2 infection activates caspase dependent and independent apoptosis in multiple breast cancer lines but not in normal mammary epithelial cells
Molecular Cancer (2011)
-
Taurine: a novel tumor marker for enhanced detection of breast cancer among female patients
Angiogenesis (2011)
-
Effects of Extremely Low-Frequency Magnetic Field on Caspase Activities and Oxidative Stress Values in Rat Brain
Biological Trace Element Research (2010)
-
Short-term presurgical treatment for all?
Breast Cancer Research (2007)