Abstract
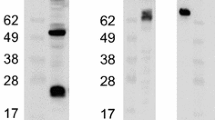
The envelope of the human immunodeficiency virus (HIV) is the main target for neutralizing antibodies. We report the cloning, purification, and characterization of two recombinant forms of the envelope glycoprotein gp125 from a primary HIV-2SBL-6669 isolate. Both constructs were truncated at the N- and C-termini, and in the gp125Δv1v2 construct the variable V1 and V2 loops were deleted. The recombinant glycoproteins were stably expressed in Chinese hamster ovarian cells, producing soluble gp125 and gp125Δv1v2 at molecular weights of 74.2 and 56.9 kDa, respectively, and were purified from cell culture supernatants in a single step using Galanthus nivalis lectin chromatography. Circular dichroism analysis indicated a similar secondary structure for gp125 and gp125Δv1v2, and both proteins were recognized by HIV-2 serum antibodies in surface plasmon resonance assays. The high yield and purity of these constructs makes them suitable for structural and functional analyses, as well as vaccine studies.
Article PDF
Similar content being viewed by others
References
Albert, J., Bredberg, U., Chiodi, F., et al. (1987) A new human retrovirus isolate of West African origin (SBL-6669) and its relationship to HTLV-IV, LAV-II, and HTLV-IIIB. AIDS Res. Hum. Retroviruses 3, 3–10.
Schim van der Loeff, M. and Aaby, P. (1999) Towards a better understanding of the epidemiology of HIV-2. AIDS 13, 69–84.
Kanki, P. J., Travers, K. U., Marlink, R. G., et al. (1994) Slower heterosexual spread of HIV-2 than HIV-1. Lancet 343, 943–946.
Marlink, R., Kanki, P., Thior, I., et al. (1994) Reduced rate of disease development after HIV-2 infection as compared to HIV-1. Science 265, 1587–1590.
Choe, H., Farzan, M., Sun, Y., et al. (1996) The B-chemokine receptors facilitate infection by primary HIV-1 isolates. Cell 85, 1135–1148.
McKnight, A., Dittmar, M. T., Moniz-Periera, J., et al. (1998) A broad range of chemokine receptors are used by primary isolates of human immunodeficiency virus type 2 as coreceptors with CD4. J. Virol. 72, 4065–4071.
Guyader, M., Emerman, M., Sonigo P., Clavel, F., Montagnier, L., and Alizon, M. (1987) Genome organization and transactivation of the human immunodeficiency virus type 2. Nature 326, 662–669.
Chakrabarti, L., Guyader, M., Alizon, M., et al. (1987) Sequence of simian immunodeficiency virus from macaque and its relationship to other human and simian retroviruses. Nature 328, 543–547.
Center, R. J., Leapman, R. D., Lebowitz, J., Arthur, L. O., Earl, P. L., and Moss, B. (2002) Oligomeric structure of the human immunodeficiency virus type 1 envelope protein on the virion surface. J. Virol. 76, 7863–7867.
Lu, M., Blacklow, S. C., and Kim, P. S. (1995) A trimeric structural domain of the HIV-1 transmembrane glycoprotein. Nat. Struct. Biol. 2, 1075–1082.
Modrow, S., Hahn, B. H., Shaw, G. M., Gallo, R. C., Wong-Staal, F., and Wolf, H. (1987) Computer-assisted analysis of envelope protein sequences of seven human immunodeficiency virus isolates: prediction of antigenic epitopes in conserved and variable regions. J. Virol. 61, 570–578.
Starcich, B. R., Hahn, B. H., Shaw, G. M., et al. (1986) Identification and characterization of conserved and variable regions in the envelope gene of HTLV-III/LAV, the retrovirus of AIDS. Cell 45, 637–648.
Willey, R. L., Rutledge, R. A., Dias, S., et al. (1986) Identification of conserved and divergent domains within the envelope gene of the acquired immunodeficiency syndrome retrovirus. Proc. Natl. Acad. Sci. USA 83, 5038–5042.
Kwong, P. D., Wyatt, R., Desjardins, E., et al. (1999) Probability analysis of variational crystallization and its application to gp120, the exterior envelope glycoprotein of type I human immunodeficiency virus (HIV-1). J. Biol. Chem. 274, 4115–4123.
Barnett, S. W., Lu, S., Srivastava, I., et al. (2001) The ability of an oligomeric human immunodeficiency virus type 1 (HIV-1) envelope antigen to elicit neutralizing antibodies against primary isolates is improved following partial deletion of the second hypervariable region. J. Virol. 75, 5526–5540.
Srivastava, I. K., Stamatatos, L., Kan, E., et al. (2003) Purification, characterization, and immunogenicity of a soluble trimeric envelope protein containing a partial deletion of the V2 loop derived from SF162, an R5-tropic human immunodeficiency virus type 1 isolate. J. Virol. 77, 11244–11259.
Weiss, R. A., Clapham, P. R., Weber, J. N., et al. (1988) HIV-2 antisera cross-neutralize HIV-1. AIDS 2, 95–100.
Bottiger, B., Karlsson, A., Andreasson, P. A., et al. (1990) Envelope cross-reactivity between human immunodeficiency virus types 1 and 2 detected by different serological methods: correlation between cross-neutralization and reactivity against the main neutralizing site. J. Virol. 64, 3492–3499.
Thali, M., Olshevsky, U., Furman, C., Gabuzda, D., Posner, M., and Sodroski, J. (1991). Characterization of a discontinuous human immunodeficiency virus type 1 gp120 epitope recognized by a broadly reactive neutralizing human monoclonal antibody. J. Virol. 65, 6188–6193.
Casasnovas, J. M. and Springer, T. A. (1995). Kinetics and thermodynamics of virus binding to receptor. Studies with rhinovirus, intercellular adhesion molecule-1 (ICAM-1), and surface plasmon resonance. J. Biol. Chem. 270, 13216–13224.
Takebe, Y., Seiki, M., Fujisawa, J., et al. (1988) SR alpha promoter: an efficient and versatile mammalian cDNA expression system composed of the simian virus 40 early promoter and the R-U5 segment of human T-cell leukemia virus type 1 long terminal repeat. Mol. Cell Biol. 8, 466–472.
Rothlein, R., Mainolfi, E. A., Czajkowski, M., and Marlin, S. D. (1991) A form of circulating ICAM-1 in human serum. J. Immunol. 147, 3788–3793.
Conley, A. J., Gorny, M. K., Kessler, J. A. II, et al. (1994) Neutralization of primary human immunodeficiency virus type 1 isolates by the broadly reactive anti-V3 monoclonal antibody, 447-52D. J. Virol. 68, 6994–7000.
Beirnaert, E., Nyambi, P., Willems, B., et al. (2000) Identification and characterization of sera from HIV-infected individuals with broad cross-neutralizing activity against group M (env clade A-H) and group O primary HIV-1 isolates. J. Med. Virol. 62, 14–24.
Björling, E., Scarlatti, G., von Gegerfelt, A., et al. (1993) Autologous neutralizing antibodies prevail in HIV-2 but not in HIV-1 infection. Virology 193, 528–530.
Thomas, E. R., Shotton, C., Weiss, R. A., Clapham, P. R., and McKnight, A. (2003). CD4-dependent and CD4-independent HIV-2: consequences for neutralization. AIDS 17, 291–300.
Zhang, J. Y., Jin, Y. N., Jiang, Z. W., Zhu, X. J., and Shen, J. C. (2001) Cloning and expression of the external-glycoprotein gene mutant from HIV-2 in the methylotrophic yeast Pichia pastoris and identification of the glycoprotein. Biotechnol. Appl. Biochem 34, 1–4.
Doyle, R. J. (1994). Introduction to lectins and their interactions with microorganisms. In: Lectin—Microorganisms Interactions (Doyle, R. J. and Slifkin, M., eds,), Marcel Dekker, New York, pp. 1–65.
Gilljam, G. (1993). Envelope glycoproteins of HIV-1, HIV-2, and SIV purified with Galanthus nivalis agglutinin induce strong immune response. AIDS Res. Hum. Retroviruses 9, 431–438.
Mascola, J. R., Snyder, S. W., Weislow, O. S., et al. (1996) Immunization with envelope subunit vaccine products elicits neutralizing against laboratory-adapted but not primary isolates of human immunodeficiency virus type 1. The National Institute of Allergy and Infectious Diseases AIDS Vaccine Evaluation Group. J. Infect. Dis. 173, 340–348.
Yang, X., Wyatt, R., and Sodroski, J. (2001) Improved elicitation of neutralizing antibodies against primary human immunodeficiency viruses by soluble stabilized envelope glycoprotein trimers. J. Virol. 75, 1165–1171.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sourial, S., Wärnmark, A., Nilsson, C. et al. Cloning, expression, and purification of HIV-2 gp125. Mol Biotechnol 30, 155–162 (2005). https://doi.org/10.1385/MB:30:2:155
Issue Date:
DOI: https://doi.org/10.1385/MB:30:2:155