Abstract
Background
Primary treatments for stage IV breast cancer are chemotherapy and radiation, with surgery usually reserved for tumor-related complications. We sought to determine whether surgical removal of the primary tumor provides a survival advantage for women with metastatic breast cancer.
Methods
We conducted a retrospective, population-based cohort study by using the 1988–2003 Surveillance, Epidemiology, and End Results (SEER) program data. By use of multivariate Cox regression models, overall survival in women with stage IV disease was compared between women who underwent surgical excision of their breast tumor with women who did not, controlling for potential confounding demographic, tumor- and treatment-related variables, and propensity scores (accounting for variables associated with the likelihood of having surgery).
Results
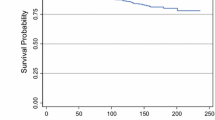
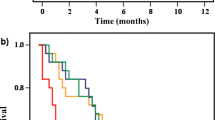
Of 9734 SEER patients with stage IV breast cancer, 47% underwent breast cancer surgery and 53% did not. Median survival was longer for women who had surgery than for women who did not, both among women who were alive at the end of the study period (36.00 vs. 21.00 months; P < .001) and among women who had died during follow-up (18.00 vs. 7.00 months; P < .001). After controlling for potential confounding variables and propensity scores, patients who underwent surgery were less likely to die during the study period compared with women who did not undergo surgery (adjusted hazard ratio, .63, 95% confidence interval, .60–.66).
Conclusions
Analysis of the 1988–2003 SEER data indicated that extirpation of the primary breast tumor in patients with stage IV disease was associated with a marked reduction in risk of dying after controlling for variables associated with survival.

Similar content being viewed by others
References
Falkson G, Holcroft C, Gelman RS, Tormey DC, Wolter JM, Cummings FJ. Ten-year follow-up study of premenopausal women with metastatic breast cancer: an Eastern Cooperative Oncology Group Study. J Clin Oncol 1995;13:1453–8
Hortobagyi GN. Treatment of breast cancer. N Engl J Med 1998;339:974–84
Bernard-Marty C, Cardoso F, Piccaart MJ. Facts and controversies in systemic treatment of metastatic breast cancer. Oncology 2004;9:617–32
Singletary SE, Walsh G, Vauthey JN, et al. A role for curative surgery in the treatment of selected patients with metastatic breast cancer. Oncologist 2003;8:241–51
National Cancer Institute. Breast cancer (PDQ). Treatment. Stage IIIB, inoperable IIIC, IV, recurrent, and metastatic breast cancer. Available at: http://www.cancer.gov/cancertopics/pdq/treatment/breast/HealthProfessional/page8. Accessed January 30, 2007
Khan SA, Stewart AK, Morrow M. Does aggressive local therapy improve survival in metastatic breast cancer? Surgery 2002;132:620–7
Babiera GV, Rao R, Feng L, et al. Effect of primary tumor extirpation in breast cancer patients who present with stage IV disease and an intact primary tumor. Ann Surg Oncol 2006;13:776–82
Rapiti E, Verkooijen HM, Vlastos G, et al. Complete excision of primary breast tumor improves survival of patients with metastatic breast cancer at diagnosis. J Clin Oncol 2006;24:2743–9
Griffiths CT, Fuller AR. Intensive surgical and chemotherapeutic management of advanced ovarian cancer. Symposium on Gynecologic Cancer. Surg Clin North Am 1978;58:131–42
Dauplat J, Le Bouedec G, Pomel C, Scherer C. Cytoreductive surgery for advanced stages of ovarian cancer. Semin Surg Oncol 2000;19:42–8
Chi DS, Eisenhauer EL, Lang J, et al. What is the optimal goal of primary cytoreductive surgery for bulky stage IIIC epithelial ovarian carcinoma (EOC)? Gynecol Oncol 2006;103:559–64
Griffiths CT, Parker LM, Lee S, Finkler NJ. The effect of residual mass size on response to chemotherapy after surgical cytoreduction for advanced ovarian cancer: long-term results. Int J Gynecol Cancer 2002;12:323–31
Goldie JH, Coldman AJ. A mathematical model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat Rep 1979;63:1727–33
Flanigan RC, Salmon SE, Blumenstein BA, et al. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal cell cancer. N Engl J Med 2001;345:1655–9
Rosen SA, Buell JF, Yoshida A, et al. Initial presentation with stage IV colorectal cancer: how aggressive should we be? Arch Surg 2000;135:530–4
Martin R, Paty P, Fong Y, et al. Simultaneous liver and colorectal resections are safe for synchronous colorectal liver metastasis. J Am Coll Surg 2003;197:233–41
Abdalla EK, Vauthey JN, Ellis LM, et al. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg 2004;239:818–25
Tanaka K, Shimada H, Matsuo K, et al. Outcome after simultaneous colorectal and hepatic resection for colorectal cancer with synchronous metastases. Surgery 2004;136:650–9
Hallissey MT, Allum WH, Roginski C, Fielding JWL. Palliative surgery for gastric cancer. Cancer 1988;62:440–4
National Cancer Institute. Surveillance, Epidemiology, and End Results. Overview of the SEER program. Available at: http://www.seer.cancer.gov/about/. Accessed February 1, 2007
Brookhart MA, Schneeweiss S, Rothman KJ, Glynn RJ, Avorn J, Sturmer T. Variable selection for propensity score models. Am J Epidemiol 2006;163:1149–56
Carmichael AR, Anderson EDC, Chetty U, Dixon JM. Does local surgery have a role in the management of stage IV breast cancer? Eur J Surg Oncol 2002;29:17–9
Cristofanilli M, Budd GT, Ellis MJ, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 2004;351:781–91
Retsky M, Bonadonna G, Demicheli R, Folkman J, Hrushesky W, Valagussa P. Hypothesis: induced angiogenesis after surgery in premenopausal node-positive breast cancer patients is a major underlying reason why adjuvant chemotherapy works particularly well for those patients. Breast Cancer Res 2004;6:R372–4
Retsky M, Demicheli R, Hrushesky W. Premenopausal status accelerates relapse in node positive breast cancer: hypothesis links angiogenesis, screening controversy. Breast Cancer Res Treat 2001;65:217–24
O’Reilly MS, Holmgren L, Shing Y, et al. Angiostatin: a novel angiogenesis inhibitor that mediates suppression of metastases by a Lewis lung carcinoma. Cell 1994;79:315–28
Danna EA, Sinha P, Gilbert M, et al. Surgical removal of the primary tumor reverses tumor-induced immunosuppression despite the presence of metastatic disease. Cancer Res 2004;64:2205–11
Dooley WC. A surgical indication in incurable breast cancer (editorial). Ann of Surg Oncol 2006;13:759–60
Esteva FJ, Valero V, Pusztai L, et al. Chemotherapy of metastatic breast cancer: what to expect in 2001 and beyond. Oncologist 2001;6:133–46
Acknowledgments
We thank the Alvin J. Siteman Cancer Center at Barnes-Jewish Hospital and Washington University School of Medicine in St. Louis, Missouri, for the use of the Health Behavior and Outreach Core, especially James Struthers and Mario Schootman, PhD, for data management and statistical services. The Siteman Cancer Center is supported in part by a National Cancer Institute Cancer Center Support Grant P30 CA91842.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gnerlich, J., Jeffe, D.B., Deshpande, A.D. et al. Surgical Removal of the Primary Tumor Increases Overall Survival in Patients With Metastatic Breast Cancer: Analysis of the 1988–2003 SEER Data. Ann Surg Oncol 14, 2187–2194 (2007). https://doi.org/10.1245/s10434-007-9438-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-007-9438-0