Abstract
Background
Pancreatic cancer, a particularly deadly form of malignancy, has increased in the last decade worldwide. The purpose of this study is to identify markers for determining and identifying possible long-term survivors in cases of advanced pancreatic cancer.
Patients and methods
117 patients with pancreatic ductal carcinoma, including 89 with invasive tubular adenocarcinoma of the pancreas, Japan Pancreas Society (JPS) stage III–IVb patients, who underwent tumor resection between 1986 and 2006.
Results
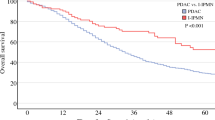
Univariate prognostic analyses of the 5-year disease-specific survival (DSS) revealed that JPS stage (P < 0.0001), preoperative serum carbohydrate antigen 19-9 (CA19-9) level (preCA19-9; P < 0.0001), dissected peripancreatic tissue margin (DPM; P < 0.0001), residual tumor (R factor; P = 0.0007), lymph node metastasis density over 10% (ND10; P = 0.006), volume of the stromal connective tissue (stroma factor; P = 0.008), growth pattern (P = 0.01), and histology (P = 0.03) were all significantly associated with poor outcome in advanced pancreatic cancer. Multivariate logistic analysis confirmed that preCA19-9 [P = 0.0006, relative risk (RR) = 2.16] and DPM (P = 0.04, RR = 1.62) were prognostic factors that remained, independent of JPS stage (P = 0.001). The higher preCA19-9 was, the worse the prognosis was. Astonishingly, among JPS stage III cases, 76.9% of the patients with preCA19-9 below 37 U/ml survived more than 5 years. This, combined with an analysis of DPM, allowed us to identify those with the potentiality for long-term survival.
Conclusion
Our results reveal for the first time that it is possible with JPS stage III–IVb invasive tubular adenocarcinomas of the pancreas to differentiate prognostic groups and potential survival rates, like with other cancers.




Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71-96. Epub 2008 Feb 20.
Matsuno S, Egawa S, Fukuyama S, et al. Pancreatic Cancer Registry in Japan: 20 years of experience. Pancreas. 2004;28(3):219-30.
Benassai G, Mastrorilli M, Quarto G, et al. Factors influencing survival after resection for ductal adenocarcinoma of the head of the pancreas. J Surg Oncol. 2000;73(4):212-8.
Ni XG, Bai XF, Mao YL, et al. The clinical value of serum CEA, CA19-9, and CA242 in the diagnosis and prognosis of pancreatic cancer. Eur J Surg Oncol. 2005;31(2):164-9.
Winter JM, Cameron JL, Campbell KA, et al. 1423 pancreaticoduodenectomies for pancreatic cancer: A single-institution experience. J Gastrointest Surg. 2006;10(9):1199-210; discussion 1210-1.
Smith RA, Bosonnet L, Ghaneh P, et al. Preoperative CA19-9 levels and lymph node ratio are independent predictors of survival in patients with resected pancreatic ductal adenocarcinoma. Dig Surg. 2008;25(3):226-232.
Kim J, Reber HA, Dry SM, et al. Unfavourable prognosis associated with K-ras gene mutation in pancreatic cancer surgical margins. Gut. 2006;55(11):1598-605. Epub 2006 May 8.
Vimalachandran D, Greenhalf W, Thompson C, et al. High nuclear S100A6 (Calcyclin) is significantly associated with poor survival in pancreatic cancer patients. Cancer Res. 2005;65(8):3218-25.
Tascilar M, Skinner HG, Rosty C, et al. The SMAD4 protein and prognosis of pancreatic ductal adenocarcinoma. Clin Cancer Res. 2001;7(12):4115-21.
Kawesha A, Ghaneh P, Andren-Sandberg A, et al. K-ras oncogene subtype mutations are associated with survival but not expression of p53, p16(INK4A), p21(WAF-1), cyclin D1, erbB-2 and erbB-3 in resected pancreatic ductal adenocarcinoma. Int J Cancer. 2000;89(6):469-74.
Nakao A, Fujii T, Sugimoto H, et al. Oncological problems in pancreatic cancer surgery. World J Gastroenterol. 2006;12(28):4466-72.
Nimura Y, Nagino M, Kato H, et al. Regional versus extended lymph node dissection in radical pancreatoduodenectomy for pancreatic cancer: a multicenter, randomized contorolled trial. Official J Int Hepatopancreatobiliary Assoc. 2004;6:(supplement I)2.
Japan Pancreas Society, ed. General rules for study of pancreatic cancer, April 2002 (5th ed). Tokyo: Kanahara; 2002.
Sobin LH, Wittekind CH, ed. International Union Againt Cancer (UICC): TNM classification of malignant tumors, 6th ed. New York: Wiley and Liss; 2002.
Slidell MB, Chang DC, Cameron JL, et al. Impact of total lymph node count and lymph node ratio on staging and survival after pancreatectomy for pancreatic adenocarcinoma: a large, population-based analysis. Ann Surg Oncol. 2008;15(1):165-74. Epub 2007 Sep 26.
Schwarz RE, Smith DD. Extent of lymph node retrieval and pancreatic cancer survival: information from a large US population database. Ann Surg Oncol. 2006;13(9):1189-200. Epub 2006 Sep 6.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53(282):457-481.
Cox DR. Regression models and life-tables. J R Stat Soc B (Methdological). 1972;34(2):187-220.
Sperti C, Pasquali C, Catalini S, et al. CA 19-9 as a prognostic index after resection for pancreatic cancer. J Surg Oncol. 1993;52(3):137-41.
Lundin J, Roberts PJ, Kuusela P, Haglund C. The prognostic value of preoperative serum levels of CA 19-9 and CEA in patients with pancreatic cancer. Br J Cancer. 1994;69(3):515-9.
Berger AC, Meszoely IM, Ross EA, Watson JC, Hoffman JP. Undetectable preoperative levels of serum CA 19-9 correlate with improved survival for patients with resectable pancreatic adenocarcinoma. Ann Surg Oncol. 2004;11(7):644-9. Epub 2004 Jun 14.
Ferrone CR, Finkelstein DM, Thayer SP, Muzikansky A, Fernandez-del Castillo C, Warshaw AL. Perioperative CA19-9 levels can predict stage and survival in patients with resectable pancreatic adenocarcinoma. J Clin Oncol. 2006;24(18):2897-902.
Zhang S, Wang YM, Sun CD, Lu Y, Wu LQ. Clinical value of serum CA19-9 levels in evaluating resectability of pancreatic carcinoma. World J Gastroenterol. 2008;14(23):3750-3.
Mandelker DL, Yamashita K, Tokumaru Y, et al. PGP95 promoter methylation is an independent prognostic factor for esophageal squamous cell carcinoma. Cancer Res. 2005;65(11):4963-8.
Yamashita K, Sakuramoto S, Kikuchi S, Katada N, Kobayashi N, Watanabe M. Surgical resection of stage IV gastric cancer and prognosis. Anticancer Res. 2007;27(6):4381-6.
Katoh H, Yamashita K, Kokuba Y, et al. Surgical resection of stage IV colorectal cancer and prognosis. World J Surg. 2008;32(6):1130-7.
Magnani JL. The discovery, biology, and drug development of sialyl Lea and sialyl Lex. Arch Biochem Biophys. 2004;426(2):122-31.
Yokoigawa N, Takeuchi N, Toda M, et al. Enhanced production of interleukin 6 in peripheral blood monocytes stimulated with mucins secreted into the bloodstream. Clin Cancer Res. 2005;11(17):6127-32.
Yokoigawa N, Takeuchi N, Toda M, et al. Overproduction of PGE2 in peripheral blood monocytes of gastrointestinal cancer patients with mucins in their bloodstream. Cancer Lett. 2007;245(1-2):149-55. Epub 2006 Feb 20.
Kannagi R, Izawa M, Koike T, Miyazaki K, Kimura N. Carbohydrate-mediated cell adhesion in cancer metastasis and angiogenesis. Cancer Sci. 2004;95(5):377-84.
Honn KV, Tang DG, Crissman JD. Platelets and cancer metastasis: a causal relationship? Cancer Metastasis Rev. 1992;11(3-4):325-51.
Matsumoto S. Cimetidine and survival with colorectal cancer. Lancet. 1995;346(8967):115.
Matsumoto S, Imaeda Y, Umemoto S, Kobayashi K, Suzuki H, Okamoto T. Cimetidine increases survival of colorectal cancer patients with high levels of sialyl Lewis-X and sialyl Lewis-A epitope expression on tumour cells. Br J Cancer. 2002;86(2):161-7.
Takai S, Satoi S, Toyokawa H, et al. Clinicopathologic evaluation after resection for ductal adenocarcinoma of the pancreas: a retrospective, single-institution experience. Pancreas. 2003;26(3):243-9.
Richter A, Niedergethmann M, Sturm JW, Lorenz D, Post S, Trede M. Long-term results of partial pancreaticoduodenectomy for ductal adenocarcinoma of the pancreatic head: 25-year experience. World J Surg. 2003;27(3):324-9. Epub 2003 Feb 27.
Wagner M, Redaelli C, Lietz M, Seiler CA, Friess H, Buchler MW. Curative resection is the single most important factor determining outcome in patients with pancreatic adenocarcinoma. Br J Surg. 2004;91(5):586-94.
Verbeke CS. Resection margins and R1 rates in pancreatic cancer–are we there yet? Histopathology. 2008;52(7):787-96. Epub 2007 Dec 13.
Raut CP, Tseng JF, Sun CC, et al. Impact of resection status on pattern of failure and survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. Ann Surg. 2007;246(1):52-60.
Esposito I, Kleeff J, Bergmann F, et al. Most pancreatic cancer resections are R1 resections. Ann Surg Oncol. 2008;15(6):1651-60. Epub 2008 Mar 20.
Takahashi S, Ogata Y, Tsuzuki T. Combined resection of the pancreas and portal vein for pancreatic cancer. Br J Surg. 1994;81(8):1190-3.
Nakao A, Takeda S, Sakai M, et al. Extended radical resection versus standard resection for pancreatic cancer: the rationale for extended radical resection. Pancreas. 2004;28(3):289-92.
Luttges J, Vogel I, Menke M, Henne-Bruns D, Kremer B, Kloppel G. The retroperitoneal resection margin and vessel involvement are important factors determining survival after pancreaticoduodenectomy for ductal adenocarcinoma of the head of the pancreas. Virchows Arch. 1998;433(3):237-42.
Westgaard A, Tafjord S, Farstad IN, et al. Resectable adenocarcinomas in the pancreatic head: the retroperitoneal resection margin is an independent prognostic factor. BMC Cancer. 2008;8:5.
Pedrazzoli S, DiCarlo V, Dionigi R, et al. Standard versus extended lymphadenectomy associated with pancreatoduodenectomy in the surgical treatment of adenocarcinoma of the head of the pancreas: a multicenter, prospective, randomized study. Lymphadenectomy Study Group. Ann Surg. 1998;228(4):508-17.
Yeo CJ, Cameron JL, Lillemoe KD, et al. Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma, part 2: randomized controlled trial evaluating survival, morbidity, and mortality. Ann Surg. 2002;236(3):355-66; discussion 366-8.
Capussotti L, Massucco P, Ribero D, Vigano L, Muratore A, Calgaro M. Extended lymphadenectomy and vein resection for pancreatic head cancer: outcomes and implications for therapy. Arch Surg. 2003;138(12):1316-22.
Farnell MB, Pearson RK, Sarr MG, et al. A prospective randomized trial comparing standard pancreatoduodenectomy with pancreatoduodenectomy with extended lymphadenectomy in resectable pancreatic head adenocarcinoma. Surgery. 2005;138(4):618-28; discussion 628-30.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Waraya, M., Yamashita, K., Katagiri, H. et al. Preoperative Serum CA19-9 and Dissected Peripancreatic Tissue Margin as Determiners of Long-Term Survival in Pancreatic Cancer. Ann Surg Oncol 16, 1231–1240 (2009). https://doi.org/10.1245/s10434-009-0415-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-009-0415-7