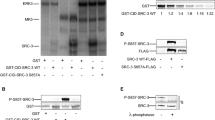
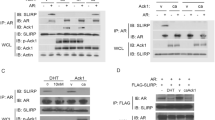
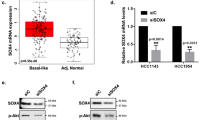
Abstract
Steroid receptor coactivator-3 (SRC-3, also known as NCoA3, AIB1, p/CIP, RAC3, ACTR, and TRAM1), localized on a frequently amplified region, 20q12, has been associated with multiple cancers, including breast, gastric and prostate cancers. Although SRC-3 has been implicated as an oncogene, compelling evidence has only recently emerged implicating it as a causal factor in the genesis of human cancers. Here, we summarize recent evidence that indicates aberrant SRC-3 expression is important in hormone-sensitive and -insensitive human cancers.
Similar content being viewed by others
Article PDF
References
Anzick SL, Kononen J, Walker RL, Azorsa DO, Tanner MM, Guan XY, et al. AIB1, a novel estrogen receptor co-activator amplified in breast and ovarian cancer. Science 1997; 277: 965–8.
Torchia J, Rose DW, Inostroza J, Kamei Y, Westin S, Glass CK, et al. The transcriptional coactivator p/CIP binds CBP and me diates nuclear receptor function. Nature 1997; 387: 677–84.
Li H, Gomes PJ, Chen JD . RAC3 a steroid/nuclear receptor-associated coactivator that is related to SRC-1 and TIF2. Proc Natl Acad Sci USA 1997; 94: 8479–84.
Chen H, Lin RJ, Schiltz RL, Chakravarti D, Nash A, Nagy L, et al. Nuclear receptor coactivator ACTR is a novel histone acetyl-transferase and forms a multimeric activation complex with p/ CAF and CBP/p300. Cell 1997; 90: 569–80.
Takeshita A, Cardona GR, Koibuchi N, Suen CS, Chin WW . TRAM-1, a novel 160 kDa thyroid hormone receptor activator molecule, exhibits distinct properties from steroid receptor coactivator-1. J Biol Chem 1997; 272: 27629–34.
Suen CS, Berrodin TJ, Mastroeni R, Cheskis BJ, Lyttle CR, Frail DE . A transcriptional coactivator, steroid receptor coactivator-3, selectively augments steroid receptor transcriptional activity. J Biol Chem 1998; 273: 27645–53.
Han SJ, Demayo FJ, Xu J, Tsai SY, Tsai MJ, O'Malley BW . Steroid receptor coactivators SRC-1 and SRC-3 differentially modulate tissue-specific activation functions of the progester one receptor. Mol Endocrinol 2006; 20: 45–55.
Ying H, Furuya F, Willingham MC, Xu J, O'Malley BW, Cheng SY . Dual functions of the steroid hormone receptor coactivator 3 in modulating resistance to thyroid hormone. Mol Cell Biol 2005; 25: 7687–95.
Lee SK, Kim HJ, Na SY, Kim TS, Choi HS, Im SY, et al. Steroid receptor coactivator-1 coactivates activating protein-1-mediated transactivations through interaction with the c-Jun and c-Fos subunits. J Biol Chem 1998; 273: 16651–4.
Werbajh S, Nojek I, Lanz R, Costas MA . RAC-3 is a NF-kappa B coactivator. FEBS Lett 2000; 485: 195–9.
Arimura A, Van Peer M, Schroder AJ, Rothman PB . The transcriptional co-activator p/CIP (NCoA-3) is up-regulated by STAT6 and serves as a positive regulator of transcriptional activation by STAT6. J Biol Chem 2004; 279: 31105–12.
Louie MC, Zou JX, Rabinovich A, Chen HW . ACTR/AIB1functions as an E2F1 coactivator to promote breast cancer cell proliferation and antiestrogen resistance. Mol Cell Biol 2004; 24: 5157–71.
Chen D, Ma H, Hong H, Koh SS, Huang SM, Schurter BT, et al. Regulation of transcription by a protein methyltransferase. Science 1999; 284: 2174–7.
McKenna NJ, O'Malley BW . Combinatorial control of gene expression by nuclear receptors and coregulators. Cell 2002; 108: 465–74.
Huang ZJ, Edery I, Rosbash M . PAS is a dimerization domain common to Drosophila period and several transcription factors. Nature 1993; 364: 259–62.
Belandia B, Parker MG . Functional interaction between the p160 coactivator proteins and the transcriptional enhancer factor family of transcription factors. J Biol Chem 2000; 275: 30801–5.
Chen SL, Dowhan DH, Hosking BM, Muscat GE . The steroid receptor coactivator, GRIP-1, is necessary for MEF-2C-dependent gene expression and skeletal muscle differentiation. Genes Dev 2000; 14: 1209–28.
Kim JH, Li H, Stallcup MR . CoCoA, a nuclear receptor coactivator which acts through an N-terminal activation domain of p160 coactivators. Mol Cell 2003; 12: 1537–49.
Shiau AK, Barstad D, Loria PM, Cheng L, Kushner PJ, Agard DA, et al. The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell 1998; 95: 927–37.
Brzozowski AM, Pike AC, Dauter Z, Hubbard RE, Bonn T, Engstrom O, et al. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature 1997; 389: 753–8.
Spencer TE, Jenster G, Burcin MM, Allis CD, Zhou J, Mizzen CA, et al. Steroid receptor coactivator-1 is a histone acetyltransferase. Nature 1997; 389: 194–8.
Chen D, Ma H, Hong H, Koh SS, Huang SM, Schurter BT, et al. Regulation of transcription by a protein methyltransferase. Science 1999; 284: 2174–7.
Koh SS, Chen D, Lee YH, Stallcup MR . Synergistic enhancement of nuclear receptor function by p160 coactivators and two coactivators with protein methyltransferase activities. J Biol Chem 2001; 276: 1089–98.
Gnanapragasam VJ, Leung HY, Pulimood AS, Neal DE, Robson CN . Expression of RAC 3, a steroid hormone receptor co-activator in prostate cancer. Br J Cancer 2001; 85: 1928–36.
Zhou HJ, Yan J, Luo W, Ayala G, Lin SH, Erdem H, et al. SRC-3 is required for prostate cancer cell proliferation and survival. Cancer Res 2005; 65: 7976–83.
Tanner MM, Grenman S, Koul A, Johannsson O, Meltzer P, Pejovic T, et al. Frequent amplification of chromosomal region 20q12-q13 in ovarian cancer. Clin Cancer Res 2000; 6: 1833–9.
Carroll RS, Brown M, Zhang J, DiRenzo J, De Mora JF, Black PM . Expression of a subset of steroid receptor cofactors is associated with progesterone receptor expression in meningiomas. Clin Cancer Res 2000; 6: 3570–5.
List HJ, Reiter R, Singh B, Wellstein A, Riegel AT . Expression of the nuclear coactivator AIB1 in normal and malignant breast tissue. Breast Cancer Res Treat 2001; 68: 21–8.
Bouras T, Southey MC, Venter DJ . Overexpression of the steroid receptor coactivator AIB1 in breast cancer correlates with the absence of estrogen and progesterone receptors and positivity for p53 and HER2/neu. Cancer Res 2001; 61: 903–7.
Osborne CK, Bardou V, Hopp TA, Chamness GC, Hilsenbeck SG, Fuqua SA, et al. Role of the estrogen receptor coactivator AIB1 (SRC-3) and HER-2/neu in tamoxifen resistance in breast cancer. J Natl Cancer Inst 2003; 95: 353–61.
Shou J, Massarweh S, Osborne CK, Wakeling AE, Ali S, Weiss H, et al. Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J Natl Cancer Inst 2004; 96: 926–35.
Reiter R, Wellstein A, Riegel AT . An isoform of the coactivator AIB1 that increases hormone and growth factor sensitivity is overexpressed in breast cancer. J Biol Chem 2001; 276: 39736–41.
Henke RT, Haddad BR, Kim SE, Rone JD, Mani A, Jessup JM, et al. Overexpression of the nuclear receptor coactivator AIB1 (SRC-3) during progression of pancreatic adenocarcinoma. Clin Cancer Res 2004; 10: 6134–42.
Ghadimi BM, Schrock E, Walker RL, Wangsa D, Jauho A, et al. Specific chromosomal aberrations and amplification of the AIB1 nuclear receptor coactivator gene in pancreatic carcinomas. Am J Pathol 1999; 154: 525–36.
Sakakura C, Hagiwara A, Yasuoka R, Fujita Y, Nakanishi M, Masuda K, et al. Amplification and over-expression of the AIB1 nuclear receptor co-activator gene in primary gastric cancers. Int J Cancer 2000; 89: 217–23.
Xie D, Sham JS, Zeng WF, Lin HL, Bi J, Che LH, et al. Correlation of AIB1 overexpression with advanced clinical stage of human colorectal carcinoma. Hum Pathol 2005; 36: 777–83.
Wang Y, Wu MC, Sham JS, Zhang W, Wu WQ, Guan XY . Prog nostic significance of c-myc and AIB1 amplification in hepatocellular carcinoma. A broad survey using high-throughput tissue microarray. Cancer 2002; 95: 2346–52.
Torres-Arzayus MI, De Mora JF, Yuan J, Vazquez F, Bronson R, Rue M, et al. High tumor incidence and activation of the PI3K/ AKT pathway in transgenic mice define AIB1 as an oncogene. Cancer Cell 2004; 6: 263–74.
Tilli MT, Reiter R, Oh AS, Henke RT, McDonnell K, Gallicano GI, et al. Overexpression of an N-terminally truncated isoform of the nuclear receptor coactivator amplified in breast cancer 1 leads to altered proliferation of mammary epithelial cells in transgenic mice. Mol Endocrinol 2005; 19: 644–56.
Xu J, Liao L, Ning G, Yoshida-Komiya H, Deng C, O'Malley BW . The steroid receptor coactivator SRC-3 (p/CIP/RAC3/AIB1/ ACTR/TRAM-1) is required for normal growth, puberty, female reproductive function, and mammary gland development. Proc Natl Acad Sci USA 2000; 97: 6379–84.
Wang Z, Rose DW, Hermanson O, Liu F, Herman T, Wu W, et al. Regulation of somatic growth by the p160 coactivator p/CIP. Proc Natl Acad Sci USA 2000; 97: 13549–54.
Kuang SQ, Liao L, Zhang H, Lee AV, O'Malley BW, Xu J . AIB1/ SRC-3 deficiency affects insulin-like growth factor I signaling pathway and suppresses v-Ha-ras-induced breast cancer initiation and progression in mice. Cancer Res 2004; 64: 1875–85.
Kuang SQ, Liao L, Wang S, Medina D, O'Malley BW, Xu J . Mice lacking the amplified in breast cancer 1/steroid receptor coactivator-3 are resistant to chemical carcinogen-induced mammary tumorigenesis. Cancer Res 2005; 65: 7993–8002.
Azorsa DO, Cunliffe HE, Meltzer PS . Association of steroid receptor coactivator AIB1 with estrogen receptor-alpha in breast cancer cells. Breast Cancer Res Treat 2001; 70: 89–101.
List HJ, Lauritsen KJ, Reiter R, Powers C, Wellstein A, Riegel AT . Ribozyme targeting demonstrates that the nuclear receptor coactivator AIB1 is a rate-limiting factor for estrogen-dependent growth of human MCF-7 breast cancer cells. J Biol Chem 2001; 276: 23763–8.
Shao W, Keeton EK, McDonnell DP, Brown M . Coactivator AIB1 links estrogen receptor transcriptional activity and stability. Proc Natl Acad Sci USA 2004; 101: 11599–604.
Planas-Silva MD, Shang Y, Donaher JL, Brown M, Weinberg RA . AIB1 enhances estrogen-dependent induction of cyclin D1 expression. Cancer Res 2001; 61: 3858–62.
Reiter R, Oh AS, Wellstein A, Riegel AT . Impact of the nuclear receptor coactivator AIB1 isoform AIB1-Delta3 on estrogenic ligands with different intrinsic activity. Oncogene 2004; 23: 403–9.
Harbour JW, Dean DC . The Rb/E2F pathway: expanding roles and emerging paradigms. Genes Dev 2000; 14: 2393–409.
Dimova DK, Dyson NJ . The E2F transcriptional network: old acquaintances with new faces. Oncogene 2005; 24: 2810–26.
Hunt KK, Keyomarsi K . Cyclin E as a prognostic and predictive marker in breast cancer. Semin Cancer Biol 2005; 15: 319–26.
Keyomarsi K, Tucker SL, Buchholz TA, Callister M, Ding Y, Hortobagyi GN, et al. Cyclin E and survival in patients with breast cancer. N Engl J Med 2002; 347: 1566–75.
Han S, Park K, Bae BN, Kim KH, Kim HJ, Kim YD, et al. E2F1 expression is related with the poor survival of lymph node-positive breast cancer patients treated with fluorouracil, doxorubicin and cyclophosphamide. Breast Cancer Res Treat 2003; 82: 11–6.
Dhillon NK, Mudryj M . Ectopic expression of cyclin E in estrogen responsive cells abrogates antiestrogen mediated growth arrest. Oncogene 2002; 21: 4626–34.
Grimberg A . Mechanisms by which IGF-I may promote cancer. Cancer Biol Ther 2003; 2: 630–5.
Oh A, List HJ, Reiter R, Mani A, Zhang Y, Gehan E, et al. The nuclear receptor coactivator AIB1 mediates insulin-like growth factor I-induced phenotypic changes in human breast cancer cells. Cancer Res 2004; 64: 8299–308.
Zhou G, Hashimoto Y, Kwak I, Tsai SY, Tsai MJ . Role of the steroid receptor coactivator SRC-3 in cell growth. Mol Cell Biol 2003; 23: 7742–55.
Lin A, Karin M . NF-κB in cancer: a marked target. Semin Cancer Biol 2003; 13: 107–14.
Werbajh S, Nojek I, Lanz R, Costas MA . RAC-3 is an NF-kappa B coactivator. FEBS Lett 2000; 485: 195–9.
Wu RC, Qin J, Hashimoto Y, Wong J, Xu J, Tsai SY, et al. Regulation of SRC-3 (pCIP/ACTR/AIB-1/RAC-3/TRAM-1) coactivator activity by I kappa B kinase. Mol Cell Biol 2002; 22: 3549–61.
Pages F, Vives V, Sautes-Fridman C, Fossiez F, Berger A, Cugnenc PH, et al. Control of tumor development by intratumoral cytokines. Immunol Lett 1999; 68: 135–9.
Giri D, Ozen M, Ittmann M . Interleukin-6 is an autocrine growth factor in human prostate cancer. Am J Pathol 2001; 159: 2159–65.
Shariat SF, Andrews B, Kattan MW, Kim J, Wheeler TM, Slawin KM . Plasma levels of interleukin-6 and its soluble receptor are associated with prostate cancer progression and metastasis. Urol ogy 2001; 58: 1008–15.
Wu RC, Qin J, Yi P, Wong J, Tsai SY, Tsai MJ, et al. Selective phosphorylations of the SRC-3/AIB1 coactivator integrate genomic responses to multiple cellular signaling pathways. Mol Cell 2004; 15: 937–49.
Wu RC, Smith CL, O'Malley BW . Transcriptional regulation by steroid receptor coactivator phosphorylation. Endocrine Rev 2005; 26: 393–9.
Raught B, Liao WSL, Rosen JM . Developmentally and hormonally regulated CCAAT/enhancer-binding protein isoforms influence beta-casein gene expression. Mol Endocrinol 1995; 9: 1223–32.
Kagan BL, Henke RT, Cabal-Manzano R, Stoica GE, Nguyen Q, Wellstein A, et al. Complex regulation of the fibroblast growth factor-binding protein in MDA- MB-468 breast cancer cells by CCAAT/enhancer-binding protein beta. Cancer Res 2003; 63: 1696–705.
Font de Mora J, Brown M . AIB1 is a conduit for kinase-mediated growth factor signaling to the estrogen receptor. Mol Cell Biol 2000; 20: 5041–7.
Goel A, Janknecht R . Concerted activation of ETS protein ER81 by p160 coactivators, the acetyltransferase p300 and the receptor tyrosine kinase HER2/Neu. J Biol Chem 2004; 279: 14909–16.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from the National Institutes of Health (U01DK62424) and Prostate SPORE (CA58204) to Ming-jer TSAT.
Rights and permissions
About this article
Cite this article
Yan, J., Tsai, S. & Tsai, Mj. SRC-3/AIB1: transcriptional coactivator in oncogenesis. Acta Pharmacol Sin 27, 387–394 (2006). https://doi.org/10.1111/j.1745-7254.2006.00315.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00315.x
Keywords
This article is cited by
-
SRC-3/TRAF4 facilitates ovarian cancer development by activating the PI3K/AKT signaling pathway
Medical Oncology (2023)
-
A genome-scale CRISPR Cas9 dropout screen identifies synthetically lethal targets in SRC-3 inhibited cancer cells
Communications Biology (2021)
-
Targeting NSD2-mediated SRC-3 liquid–liquid phase separation sensitizes bortezomib treatment in multiple myeloma
Nature Communications (2021)
-
Amplified in breast cancer 1 promotes colorectal cancer progression through enhancing notch signaling
Oncogene (2015)
-
RAC3 more than a nuclear receptor coactivator: a key inhibitor of senescence that is downregulated in aging
Cell Death & Disease (2015)