-
PDF
- Split View
-
Views
-
Cite
Cite
J.A.Z. Loudon, S.R. Sooranna, P.R. Bennett, M.R. Johnson, Mechanical stretch of human uterine smooth muscle cells increases IL-8 mRNA expression and peptide synthesis, Molecular Human Reproduction, Volume 10, Issue 12, December 2004, Pages 895–899, https://doi.org/10.1093/molehr/gah112
Close - Share Icon Share
Abstract
Labour is associated with increased synthesis of interleukin-8 (IL-8) by the fetal membranes and myometrium, which leads to an inflammatory infiltrate. Stretch has been shown to increase the expression of contraction-associated proteins in animal models of labour and in human myocytes in vitro. In this study, we tested the hypothesis that mechanical stretch of human myometrial cells increases IL-8 messenger ribonucleic acid (mRNA) expression. We isolated myocytes from non-pregnant women undergoing hysterectomy and pregnant women undergoing Caesarean section before and after the onset of labour. Myocytes in culture were subjected to stretch of varying intensity (6–16%) and duration (1 or 6 h) using the Flexercell system. IL-8 mRNA expression was lowest in myocytes from pregnant women not in labour, intermediate in those from non-pregnant women and greatest in those from pregnant women in labour. Stretch increased IL-8 mRNA expression independent of reproductive state. The stretch-induced increase in IL-8 mRNA expression was associated with higher IL-8 levels in the culture supernatant and enhanced promoter activity. These data suggest that stretch contributes to the increase in myometrial IL-8 synthesis associated with the onset of labour in humans.
Introduction
During pregnancy, the uterus grows by both hyperplasia and hypertrophy allowing the uterus to accommodate the growing feto-placental unit. Stretch is an important stimulus to uterine growth, but in conditions such as multiple pregnancy, polyhydramnios or unicornuate uterus, stretch is excessive and associated with an increased risk of preterm labour and delivery. Animal studies suggest that uterine stretch in the absence of progesterone is associated with an increase in the expression of some contraction-associated proteins such as oxytocin receptor (OTR), cycloxygenase 2 (COX-2) and connexin-43 (Ou et al., 1997; Parry et al., 1997; Ou et al., 1998; Wu et al., 1999), but not others such as prostaglandin (PG) F2α receptor and connexin-26 (Ou et al., 1997, 2000). Uterine stretch in humans, achieved by the insertion and inflation of a 150 ml balloon, results in increased prostaglandin synthesis (Manabe et al., 1982). However, one study, comparing the expression of prostaglandin E (PGE) receptors and connexin proteins in singleton and twin pregnancies found no difference in their expression, questioning the role of stretch in the expression of these proteins (Lyall et al., 2002). In support of a role for stretch in the expression of pro-labour proteins we have shown that stretch of human uterine myocytes in vitro results in up-regulation of COX-2 messenger ribonucleic acid (mRNA) expression and protein synthesis in association with activator protein-1 (AP-1) activation (Sooranna et al., 2004) and of OTR expression in association with CCAAT/enhancer binding protein (C/EBP) activation (Terzidou et al., 2004). Consistent with our observation of stretch-induced AP-1 and C/EBP activation, in a series of studies, Lye et al. have demonstrated stretch-activated mitogen-activated protein kinase (MAPK) in uterine smooth muscle cells both in vivo and in vitro (Mitchell and Lye, 2002; Oldenhof et al., 2002; Shynlova et al., 2002).
Parturition begins only after weeks of preparation characterized by increased expression of contraction-associated proteins and pro-inflammatory cytokines such as interleukin-6 (IL-6) and interleukin-8 (IL-8). These changes result in increased uterine contractility and cervical compliance. The chemokine IL-8 is thought to contribute to the process of cervical maturation by stimulating extravasation of neutrophils that release matrix metalloproteinases denaturing the collagen within the extracellular matrix (ECM) (Osmers et al., 1992; Osmers et al., 1995a,b). When administered topically to the rabbit cervix, IL-8 induces the histological changes associated with cervical ripening (el Maradny et al., 1994). IL-8 expression in the human cervix, myometrium, fetal membranes and decidua during pregnancy is initially low but increases through the third trimester becoming maximal at term (Elliott et al., 2000; Elliott et al., 2001a). When prostaglandins are used to initiate human labour, IL-8 expression is greater than that observed in labour of spontaneous onset, suggesting the presence of an interaction between prostaglandins and chemokine expression (Chan et al., 2002). Furthermore, prostaglandins and IL-8 are suggested to act synergistically to promote neutrophil invasion in the cervix (Kelly, 2002).
Stretch of whole fetal membranes, of alveolar epithelial cells and of human airway smooth muscle cells increases IL-8 expression (Maehara et al., 1996; Kumar et al., 2003; Li et al., 2003). Given the effect of stretch on gene expression in animal and human studies and the role of IL-8 in the preparation for labour, we carried out this study to test the hypothesis that static stretch of uterine myocytes increases IL-8 mRNA expression and synthesis. Stretch may be constant, as might be expected during pregnancy, or episodic, as at the time of labour. In these studies, we have modelled stretch during pregnancy and consequently have used constant rather than episodic stretch.
Methods
Patient selection
Approval for the collection of myometrial biopsies was obtained from the regional ethics committee. Informed consent was sought from all patients participating in the study. Tissue was obtained from three groups of women: those undergoing hysterectomy for a benign indication and those undergoing Caesarean section both before and after the onset of labour.
Tissue specimens
Biopsies (0.5 × 0.5×0.5 cm3) of term human myometrium were collected at the time of Caesarean section from women not in labour (n=6), during active labour (n=6) and undergoing a hysterectomy (n=6). Biopsies were placed into Dulbecco's modified Eagle's medium (DMEM) containing 100 munits/ml penicillin and 100 μg/ml streptomycin. Samples were stored at 4°C for not more than 3 h prior to cell preparation for culture. Tissue from Caesarean section was removed from the upper margin of the incision made in the lower segment of the uterus. Mean maternal age (non-labour (NL), 31 [26–37] years; labour (L), 30 [27–40] years and non-pregnant (NP), 42 [39–47] median [range]). Gestational age (NL = 39 [38 + 3–39 + 2] and L = 38 + 5 [37–40 + 4] weeks) did not differ significantly. The indications for lower segment caesarean section (LSCS) in the labour group were: slow labour, fetal distress and breech presentation, and in the non-labour group, previous LSCS, breech presentation and maternal request.
Cell culture
Primary human uterine myocytes were isolated using a mixture of collagenases and cultured in DMEM medium 7.5% fetal calf serum, 100 munits/ml penicillin and 100 μg/ml streptomycin in T75 in an atmosphere of 5% CO2: 95% air at 37°C. Cells from passage 1–4 were trypsinized in 0.25% trypsin containing 0.02% EDTA in phosphate-buffered saline (PBS) and cultured in 6-well flexible-bottom culture plates pre-coated with collagen type I in 3 ml of DMEM medium. When cells were 85–95% confluent (day 3–4), old medium was removed and replaced with 3 ml of fresh medium supplemented with 7.5 mM HEPES with 1% fetal calf serum (FCS) overnight. Myometrial cells prepared according to this protocol have been previously characterized by Pieber et al. (2001). After 16–18 h these were subjected to a static stretch of 6, 11 or 16% for 1 or 6 h using a Flexercell strain unit (Flexcell International Corp., McKeesport, PA).
Quantitative RT-PCR
Total RNA was extracted and purified from myometrial cells grown in 6-well flexible-bottom culture plates using an RNAeasy mini kit from Qiagen Ltd., Crawley, West Sussex RH10 9AX. After quantification 1.0 μg was reverse transcribed with oligo dT random primers using MuLV reverse transcriptase (Applied Biosystems Ltd., Warrington, Cheshire WA3 7PB). Primer sets for IL-8 and glyceraldehyde-3-phosphatedehydrogenase (GAPDH) were designed and obtained from Amersham Pharmacia Biotech, Amersham Place, Little Chalfont, Bucks HP7 9NA:These primer sets flanked intron/exon junctions and produced amplicons of the expected size. Assays were validated for all primer sets by confirming that single amplicons of appropriate size and sequence were generated according to predictions. Quantitative PCR was performed in the presence of SYBR Green (Roche Diagnostics Ltd., Lewes, West Sussex, UK), and amplicon yield was monitored during cycling in a LightCycler Sequence Detector (Roche Diagnostics Ltd.) that continually measures fluorescence caused by the binding of the dye to double-stranded DNA. Pre-PCR cycle was 7 min at 95°C followed by 35 cycles of 95°C for 10 s, 56–60°C for 10 s and 72°C for 10 s followed by final extension at 72°C for 1 min. The cycle at which the fluorescence reached a preset threshold (cycle threshold) was used for quantitative analyses. The cycle threshold in each assay was set at a level where the exponential increase in amplicon abundance was approximately parallel between all samples. All mRNA abundance data were expressed relative to the amount of the constitutively expressed GAPDH. Conventional PCR was performed using Ampli-Taq Gold DNA polymerase (Applied Biosystems Ltd.). Pre-PCR cycle was 10 min at 95°C followed by 35 cycles of 95°C for 1 min, 56–60°C for 1 min and 72°C for 1 min followed by final extension 72°C for 10 min.
IL-8 sense 5′-GGACAAGAGCCAGGAAGAAACC-3′
Antisense 5′-GGCATCTTCACTGATTCTTGGAT-3′
GAPDH Sense 5′-TGATGACATCAAGAAGGTGGTGAAG-3′
Antisense 5′-TCCTTGGAGGCCATGTGGGCCAT-3′
Enzyme-linked immunosorbent assay (ELISA)
At the end of the incubation experiments, 1 ml of medium was collected and immediately frozen at −80°C for future analysis by IL-8 ELISA (Flexia). The IL-8 ELISA had a sensitivity of 15 pg/ml; the inter- and intra-assay variation were 7.4% and 5.3%, respectively.
Promoter studies
Transfection
Transient transfection was used to study promoter activity using a 2.2 kb of the IL-8 promoter sequence cloned into luciferase reporter vectors (PGL2, Promega) (Elliott et al., 2001b). A cytomegalovirus-driven renilla expression vector was used to control well-to-well transfection efficiency. Cells were plated and grown to 80% confluence equating to approximately 107 cells per well. Transfast (Promega) transfection agent was used to transfect 2.5 μg plasmid DNA per well (2 μg reporter and 0.5 μg renilla vector). Cells were incubated with transfection agent mastermix for 1 h then flooded with DMEM medium. After 16 h the medium was changed to serum-free conditions and the cells stretched at 11% for 6 h. The medium was aspirated from the wells and the plates frozen at −20°C. Cells were lysed using Luclite reagent and luciferase activity measured by a Wallac Luminometer. The luciferase reaction was quenched with 10 mM EDTA and renilla activity was determined from the samples.
Statistical analysis
Kolmogorov–Smirnov and Shapiro-Wilk statistics were used to determine normality of each sample pair. The data from stretch of primary cells were not normally distributed and were analysed with a Wilcoxon Signed Ranks Test (non-parametric test for related samples) using Statistics Package for Social Sciences (SPSS) 10.0 and the data expressed as median and range. The ELISA and promoter data were normally distributed and were analysed with a Student's t-test and expressed as mean (±SEM). Differences were considered statistically significant at P<0.05.
Results
Effects of reproductive state upon IL-8 mRNA expression
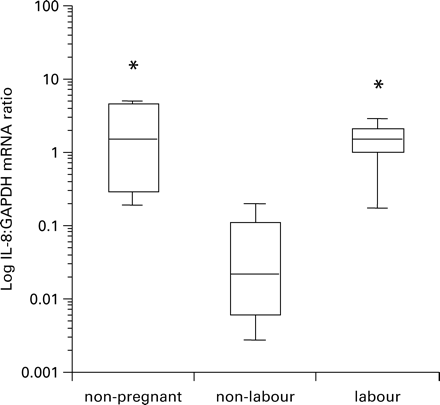
IL-8 mRNA expression was detected from myocytes in each of the patient groups. Expression was lowest in myocytes obtained from women at elective Caesarean section compared to IL-8 mRNA expression in myocytes from either non-pregnant women (P<0.05) or women in active labour (P<0.05; Figure 1, data shown in Table I).
Effects of stretch upon IL-8 mRNA and protein release
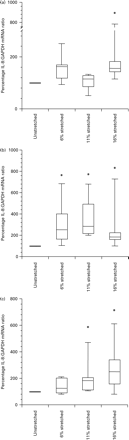
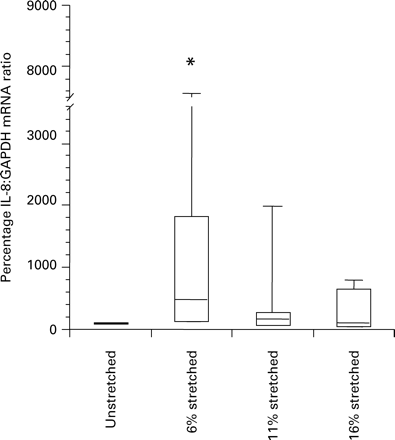
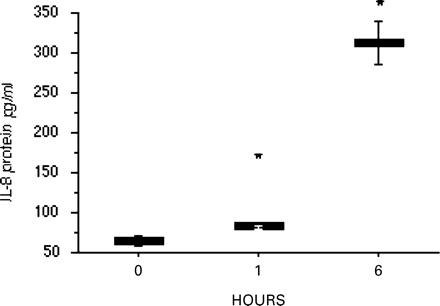
Myocytes from each patient group were exposed to static mechanical stretch for 1 h. In non-pregnant myocytes, IL-8 mRNA expression increased with stretch and was significantly increased at 16% stretch (P=0.028; Figure 2a). In myocytes from pregnant, non-labour women, 6, 11 and 16% stretch increased IL-8 mRNA expression (P=0.043, 0.028 and 0.043, respectively; Figure 2b). Stretch of myocytes grown from labouring women increased IL-8 mRNA expression at 11 and 16% (P=0.027 and 0.046, respectively; Figure 2c). In addition, myocytes from the pregnant, non-labour group were stretched for 6 h; IL-8 expression was increased at 6% stretch only (P=0.028; Figure 3). Concentrations of IL-8 protein increased in the supernatants of the stretched pregnant, non-labour myocytes at 1 and 6 h of 11% stretch (P=0.003 and 0.0008, respectively; Figure 4).
Effects of stretch upon IL-8 promoter activity
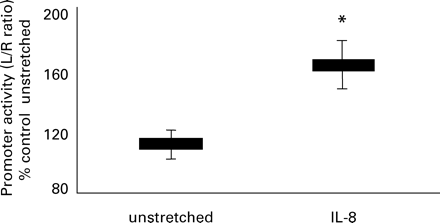
The full-length IL-8 promoter linked to a Luc reporter was transfected into myocytes collected before labour and the cells were exposed to 6 h of static stretch at 11%. Control experiments using empty plasmid showed no effect of stretch upon promoter activity (Figure 5). Promoter activity increased compared to non-stretched (P<0.05; Figure 5).
Discussion
We have shown that stretch of uterine myocytes results in increased IL-8 mRNA expression, synthesis and promoter activity. Thus, stretch not only increases myometrial contraction-associated protein expression, but also the synthesis of a potent pro-inflammatory factor that plays a key role in the preparation of the female reproductive tract for the onset of labour.
In our studies of IL-8 in different reproductive states, we found that IL-8 expression was lowest in myocytes derived from samples taken from pregnant women not in labour, intermediate in those from non-pregnant women and greatest in those from pregnant women in labour. The differences seen before and after the onset of labour are consistent with our previous work in which we demonstrated a significant increase in IL-8 mRNA levels in myometrial homogenates with the onset of labour (Elliott et al., 2000). Myocytes cultured from myometrial samples therefore maintain the expression profile and characteristics of myometrial tissue despite passage and cell culture conditions, making them a useful model to study the biochemistry of labour. The lower levels of IL-8 in myocytes derived from pregnant women before labour suggest that the pro-inflammatory process is repressed in the pregnant non-labouring myometrium but this repressive influence is lost at the time of labour allowing up-regulation of IL-8. We found a similar pattern for COX-2 and OTR (Sooranna et al., 2004; Terzidou et al., 2004). This may be due to progesterone since we have shown that progesterone suppresses IL-1β-induced IL-8 mRNA expression (Kelly et al., 1992; Loudon et al., 2003). Furthermore, the loss of this repressive effect after labour may be an example of ‘functional progesterone withdrawal’ that is suggested to occur before the onset of labour in humans.
Mechanical stretch increased IL-8 mRNA expression in uterine myocytes independent of reproductive state. The increases were greatest in the pregnant non-labour cells, which also had the lowest baseline expression of IL-8 mRNA. These data support the idea of a repression of IL-8 expression during pregnancy that is overcome by stretch, since the labour samples show a lesser degree of increase reflecting the fact that the repressive influence is lost or removed in labour and the baseline expression of IL-8 is therefore higher. These data are similar, but not identical to baseline levels of COX-2 expression, which showed a similar pattern and increase by stretch in all groups; however, the increase in COX-2 mRNA expression in response to stretch was greatest in the cells from labouring women (Sooranna et al., 2004). These data suggest that the regulation of IL-8 and COX-2 expression is not identical and that COX-2 expression may be specifically enhanced in labour. Indeed, we have found that although stretch-induced increases in the mRNA of both are dependent on ERK1/2 and p38 MAPK activation and the IL-1β-induced increases in COX-2 mRNA are similarly regulated, those of IL-8 are dependent on ERK1/2 activation alone (Sooranna et al., unpublished observation).
These data establish that stretch of uterine myocytes increases IL-8 mRNA expression and synthesis. The variations in IL-8 mRNA expression in different reproductive states support the concept of a suppression of contraction-associated proteins and of pro-inflammatory factors during pregnancy.
The effect of reproductive state upon the log of the IL-8:GAPDH mRNA ratio. Myocytes from samples collected before labour differed significantly from those collected after labour onset and also from samples from non-pregnant myometrium (* indicates P<0.05). The box plots represent median, interquartile range and range.
The effect of stretch for 1 h upon the IL-8:GAPDH mRNA ratio in non-pregnant (a), before labour onset (b) and after labour onset (c) cell preparations. In the non-pregnant preparations 16% stretch increased IL-8 mRNA in comparison to non-stretched control. Before labour all degrees of stretch significantly increased IL-8 mRNA in comparison to non-stretched control. After labour, 11 and 16% stretch significantly increased IL-8 mRNA in comparison to non-stretched control (*indicates P<0.05). The box plots represent median, interquartile range and range.
The effect of stretch for 6 h upon the IL-8:GAPDH mRNA ratio in non-pregnant cell preparations. 6% stretch increased IL-8 mRNA in comparison to non-stretched control (*indicates P<0.05). The box plots represent median, interquartile range and range.
The effect of stretch upon IL-8 protein release into the medium of stretched myocytes collected before labour. IL-8 protein release increased at 1 and 6 h of stretch at 11% (*indicates P<0.05). Data are shown as mean (±SEM).
The effect of stretch upon IL-8 promoter activity. Control experiments using empty vector showed no effect of stretch. 11% stretch for 6 h increased IL-8 promoter activity (*indicates P<0.05). Data are shown as mean (±SEM).
The IL-8/GAPDH ratio and GAPDH mRNA expression (copy number) in different reproductive states
| . | . | IL-8/GAPDH ratio (SEM) . | GAPDH (×106; SEM) . |
|---|---|---|---|
| Baseline | NP | 2.18 (0.9) | 114 (8) |
| NL | 0.06 (0.03) | 1076 (378) | |
| L | 1.55 (0.4) | 113 (34) |
| . | . | IL-8/GAPDH ratio (SEM) . | GAPDH (×106; SEM) . |
|---|---|---|---|
| Baseline | NP | 2.18 (0.9) | 114 (8) |
| NL | 0.06 (0.03) | 1076 (378) | |
| L | 1.55 (0.4) | 113 (34) |
NP, non-pregnant; NL, non-labour; L, labour.
The IL-8/GAPDH ratio and GAPDH mRNA expression (copy number) in different reproductive states
| . | . | IL-8/GAPDH ratio (SEM) . | GAPDH (×106; SEM) . |
|---|---|---|---|
| Baseline | NP | 2.18 (0.9) | 114 (8) |
| NL | 0.06 (0.03) | 1076 (378) | |
| L | 1.55 (0.4) | 113 (34) |
| . | . | IL-8/GAPDH ratio (SEM) . | GAPDH (×106; SEM) . |
|---|---|---|---|
| Baseline | NP | 2.18 (0.9) | 114 (8) |
| NL | 0.06 (0.03) | 1076 (378) | |
| L | 1.55 (0.4) | 113 (34) |
NP, non-pregnant; NL, non-labour; L, labour.
This study was funded by an Action Research Fellowship grant and a Wellbeing grant.
References
Chan EC, Fraser S, Yin S, Yeo G, Kwek K, Fairclough RJ and Smith R (
el Maradny E, Kanayama N, Halim A, Maehara K, Sumimoto K and Terao T (
Elliott CL, Slater DM, Dennes W, Poston L and Bennett PR (
Elliott CL, Allport VC, Loudon JA, Wu GD and Bennett PR (
Elliott CL, Loudon JA, Brown N, Slater DM, Bennett PR and Sullivan MH (
Kelly RW, Leask R and Calder AA (
Kumar A, Knox AJ and Boriek AM (
Li LF, Ouyang B, Choukroun G, Matyal R, Mascarenhas M, Jafari B, Bonventre JV, Force T and Quinn DA (
Loudon JA, Elliott CL, Hills F and Bennett PR (
Lyall F, Lye S, Teoh T, Cousins F, Milligan G and Robson S (
Maehara K, Kanayama N, Maradny EE, Uezato T, Fujita M and Terao T (
Manabe Y, Manabe A and Takahashi A (
Mitchell JA and Lye SJ (
Oldenhof AD, Shynlova OP, Liu M, Langille BL and Lye SJ (
Osmers R, Rath W, Adelmann-Grill BC, Fittkow C, Kuloczik M, Szeverenyi M, Tschesche H and Kuhn W (
Osmers RG, Adelmann-Grill BC, Rath W, Stuhlsatz HW, Tschesche H and Kuhn W (
Osmers RG, Blaser J, Kuhn W and Tschesche H (
Ou CW, Orsino A and Lye SJ (
Ou CW, Chen ZQ, Qi S and Lye SJ (
Ou CW, Chen ZQ, Qi S and Lye SJ (
Parry LJ, Bathgate RA, Shaw G, Renfree MB and Ivell R (
Pieber D, Allport VC, Hills F, Johnson M and Bennett PR (
Shynlova OP, Oldenhof AD, Liu M, Langille L and Lye SJ (
Sooranna SR, Lee Y, Ki LU, Mohan AR, Bennett PR and Johnson MR (
Terzidou V, Sooranna SR, Kim LU, Thornton S, Bennett PR and Johnson MR (
Wu WX, Ma XH, Yoshizato T, Shinozuka N and Nathanielsz PW (
Author notes
Imperial College Parturition Research Group 1Institute of Reproductive and Developmental Biology, Hammersmith Hospital Campus DuCane Road, London W12 0NN and 2Department of Maternal Fetal Medicine, Chelsea and Westminster Hospital, 369 Fulham Road, London SW10 9NH, UK