Abstract
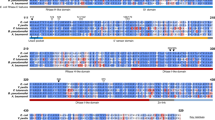
Most mycobacteria appear to be naturally resistant to β-lactam antibiotics such as penicillin. However, very few β-lactamases and their regulation have been clearly characterized in Mycobacterium tuberculosis H37Rv. In this study, a unique bifunctional protein, Rv2752c, from M. tuberculosis showed both β-lactamase and RNase activities. Two residues, D184 and H397, appear to be involved in Zn2+-binding and are essential for the dual functions. Both activities are lost upon deletion of the C-terminal 100 a.a. long Rv2752c tail, which contains an additional loop when compared with the RNase J of Bacillus subtilis. A chaperone-like protein, Rv2373c, physically interacted with Rv2752c and inhibited both activities. This is the first report of characterization of a bifunctional β-lactamase and its regulation in mycobacteria. These data offered important clues for further investigation of the structure and function of microbial β-lactamases. Increased understanding of this protein will provide further insights into the mechanism of microbial drug resistance.
Similar content being viewed by others
Abbreviations
- 3-AT:
-
3-amino-1,2,4-triazole
- GST:
-
glutathione-S-transferase
- Ni-NTA:
-
Ni2+-nitrilotriacetate
References
Fisher, J. F., Meroueh, S. O., and Mobashery, S. (2005) Chem. Rev., 105, 395–424.
Wilke, M. S., Lovering, A. L., and Strynadka, N. C. (2005) Curr. Opin. Microbiol., 8, 525–533.
Bush, K., Jacoby, G. A., and Medeiros, A. A. (1995) Antimicrob. Agents Chemother., 39, 1211–1233.
Carfi, A., Pares, S., Duee, E., Galleni, M., Duez, C., Frere, J. M., and Dideberg, O. (1995) EMBO J., 14, 4914–4921.
Orellano, E. G., Girardini, J. E., Cricco, J. A., Ceccarelli, E. A., and Vila, A. J. (1998) Biochemistry, 37, 10173–10180.
Fabiane, S. M., Sohi, M. K., Wan, T., Payne, D. J., Bateson, J. H., Mitchell, T., and Sutton, B. J. (1998) Biochemistry, 37, 12404–12411.
Garau, G., Bebrone, C., Anne, C., Galleni, M., Frere, J. M., and Dideberg, O. A. (2005) J. Mol. Biol., 345, 785–795.
Garrity, J. D., Carenbauer, A. L., Herron, L. R., and Crowder, M. W. (2004) J. Biol. Chem., 279, 920–927.
Wang, Z., and Benkovic, S. J. (1998) J. Biol. Chem., 273, 22402–22408.
De Seny, D., Heinz, U., Wommer, S., Kiefer, J. H., Meyer-Klaucke, W., Galleni, M., Frere, J., Bauer, R., and Adolph, H. W. (2001) J. Biol. Chem., 276, 45065–45078.
Rasia, R. M., and Vila, A. J. (2002) Biochemistry, 41, 1853–1860.
Bebrone, C., Delbruck, H., Kupper, M. B., Schlomer, P., Willmann, C., Frere, J. M., Fischer, R., Galleni, M., and Hoffmann, K. M. (2009) Antimicrob. Agents Chemother., 53, 4464–4471.
Fast, W., Wang, Z. G., and Benkovic, S. J. (2001) Biochemistry, 40, 1640–1650.
Hu, Z., Spadafora, L. J., Hajdin, C. E., Bennett, B., and Crowder, M. W. (2009) Biochemistry, 48, 2981–2999.
Even, S., Pellegrini, O., Zig, L., Labas, V., Vinh, J., and Putzer, H. (2005) Nucleic Acids Res., 33, 2141–2152.
Britton, R. A., Tingyi, W., Laura, S., Olivier, P., William, C. U., Nathalie, M., Crystal, T., Roula, D., and Ciaran, C. (2007) Mol. Microbiol., 63, 127–138.
Callebaut, I., Moshous, D., Mornon, J. P., and de Villartay, J. P. (2002) Nucleic Acids Res., 30, 3592–3601.
Nathalie, M., Lionel, B., Olivier, P., Roula, D., Tingyi, W., and Ciaran, C. (2007) Cell, 129, 681–692.
Sierra-Gallay, I., Zig, L., Jamalli, A., and Putzer, H. (2008) Nat. Struct. Mol. Biol., 15, 206–212.
Feng, W., Cassidy, C., and James, C. S. (2006) Antimicrob. Agents Chemother., 50, 2762–2771.
Nampoothiri, K. M., Rubex, R., Patel, A. K., Narayanan, S. S., Krishna, S., Das, S. M., and Pandey, A. (2008) J. Appl. Microbiol., 105, 59–67.
Tremblay, L. W., Hugonnet, J. E., and Blanchard, J. S. (2008) Biochemistry, 47, 5312–5316.
Cui, T., Zhang, L., Wang, X., and He, Z. G. (2009) BMC Genomics, 10, 118.
Gill, S. C., and von Hippel, P. H. (1989) Anal. Biochem., 182, 319–326.
Zhang, L., Zhang, L., Liu, Y., Yang, S., Gao, C., Gong, H., Feng, Y., and He, Z. G. (2009) Proc. Natl. Acad. Sci. USA, 106, 7792–7797.
Zeng, J., Zhang, L., Li, Y., Wang, Y., Wang, M., Duan, X., and He, Z. G. (2010) Protein Expr. Purif., 69, 47–53.
Arnold, K., Bordoli, L., Kopp, J., and Schwede, T. (2006) Bioinformatics, 22, 195–201.
Kolev, N. G., Yario, T. A., Benson, E., and Steitz, J. A. (2008) EMBO Rep., 9, 1013–1018.
Massova, I., and Mobashery, S. (1998) Antimicrob. Agents Chemother., 42, 1–17.
Nampoothiri, K. M. (2003) J. Biotechnol., 2, 342–345.
Stewart, G. R., Robertson, B. D., and Young, D. B. (2004) Tuberculosis, 84, 180–187.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2011, Vol. 76, No. 3, pp. 429–439.
Rights and permissions
About this article
Cite this article
Sun, L., Zhang, L., Zhang, H. et al. Characterization of a bifunctional β-lactamase/ribonuclease and its interaction with a chaperone-like protein in the pathogen Mycobacterium tuberculosis H37Rv. Biochemistry Moscow 76, 350–358 (2011). https://doi.org/10.1134/S0006297911030096
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297911030096