Abstract
Background
Alterations in HDACs gene expression have been reported in a number of human cancers. No information is available concerning the status of HDACs in pancreatic cancer tumors. The aim of the present study was to evaluate the expression levels of members of class I (HDAC1, 2,, 3), class II (HDAC4, 5, 6, and 7), and class III (SIRT1, 2, 3, 4, 5, and 6) in a set of surgically resected pancreatic tissues.
Methods
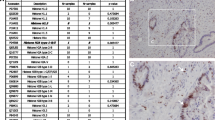
Total RNA was isolated from 11 pancreatic adenocarcinomas (PA): stage 0 (n = 1), IB (n = 1), IIB (n = 6), III (n = 1), IV (n = 2), one serous cystadenoma (SC), one intraductal papillary mucinous tumor of the pancreas (IMPN), one complicating chronic pancreatitis (CP), and normal pancreas (NP) obtained during donor liver transplantation. Moreover, six other control pancreatic were included. HDACs gene expression was conducted using quantitative real-time polymerase chain reaction (qPCR). Protein expression levels were analyzed by Western blot and their localization by immunohistochemistry analyses of cancer tissues sections.
Results
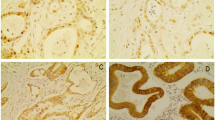
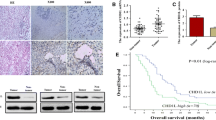
Remarkably, 9 of the 11 PA (approximately 81%) showed significant increase of HDAC7 mRNA levels. In contrast to PA samples, message for HDAC7 was reduced in CP, SC, and IMPN specimens. The Western blot analysis showed increased expression of HDAC7 protein in 9 out of 11 PA samples, in agreement with the qPCR data. Most of the PA tissue sections examined showed intense labeling in the cytoplasm when reacted against antibodies to HDAC7.
Conclusion
The data showed alteration of HDACs gene expression in pancreatic cancer. Increased expression of HDAC7 discriminates PA from other pancreatic tumors.




Similar content being viewed by others
References
Greenlee RT, Murray T, Bolden S, et al. Cancer statistics. Cancer J Clin 2000; 50:7–33.
Goggins M, Canto M, Hruban R. Can we screen high-risk individuals to detect early pancreatic carcinoma? J Surg Oncol 2000; 74:243–8.
Rosenberg L. Pancreatic cancer: a review of emerging therapies. Drugs 2000; 59:1071–89.
Gerdes B, Ramaswamy A, Ziegler A, et al. p16INK4a is a prognostic marker in resected ductal pancreatic cancer. Ann Surg 2002; 235:51–9.
Cress WD, Seto E. Histone deacetylase, transcriptional control and cancer. J Cell Physiol 2000; 184:1–16.
Marks PA. Histone deacetylase inhibitors as a new drugs. Curr Opin Oncol 2001; 13:477–83.
Timmermann S. Histone acetylation and disease. Cell Mol Life Sci 2001; 58:728–36.
Amunziato AT, Hansen JC. Role of histone acetylation in the assembly and modulation of chromatin structures. Gene Expr 2000; 9:37–61.
Bolden JE, Peart MJ, Johnstone RW. Anticancer activities of histone deacetylase inhibitors. Nat Rev Drug Discov 2006; 5:769–84.
Frye RA. Phylogenetic classification of prokaryotic and eukaryotic Sir2-like proteins. Biochem Biophys Res Commun 2000; 273:793–8.
De Nigris F, Cerutti J, Morelli C, et al. Isolation of a SIR-like gene, SIR-T8, that is overexpressed in thyroid carcinoma cell lines and tissues. Br J Cancer 2002; 86:917–23.
Greene FL, Page DL, Fleming ID, et al., eds. AJCC Cancer Staging Handbook, 6th edition. American Joint Committee on Cancer. New York: Springer Verlag, 2002
Corpet F. Multiple sequence alignment with hierarchical clustering. Nucl Acids Res 1988; 22:10881–90.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001; 25:402–8.
Avila AM, Burnett BG, Taye AA, et al. Trichostatin A increases SMN expression and survival in a mouse model of spinal muscular atrophy. J Clin Inv 2007; 117:659–71.
Shen S, Li J, Casaccia-Bonnefil P. Histone modifications affect timing of oligodendrocyte progenitor differentiation in the developing brain. J Cell Biol 2005; 169:577–88.
Gress TM, Muller-Pillasch F, Geng M, et al. A pancreatic cancer-specific expression profile. Oncogene 1996; 13:1819–30.
Crnogorac-Jurcevic T, Efthimiou E, Capelli P, et al. Gene expression profiles of pancreatic cancer and stromal desmoplasia. Oncogene 2001; 20:7437–46.
Sidransky D. Emerging molecular markers of cancer. Nat Rev Cancer 2002; 2:210–9.
Rosty C, Goggind M. Early detection of pancreatic carcinoma. Hematol Oncol Clin North Am 2002; 16:37–52.
Suzuki H, Gabrielson E, Chen W, et al. A genomic screen for genes upregulated by demethylation and histone deacetylase inhibition in human colorectal cancer. Nat Genet 2002; 31:141–9.
Iacobuzio-Donahue CA, Ashfaq A, Maitra A, et al. Highly expressed genes in pancreatic ductal adenocarcinomas: a comprehensive characterization and comparison of the transcription profiles obtained from three major technologies. Cancer Res 2003; 63:8614–22.
Bardeey N, DePinho RA. Pancreatic cancer biology and genetics. Nat Rev Cancer 2002; 2:897–909.
Crnogorac-Jurcevic T, Missiaglia E, Blaveri E, et al. Molecular alterations in pancreatic carcinoma expression profiling shows that dysregulated expression of S100 genes is highly prevalent. J Pathol 2003; 201:63–74.
Shekouh A, Thompson CC, Prime W, et al. Development and application of laser capture microdissection combined with two-dimensional electrophoresis for the discovery of differentially regulated proteins in pancreatic ductal adenocarcinoma. Proteomics 2003; 3:1988–2001.
Vimalachandran D, Greenhalf W, Thompson C, et al. High nuclear S100A6 (Calcyclin) is significantly associated with poor survival in pancreatic cancer patients. Cancer Res 2005; 65:3218–25.
Hruban RH, Iacobuzio-Donahue C, Wilentz RE, et al. Molecular pathology of pancreatic cancer. Cancer J 2001; 7:251–8.
Ebert M, Yokoyama M, Friess H, et al. Induction of platelet-derived growth factor A and B chains and over-expression of their receptors in human pancreatic cancer. Int J Cancer 1995; 62:529–35.
Friess H, Yamanaka Y, Büchler M. Enhanced expression of the type II transforming growth factor beta receptor in human pancreatic cancer cells without alteration of type III receptor expression. Cancer Res 1993; 53:2704–7.
Choi JH, Kwon HJ, Yoon BI, et al. Expression profile of histone deacetylase 1 in gastric cancer tissues. Jpn J Cancer Res 2001; 92:1300–4.
Zhu P, Martin E, Mengwasser J, Schlag P, et al. Induction of HDAC2 expression upon loss of APC in colorectal tumorogenesis. Cancer Cell 2004; 5:455–63.
Wilson AJ, Byun DS, Popova N, et al. Histone deacetylase 3 (HDAC3) and other class I HDACs regulate colon cell maturation and p21 expression and are deregulated in human colon cancer. J Biol Chem 2006; 281:13548–58.
Scanlan MJ, Welt S, Gordon CM, et al. Cancer-related serological recognition of human colon cancer: identification of potential diagnostic and immunotherapeutic targets. Cancer Res 2002; 62:4041–7.
Hiratsuka M, Inoue T, Toda T, et al. Proteomics-based identification of differentially expressed genes in human gliomas: down-regulation of SIRT2 gene. Biochem Biophys Res Commun 2003; 309:558–66.
Ouaissi M, Ouaissi A. Histone deacetylase enzymes as potential drug targets in cancer and parasitic diseases. J Biomed Biotechnol 2006; 13474:1–10.
Marks PA, Breslow R. Dimethyl sulfoxide to vorinostat: development of the histone deacetylase inhibitor as an anticancer drug. Nat Biotechnol 2007; 25:84–90.
Dokmanovic M, Perez G, Xu W, et al. Histone deacetylase inhibitors selectively suppress expression of HDAC7. Mol Cancer Ther 2007; 6:2525–34.
Mottet D, Bellahcène A, Pirotte S. HDAC7 silencing alters endothelial cell migration, a key step in angiogenesis. Cir Res 2007; 18:1794–801.
Dequiedt F, Kasler H, Fischle W, et al. HDAC7, a thymus-specific class II histone deacetylase, regulates Nur77 transcription and TCR-mediated apoptosis. Immunity 2003:18:687–98.
Kao HY, Verdel A, Tsai CC, et al. Mechanism for nucleocytoplasmic shuttling of histone deacetylase 7. J Biol Chem 2001; 276:47496–507.
Parra M, Kasler H, McKinsey TA, et al. Protein kinase D1 phosphorylates HDAC7 and induces its nuclear export after T-cell receptor activation. J Biol Chem 2005; 280:13762–70.
Sener S, Frengen A, Menck HR, et al. Pancreatic cancer: a report of treatment and survival trends for 100313 patients diagnosed from 1985–1995, using the National Cancer Database. J Am Coll Surg 1999; 189:1–7.
Acknowledgments
This work received support from INSERM. The authors wish to thank Ms Sylvie PICHET from the NOVARTIS laboratory for her support, and Ms Deborah Garcia for her efficient assistance in the organization of the reagents supply. The authors thank Dr. Laurent LAPLAZE for access to the DMRB microscope and images recording and Miss Joana TAVARES for the Western blot image analysis. We are indebted to Ms M. RADAL for her technical assistance in the preparation of cryosections of tissue samples. Dr. Leal Silvestre received a fellowship from the Fundação para a Ciência e Tecnologia (FCT) Portugal. Dr. Mehdi Ouaïssi was supported by Assistance Pubique-Hôpitaux de Marseille and University of Aix Marseille and has received a grant from the NOVARTIS Laboratory.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ouaïssi, M., Sielezneff, I., Silvestre, R. et al. High Histone Deacetylase 7 (HDAC7) Expression Is Significantly Associated with Adenocarcinomas of the Pancreas. Ann Surg Oncol 15, 2318–2328 (2008). https://doi.org/10.1245/s10434-008-9940-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-008-9940-z