Abstract
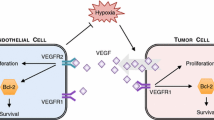
Vascular endothelial growth factor (VEGF) is a key mediator of angiogenesis, whose effect on cancer growth and development is well characterized. Alternative splicing of VEGF leads to several different isoforms, which are differentially expressed in various tumor types and have distinct functions in tumor blood vessel formation. Many cancer therapies aim to inhibit angiogenesis by targeting VEGF and preventing intracellular signaling leading to tumor vascularization; however, the effects of targeting specific VEGF isoforms have received little attention in the clinical setting. In this work, we investigate the effects of selectively targeting a single VEGF isoform, as compared with inhibiting all isoforms. We utilize a molecular-detailed whole-body compartment model of VEGF transport and kinetics in the presence of breast tumor. The model includes two major VEGF isoforms, VEGF121 and VEGF165, receptors VEGFR1 and VEGFR2, and co-receptors Neuropilin-1 and Neuropilin-2. We utilize the model to predict the concentrations of free VEGF, the number of VEGF/VEGFR2 complexes (considered to be pro-angiogenic), and the receptor occupancy profiles following inhibition of VEGF using isoform-specific anti-VEGF agents. We predict that targeting VEGF121 leads to a 54% and 84% reduction in free VEGF in tumors that secrete both VEGF isoforms or tumors that overexpress VEGF121, respectively. Additionally, 21% of the VEGFR2 molecules in the blood are ligated following inhibition of VEGF121, compared with 88% when both isoforms are targeted. Targeting VEGF121 reduces tumor free VEGF and is an effective treatment strategy. Our results provide a basis for clinical investigation of isoform-specific anti-VEGF agents.






Similar content being viewed by others
References
Rennel ES, Harper SJ, Bates DO. Therapeutic potential of manipulating VEGF splice isoforms in oncology. Futur Oncol. 2009;5(5):703–12.
Bates DO, Cui T-G, Doughty JM, Winkler M, Sugiono M, Shields JD, et al. VEGF165b, and inhibitory splice variant of vascular endothelial growth factor, is down-regulated in renal cell carcinoma. Cancer Res. 2002;62:4123–31.
Nowak DG, Woolard J, Amin EM, Konopatskaya O, Saleem MA, Churchill AJ, et al. Expression of pro- and anti-angiogenic isoforms of VEGF is differentially regulated by splicing and growth factors. J Cell Sci. 2008;121:3487–95.
Dokun AO, Annex BH. The VEGF165b "ICE-o-form" puts a chill on the VEGF story. Circul Res. 2011;109:246–7.
Manetti M, Guiducci S, Romano E, Ceccarelli C, Bellando-Randone S, Conforti ML, et al. Overexpression of VEGF165b, an inhibitory splice variant of vascular endothelial growth factor, leads to insufficient angiogenesis in patients with systemic sclerosis. Circul Res. 2011;109:e14–26.
Catena R, Larzabal L, Larrayoz M, Molina E, Hermida J, Agorreta J, et al. VEGF121b and VEGF165b are weakly angiogenic isoforms of VEGF-A. Mol Cancer. 2010;9:320.
Soker S, Miao H-Q, Nomi M, Takashima S, Klagsbrun M. VEGF165 mediates formation of complexes containing VEGFR-2 and neuropilin-1 that enhance VEGF165-receptor binding. J Cell Biochem. 2002;85(2):357–68.
Fuh G, Garcia KC, De Vos AM. The interaction of neuropilin-1 with vascular endothelial growth factor and its receptor flt-1. J Biol Chem. 2000;275:26690–5.
Park JE, Keller G-A, Ferrara N. The vascular endothelial growth factor (VEGF) isoforms: differential deposition into the subepithelial extracellular matrix and bioactivity of extracellular matrix-bound VEGF. Mol Biol Cell. 1993;4:1317–26.
Houck K, Leung DW, Rowland AM, Winer J, Ferrara N. Dual regulation of vascular endothelial growth factor bioavailability by genetic and proteolytic mechanisms. J Biol Chem. 1992;268(36):26031–7.
Grunstein J, Masbad JJ, Hickey R, Giordano F, Johnson RS. Isoforms of vascular endothelial growth factor act in a coordinate fashion to recruit and expand tumor vasculature. Mol Cell Biol. 2000;20(19):7292–1.
Tozer GM, Akerman S, Cross NA, Barber PR, Bjorndahl MA, Greco O, et al. Blood vessel maturation and response to vascular-disrupting therapy in single vascular endothelial growth factor-A isoform-producing tumors. Cancer Res. 2008;68:2301–11.
Yu JL, Rak JW, Klement G, Kerbel RS. Vascular endothelial growth factor isoform expression as a determinant of blood vessel patterning in human melanoma xenografts. Cancer Res. 2002;62:1838–46.
Yuan A, Yu C-J, Kuo S-H, Chen W-J, Lin F-Y, Luh K-T, et al. Vascular endothelial growth factor 189 mRNA isoform expression specifically correlates with tumor angiogenesis, patient survival, and postoperative relapse in non-small-cell lung cancer. J Clin Oncol. 2001;19(2):432–41.
Cheng S-Y, Nagane M, Huang H-JS, Cavenee WK. Intracerebral tumor-associated hemorrhage caused by overexpression of the vascular endothelial growth factor isoforms VEGF121 and VEGF165 but not VEGF189. Proc Natl Acad Sci U S A. 1997;94(22):12081–7.
Yuan A, Lin C-Y, Chou C-H, Shih C-M, Chen C-Y, Cheng H-W, et al. Functional and structural characteristics of tumor angiogenesis in lung cancers overexpressing different VEGF isoforms assessed by DCE- and SSCE-MRI. PLoS One. 2011;6(1):e16062.
Tokunaga T, Oshika Y, Abe Y, Ozeki Y, Sadahiro S, Kijima H, et al. Vascular endothelial growth factor (VEGF) mRNA isoform expression pattern is correlated with liver metastasis and poor prognosis in colon cancer. Br J Cancer. 1998;77(6):998–1002.
Fenton BM, Paoni SF, Liu W, Cheng S-Y, Hu B, Ding I. Overexpression of VEGF121, but not VEGF165 or FGF-1, improves oxygenation in MCF-7 breast tumors. Br J Cancer. 2004;90(2):430–5.
Verhoeff JJC, Stalpers LJA, Claes A, Hovinga KE, Musters GD, Vandertop WP, et al. Tumour control by whole brain irradiation of anti-VEGF-treated mice bearing intracerebral glioma. Eur J Cancer. 2009;45(17):3074–80.
Ruckman J, Green LS, Beeson J, Waugh S, Gillette WL, Henninger DD, et al. 2′-Fluoropyrimidine RNA-based aptamers to the 165-amino acid form of vascular endothelial growth factor (VEGF165). Inhibition of receptor binding and VEGF-induced vascular permeability through interactions requiring the exon 7-encoded domain. J Biol Chem. 1998;273:20556–67.
Ng EWM, Shima DT, Calias P, Cunningham Jr ET, Guyer DR, Adamis AP. Pegaptanib, a targeted anti-VEGF aptamer for ocular vascular disease. Nat Rev Drug Discov. 2006;5:123–32.
Ishida S, Usui T, Yamashiro K, Kaji Y, Amano S, Ogura Y, et al. VEGF164-mediated inflammation is required for pathological, but not physiological, ischemia-induced retinal neovascularization. J Exp Med. 2003;198(3):483–9.
Arrell DK, Terzic A. Network systems biology for drug discovery. Clin Pharmacol Ther. 2010;8(1):120–5.
Laubenbacher R, Hower V, Jarrah A, Torti SV, Shulaev V, Mendes P, et al. A systems biology view of cancer. Biochimica et Biophysica Acta (BBA) - Reviews on Cancer. 2009;1796(2):129–39.
Finley SD, Engel-Stefanini MO, Imoukhuede PI, Popel AS. Pharmacokinetics and pharmacodynamics of VEGF-neutralizing antibodies. BMC Syst Biol. 2011;5:193.
Stefanini MO, Wu FTH, Mac Gabhann F, Popel AS. Increase of plasma VEGF after intravenous administration of bevacizumab is predicted by a pharmacokinetic model. Cancer Res. 2010;70(23):9886–94.
Padera TP, Kadambi A, Di Tomaso E, Carreira CM, Brown EB, Boucher Y, et al. Lymphatic metastasis in the absence of functional intratumor lymphatics. Science. 2002;296(5574):1883–6.
Leu AJ, Berk DA, Lymboussaki A, Alitalo K, Jain RK. Absence of functional lymphatics within a murine sarcoma: a molecular and functional evaluation. Cancer Res. 2000;60:4324–7.
Imoukhuede PI, Popel AS. Quantification and cell-to-cell variation of vascular endothelial growth factor receptors. Exp Cell Res. 2011;317(7):955–65.
Genentech, Inc. Avastin prescribing information [cited September2011]; Available from: http://www.avastin.com/avastin/hcp/overview/about/dosing/index.html.
Gordon MS, Margolin K, Talpaz M, Sledge Jr GW, Holmgren E, Benjamin R, et al. Phase I safety and pharmacokinetic study of recombinant human anti-vascular endothelial growth factor in patients with advanced cancer. J Clin Oncol. 2001;19(3):843–50.
Hsei V, DeGuzman GG, Nixon A, Gaudreault J. Complexation of VEGF with bevacizumab decreases VEGF clearance in rats. Pharm Res. 2002;19(11):1753–6.
Liang W-C, Wu X, Peale FV, Lee CV, Meng YG, Gutierrez J, et al. Cross-species vascular endothelial growth factor (VEGF)-blocking antibodies completely inhibit the growth of human tumor xenografts and measure the contribution of stromal VEGF. J Biol Chem. 2006;281:951–61.
Guo P, Xu L, Pan S, Brekken RA, Yang S-T, Whitaker GB, et al. Vascular endothelial growth factor isoforms display distinct activities in promoting tumor angiogenesis at different anatomic sites. Cancer Res. 2001;61:8569–77.
Catena R, Muniz-Medina V, Moralejo B, Javierre B, Best CJM, Emmert-Buck MR, et al. Increased expression of VEGF121/VEGF165-189 ratio results in a significant enhancement of human prostate tumor angiogenesis. Int J Cancer. 2007;120:2096–109.
Zhang H-T, Scot PAE, Morbidelli L, Peak S, Moore J, Turley H, et al. The 121 amino acid isoform of vascular endothelial growth factor is more strongly tumorigenic than other splice variants in vivo. Br J Cancer. 2000;83(1):63–8.
Stimpfl M, Tong D, Fasching B, Schuster E, Obermair A, Leodolter S, et al. Vascular endothelial growth factor splice variants and their prognostic value in breast and ovarian cancer. Clin Cancer Res. 2002;8(7):2253–9.
Yuan A, Yu CJ, Luh KT, Lin FY, Kuo SH, Yang PC. Quantification of VEGF mRNA expression in non-small cell lung cancer using a real-time quantitative reverse transcription-PCR assay and a comparison with quantitative competitive reverse transcription-PCR. Lab Invest. 2000;2000(80):11.
Cheung N, Wong MP, Yuen ST, Leung SY, Chung LP. Tissue-specific expression pattern of vascular endothelial growth factor isoforms in the malignant transformation of lung and colon. Hum Pathol. 1998;29(9):910–4.
Ljungberg B, Jacobsen J, Haggstrom-Rudolfssson S, Rasmuson T, Lindh G, Grankvist K. Tumor vascular endothelial growth factor (VEGF) mRNA in relation to serum VEGF protein levels and tumour progression in human renal cell carcinoma. Urol Res. 2003;31(5):335–40.
Zygalaki E, Tsaroucha EG, Kaklamanis L, Lianidou ES. Quantitative real-time reverse transcription-PCR study of the expression of vascular endothelial growth factor (VEGF) splice variants and VEGF receptors (VEGFR-1 and VEGFR-2) in non-small cell lung cancer. Clin Chem. 2007;53(8):1433–9.
Hata K, Watanabe Y, Nakai H, Hata T, Hoshiai H. Expression of the vascular endothelial growth factor (VEGF) gene in epithelial ovarian cancer: an approach to anti-VEGF therapy. Anticancer Res. 2011;31(2):731–7.
Wiig H, Tenstad O, Iversen PO, Kalluri R, Bjerkvig R. Interstitial fluid: the overlooked component of the tumor microenvironment? Fibrogenesis Tissue Repair. 2010;3:12.
Huang J, Moore J, Soffer S, Kim E, Rowe D, Manley CA, et al. Highly specific antiangiogenic therapy is effective in suppressing growth of experimental Wilms tumors. J Pediatr Surg. 2001;36:357–61.
Chidlow Jr JH, Glawe JD, Pattillo CB, Pardue S, Zhang S, Kevil CG. VEGF164 isoform specific regulation of T-cell-dependent experimental colitis in mice. Inflamm Bowel Dis. 2011;17:1501–12.
Sawada O, Kawamura H, Kakinoki M, Sawada T, Ohji M. Vascular endothelial growth factor in aqueous humor before and after intravitreal injection of bevacizumab in eyes with diabetic retinopathy. Arch Ophthalmol. 2007;125(10):1363–6.
Tian XJ, Wu J, Meng L, Dong ZW, Shou CC. Expression of VEGF121 in gastric carcinoma MGC803 cell line. World J Gastroenterol. 2000;6(2):281–3.
Guo P, Fang Q, Tao H-Q, Schafer CA, Fenton BM, Ding I, et al. Overexpression of vascular endothelial growth factor by MCF-7 breast cancer cells promotes estrogen-independent tumor growth in vivo. Cancer Res. 2003;63:4684–91.
Acknowledgments
The authors thank Gang Liu and Spyridon Stamatelos for helpful discussions. This work was supported by National Institutes of Health grant R01 CA138264 (ASP) and fellowship F32 CA154213 (SDF), and the UNCF/Merck Postdoctoral Fellowship (SDF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Finley, S.D., Popel, A.S. Predicting the Effects of Anti-angiogenic Agents Targeting Specific VEGF Isoforms. AAPS J 14, 500–509 (2012). https://doi.org/10.1208/s12248-012-9363-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-012-9363-4