Abstract
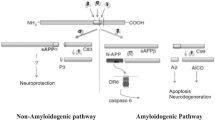
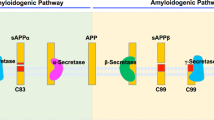
Accumulation of the amyloid-β (Aβ) peptide in the central nervous system (CNS) is considered by many to be the crucial pathological insult that ultimately leads to the development of Alzheimer’s disease (AD). Regulating the production and/or aggregation of Aβ could therefore be of considerable benefit to patients afflicted with AD. It has long been known that Aβ is derived from the proteolytic processing of the amyloid precursor protein (APP) by two enzymatic activities, β-secretase and γ-secretase. Recent breakthroughs have led to the identification of the aspartyl protease BACE (β-site APP-cleaving enzyme) as β-secretase and the probable identification of the presenilin proteins as γ-secretases. This review discusses what is know about BACE and the presenilins, focusing on their capacity as secretases, as well as the options for therapeutic advancement the careful characterization of these proteins will provide. These findings are presented in the context of the “amyloid cascade hypothesis” and its physiological relevance in AD pathogenesis.
Similar content being viewed by others
References
Selkoe D. J. (1999) Translating cell biology into therapeutic advances in Alzheimer’s disease. Nature 399(Suppl.), A23-A31.
Glenner G. G. and Wong C. W. (1984) Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem. Biophys. Res. Comm. 120, 885–890.
Roher A. E., Lowenson J. D., Clarke S, Wolkow C, Wang R, Cotter R. J., et al. (1993) Structural alterations in the peptide backbone of β-amyloid core protein may account for its deposition and stability in Alzheimer’s disease. J. Biol. Chem. 268, 3072–3083.
Iwatsubo T, Odaka A, Suzuki N, Mizusawa H, Nukina N, and Ihara Y. (1994) Visualization of Aβ 42(43) and Aβ 40 in senile plaques with end-specific Aβ monoclonals: evidence that an initially deposited species is Aβ 42(43). Neuron 13, 45–53.
Barrow C. J. and Zagorski M. G. (1991) Solution structures of β peptide and its constituent fragments: relation to amyloid deposition. Science 253, 179–182.
Jarrett J. T., Berger E. P., and Lansbury P. T., Jr. (1993) The carboxy terminus of the β amyloid protein is critical for the seeding of amyloid formation: implications for the pathogenesis of Alzheimer’s disease. Biochemistry 32, 4693–4697.
Pike C. J., Overman M. J., and Cotman C. W. (1995) Amino-terminal deletions enhance aggregation of β-amyloid peptides in vitro. J. Biol. Chem. 270, 23,895–23,898.
Kang D. E., Soriano S, Frosch M. P., Collins T, Naruse S, Sisodia S. S., et al. (1999) Presenilin 1 facilitates the constitutive turnover of β-catenin: differential activity of Alzheimer’s disease-linked PS1 mutants in the β-cateninsignaling pathway. J. Neurosci. 19, 4229–4237.
Selkoe D. J. (1994) Cell biology of the amyloid β-protein precursor and the mechanism of Alzheimer’s disease. Ann. Rev. Cell Biol. 10, 373–403.
Schubert D, Jin L. W., Saitoh T, and Cole G. (1989) The regulation of amyloid β protein precursor secretion and its modulatory role in cell adhesion. Neuron 3, 689–694.
Klier F. G., Cole G, Stallcup W., and Schubert D. (1990) Amyloid β-protein precursor is associated with extracellular matrix. Brain Res. 515, 336–342.
Breen K. C., Bruce M., and Anderton B. H. (1991) β amyloid precursor protein mediates neuronal cell-cell and cell-surface adhesion. J. Neurosci. Res. 28, 90–100.
Chen M. and Yankner B. A. (1991) An antibody to β amyloid and the amyloid precursor protein inhibits cell-substratum adhesion in many mammalian cell types. Neurosci. Lett. 125, 223–226.
Small D. H., Nurcombe V., Reed G., Clarris H., Moir R., Beyreuther K, and Masters C. L. (1994) A heparin-binding domain in the amyloid protein precursor of Alzheimer’s disease is involved in the regulation of neurite outgrowth. J. Neurosci. 14, 2117–2127.
Milward E. A., Papadopoulos R., Fuller S. J., Moir R. D., Small D., Beyreuther K., and Masters C. L. (1992) The amyloid protein precursor of Alzheimer’s disease is a mediator of the effects of nerve growth factor on neurite outgrowth. Neuron 9, 129–137.
Qiu W. Q., Ferreira A., Miller C., Koo E. H., and Selkoe D. J. (1995) Cell-surface β-amyloid precursor protein stimulates neurite outgrowth of hippocampal neurons in an isoform-dependent manner. J. Neurosci. 15, 2157–2167.
Perez R. G., Zheng H., Van der Ploeg L. H., and Koo E. H. (1997) The β-amyloid precursor protein of Alzheimer’s disease enhances neuron viability and modulates neuronal polarity. J. Neurosci. 17, 9407–9414.
Mattson M. P., Cheng B., Culwell A. R., Esch F. S., Lieberburg I., and Rydel R. E. (1993) Evidence for excitoprotective and intraneuronal calcium-regulating roles for secreted forms of the β-amyloid precursor protein. Neuron 10, 243–254.
Saitoh T., Sundsmo M., Roch J. M., Kimura N, Cole G., Schubert D., et al. (1989) Secreted form of amyloid β protein precursor is involved in the growth regulation of fibroblasts. Cell 58, 615–622.
Pietrzik C. U., Hoffmann J., Stober K., Chen C. Y., Bauer C., Otero D. A., et al. (1998) From differentiation to proliferation: the secretory amyloid precursor protein as a local mediator of growth in thyroid epithelial cells. Proc. Natl. Acad. Sci. USA 95, 1770–1775.
Zheng H., Jiang M., Trumbauer M. E., Sirinathsinghji D. J., Hopkins R., Smith D. W., et al. (1995) β-Amyloid precursor protein-deficient mice show reactive gliosis and decreased locomotor activity. Cell 81, 525–531.
Oltersdorf T, Fritz L. C., Schenk D. B., Lieberburg I, Johnson-Wood K. L., Beattie E. C., et al. (1989) The secreted form of the Alzheimer’s amyloid precursor protein with the Kunitz domain in protease nexin-II. Nature 341, 144–147.
Van Nostrand W. E., Wagner S. L., Suzuki M., Choi B. H., Farrow J. S., Geddes J. W., et al. (1989) Protease nexin-II, a potent antichymotrypsin, shows identity to amyloid β-protein precursor. Nature 341, 546–549.
Sinha S., Dovey H. F., Seubert P., Ward P. J., Blacher R. W., Blaber M., et al. (1990) The protease inhibitory properties of the Alzheimer’s β-amyloid precursor protein. J. Biol. Chem. 265, 8983–8985.
Haass C., Schlossmacher M. G., Hung A. Y., Vigo-Pelfrey C., Mellon A., Ostaszewski B. L., et al. (1992) Amyloid β-peptide is produced by cultured cells during normal metabolism. Nature 359, 322–325.
Shoji M., Golde T. E., Ghiso J., Cheung T. T., Estus S., Shaffer L. M., et al. (1992) Production of the Alzheimer amyloid β protein by normal proteolytic processing. Science 258, 126–129.
Seubert P., Vigo-Pelfrey C., Esch F., Lee M., Dovey H., Davis D., et al. (1992) Isolation and quantification of soluble Alzheimer’s β-peptide from biological fluids. Nature 359, 325–327.
Haass C., Koo E. H., Mellon A., Hung A. Y., and Selkoe D. J. (1992) Targeting of cell-surface β-amyloid precursor protein to lysosomes: alternative processing into amyloid bearing fragments. Nature 357, 500–503.
Koo E. H. and Squazzo S. L. (1994) Evidence that production and release of amyloid β-protein involves the endocytic pathway. J. Biol. Chem. 269, 17,386–17,389.
Haass C., Lemere C. A., Capell A., Citron M., Seubert P., Schenk D., et al. (1995) The Swedish mutation causes early-onset Alzheimer’s disease by β-secretase cleavage within the secretory pathway. Nat. Med. 1, 1291–1296.
Thinakaran G., Teplow D. B., Siman R., Greenberg B., and Sisodia S. S. (1996) Metabolism of the “Swedish” amyloid precursor protein variant in neuro2a (N2a) cells. J. Biol. Chem. 271, 9390–9397.
Xu H., Sweeny D., Wang R., Thinakaran G., Lo A. C. Y., Sisodia S. S., et al. (1997) Generation of Alzheimer β-amyloid protein in the transgolgi network in the apparent absence of vescicle formation. Proc. Natl. Acad. Sci. USA 94, 3748–3752.
Chyung A. S. C., Greenberg B. D., Cook D. G., Doms R. W., and Lee V. M.-Y. (1997) Novel β-secretase cleavage of β-amyloid precursor protein in the endoplasmic reticulum/intermediate compartment of NT2N cells. J. Cell. Biol. 138, 671–680.
Cook D. G., Forman M. S., Sung J. C., Leight S., Kolson D. L., Iwatsubo T., et al. (1997) Alzheimer’s Aβ(1–42) is generated in the endoplasmic reticulum/intermediate compartment of NT2N cells. Nat. Med. 3, 1021–1023.
Hartmann T., Bieger S. C., Bruhl B., Tienari P. J., Ida N., Allsop D., et al. (1997) Distinct sites of intracellular production for Alzheimer’s disease Aβ40/42 amyloid peptides. Nat. Med. 3, 1016–1020.
Skovronsky D. M., Doms R. W., and Lee V. M.-Y. (1998) Detection of a novel intraneuronal pool of insoluble amyloid β protein that accumulates with time in culture. J. Cell. Biol. 141, 1031–1039.
Esch F. S., Keim P. S., Beattie E. C., Blacher R. W., Culwell A. R., Oltersdorf T., et al. (1990) Cleavage of amyloid β peptide during constitutive processing of its precursor. Science 248, 1122–1124.
Skovronsky D. M., Moore D. B., Milla M. E., Doms R. W., and Lee V. M.-Y. (2000) Protein kinase C-dependent α-secretase competes with β-secretase for cleavage of amyloid-β precursor protein in the trans-golgi network. J. Biol. Chem. 275, 2568–2575.
Buxbaum J. D., Liu K. N., Luo Y., Slack J. L., Stocking K. L., Peschon J. J., et al. (1998) Evidence that tumor necrosis factor α converting enzyme is involved in regulated α-secretase cleavage of the Alzheimer’s amyloid protein precursor. J. Biol. Chem. 273, 27,765–27,767.
Lammich S., Kojro E., Postina R., Gilbert S., Pfeiffer R., Jasionowski M., et al. (1999) Constitutive adn regulated α-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloproteinase. Proc. Natl. Acad. Sci. USA 96, 3922–3927.
Kang J., Lemaire H. G., Unterbeck A., Salbaum J. M., Masters C. L., Grzeschik K. H., et al. (1987) The precursor of Alzheimer’s disease amyloid A4 protein resembles a cell-surface receptor. Nature 325, 733–736.
Giaccone G., Tagliavini F., Linoli G., Bouras C., Frigerio L., Frangione B., and Bugiani O. (1989) Down patients: extracellular preamyloid deposits precede neuritic degeneration and senile plaques. Neurosci. Lett. 97, 232–238.
Mann D. M., and Esiri M. M. (1989) The pattern of acquisition of plaques and tangles in the brains of patients under 50 years of age with Down’s syndrome. J. Neurol. Sci. 89, 169–179.
Motte J. and Williams R. S. (1989) Age-related changes in the density and morphology of plaques and neurofibrillary tangles in Down syndrome brain. Acta Neuropathol. 77, 535–546.
Neve R. L., Finch E. A., and Dawes L. R. (1988) Expression of the Alzheimer amyloid precursor gene transcripts in the human brain. Neuron 1, 669–677.
Tilley L., Morgan K., and Kalsheker N. (1998) Genetic risk factors in Alzheimer’s disease. Mol. Pathol. 51, 293–304.
Hardy J. (1997) Amyloid, the presenilins and Alzheimer’s disease. Trends Neurosci. 20, 154–159.
Citron M., Oltersdorf T., Haass C., McConlogue L., Hung A. Y., Seubert P., et al. (1992) Mutation of the β-amyloid precursor protein in familial Alzheimer’s disease increases β-protein production. Nature 360, 672–674.
Chartier-Harlin M. C., Crawford F., Houlden H, Warren A., Hughes D., Fidani L., et al. (1991) Early-onset Alzheimer’s disease caused by mutations at codon 717 of the β-amyloid precursor protein gene. Nature 353, 844–846.
Goate A., Chartier-Harlin M. C., Mullan M., Brown J., Crawford F., Fidani L., et al. (1991) Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349, 704–706.
Murrell J., Farlow M., Ghetti B., and Benson M. D. (1991) A mutation in the amyloid precursor protein associated with hereditary Alzheimer’s disease. Science 254, 97–99.
Suzuki N., Cheung T. T., Cai X. D., Odaka A., Otvos L. Jr., Eckman C., et al. (1994) An increased percentage of long amyloid β protein secreted by familial amyloid β protein precursor (β APP717) mutants. Science 264, 1336–1340.
Hussain I, Powell D., Howlett D. R., Tew D. G., Meek T. D., Chapman C., et al. (1999) Identification of a novel aspartic protease (Asp 2) as β-secretase. Mol. Cell Neurosci. 14, 419–427.
Sinha S., Anderson J. P., Barbour R., Basi G. S., Caccavello R., Davis D., et al. (1999) Purification and cloning of amyloid precursor protein β-secretase from human brain. Nature 402, 537–540.
Vassar R., Bennett B. D., Babu-Khan S., Kahn S., Mendiaz E. A., Denis P., et al. (1999) β-Secretase cleavage of Alzheimer’s amyloid precursor protein by the transmembrane aspartic protease BACE. Science 286, 735–741.
Yan R., Bienkowski M. J., Shuck M. E., Miao H., Tory M. C., Pauley A. M., et al. (1999) Membrane-anchored aspartyl protease with Alzheimer’s disease β-secretase activity. Nature 402, 533–537.
Lin X., Koelsch G., Wu S., Downs D, Dashti A., and Tang J. (2000) Human aspartic protease memapsin 2 cleaves the β-secretase site of β-amyloid precursor protein. Proc. Natl. Acad. Sci. USA 97, 1456–1460.
Masters C. L., Simms G., Weinman N. A., Multhaup G., McDonald B. L., and Beyreuther K. (1985) Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc. Natl. Acad. Sci. USA 82, 4245–4249.
Masters C. L., Multhaup G., Simms G., Pottgiesser J., Martins R. N., and Beyreuther K. (1985) Neuronal origin of a cerebral amyloid: neurofibrillary tangles of Alzheimer’s disease contain the same protein as the amyloid plaque cores and blood vessels. EMBO J. 4, 2757–2763.
Guiroy D. C., Miyazaki M., Multhaup G., Fisher P., Garruto R. M., Beyreuther K., et al. (1987) Amyloid of neurofibrillary tangles of Guamanian parkinsonism-dementia and Alzheimer disease share identical amino acid sequence. Proc. Natl. Acad. Sci. USA 84, 2073–2077.
Gouras G. K., Xu H., Jovanovic J. N., Buxbaum J. D., Wang R., Greengard P., et al. (1998) Generation and regulation of β-amyloid peptide variants by neurons. J. Neurochem. 71, 1920–1925.
Citron M., Diehl T. S., Capell A., Haass C., Teplow D. B., and Selkoe D. J. (1996) Inhibition of amyloid β-protein production in neural cells by the serine protease inhibitor AEBSF. Neuron 17, 171–179.
Seubert P., Oltersdorf T., Lee M. G., Barbour R., Blomquist C., Davis D. L., et al. (1993) Secretion of β-amyloid precursor protein cleaved at the amino terminus of the β-amyloid peptide. Nature 361, 260–263.
Haass C., Hung A. Y., Schlossmacher M. G., Teplow D. B., and Selkoe D. J. (1993) β-Amyloid peptide and a 3-kDa fragment are derived by distinct cellular mechanisms. J. Biol. Chem. 268, 3021–3024.
Knops J., Suomensaari S., Lee M., McConlogue L., Seubert P., and Sinha S. (1995) Cell-type and amyloid precursor protein-type specific inhibition of Aβ release by bafilomycin A1, a selective inhibitor of vacuolar ATPases. J. Biol. Chem. 270, 2419–2422.
Citron M., Teplow D. B., and Selkoe D. J. (1995) Generation of amyloid β protein from its precursor is sequence specific. Neuron 14, 661–670.
Capell A., Steiner H., Willem M., Kaiser H., Meyer C., Walter J., et al. (2000) Maturation and pro-peptide cleavage of β-secretase. J. Biol. Chem. 275, 30,849–30854.
Huse J. T., Pijak D. S., Leslie G. J., Lee V. M.-Y., and Doms R. W. (2000) Maturation and endosomal targeting of β-site amyloid precursor protein-cleaving enzyme: the Alzheimer’s disease β-secretase. J. Biol. Chem. 275, 33,729–33,737.
Haniu M., Denis P., Young Y., Mendiaz E. A., Fuller J., Hui J., et al. (2000) Characterization of Alzheimer’s β-secretase protein BACE. J. Biol. Chem. 275, 21,099–21,106.
Bennett B. D., Denis P., Haniu M., Teplow D. B., Kahn S., Louis J. C., et al. (2000) A furin-like convertase mediates propeptide cleavage of BACE, the Alzheimer’s β-secretase. J. Biol. Chem. 275, 37,712–37,717.
Hong L., Koelsch G., Lin X., Wu S., Terzyan S., Ghosh A., et al. (2000) Structure of the protease domain of memapsin 2 (β-secretase) complexed with inhibitor. Science 290, 150–153.
Sauder J. M., Arthur J. W., and Dunbrack R. L. (2000) Modeling of substrate specificity of the Alzheimer’s disease amyloid precursor protein β-secretase. J. Mol. Biol. 300, 241–248.
Acquati F., Accarino M., Nucci C., Fumagalli P., Jovine L., Ottolenghi S., and Taramelli R. (2000) The gene encoding DRAP (BACE2), a glycosylated transmembrane protein of the aspartic protease family, maps to the down critical region. FEBS Lett. 468, 59–64.
Bennett B. D., Babu-Khan S., Loeloff R., Louis J.-C., Curran E., Citron M., and Vassar R. (2000) Expression analysis of BACE2 in brain and peripheral tissues. J. Biol. Chem. 275, 20,647–20,651.
Solans A., Estivill X., and de La Luna S. (2000) A new aspartyl protease on 21q22.3, BACE2, is highly similar to Alzheimer’s amyloid precursor protein β-secretase. Cytogenet. Cell Genet. 89, 177–184.
Farzan M., Schnitzler C. E., Vasilieva N., Leung D., and Choe H. (2000) BACE2, a β-secretase homolog, cleaves at the β site and within the amyloid-β region of the amyloid-β precursor protein. Proc. Natl. Acad. Sci. USA 97, 9712–9717.
Levy-Lahad E., Wasco W., Poorkaj P., Romano D. M., Oshima J., Pettingell W. H. et al. (1995) Candidate gene for the chromosome 1 familial Alzheimer’s disease locus. Science 269, 973–977.
Rogaev E. I., Sherrington R., Rogaeva E. A., Levesque G., Ikeda M., Liang Y., et al. (1995) Familial Alzheimer’s disease in kindreds with missense mutations in a gene on chromosome 1 related to the Alzheimer’s disease type 3 gene. Nature 376, 775–778.
Sherrington R., Rogaev E. I., Liang Y., Rogaeva E. A., Levesque G., Ikeda M., et al. (1995) Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375, 754–760.
Doan A., Thinakaran G., Borchelt D. R., Slunt H. H., Ratovitsky T., Podlisny M., et al. (1996) Protein topology of presenilin 1. Neuron 17, 1023–1030.
Li X. and Greenwald I. (1996) Membrane topology of the C. elegans SEL-12 presenilin. Neuron 17, 1015–1021.
De Strooper B., Beullens M., Contreras B., Levesque L., Craessaerts K., Cordell B., et al. (1997) Phosphorylation, subcellular localization, and membrane orientation of the Alzheimer’ disease-associated presenilins. J. Biol. Chem. 272, 3590–3598.
Dewji N. N. and Singer S. J. (1997) The seven-transmembrane spanning topography of the Alzheimer disease-related presenilin proteins in the plasma membranes of cultured cells. Proc. Natl. Acad. Sci. USA 94, 14,025–14,030.
Lehmann S., Chiesa R., and Harris D. A. (1997) Evidence for a six-transmembrane domain structure of presenilin 1. J. Biol. Chem. 272, 12,047–12,051.
Li X. and Greenwald I. (1998) Additional evidence for an eight-transmembrane-domain topology for Caenorhabditis elegans and human presenilins. Proc. Natl. Acad. Sci. USA 95, 7109–7114.
Thinakaran G., Borchelt D. R., Lee M. K., Slunt H. H., Spitzer L., Kim G., et al. (1996) Endoproteolysis of presenilin 1 and accumulation of processed derivatives in vivo. Neuron 17, 181–190.
Kim T.-W., Pettingell W. H., Hallmark O. G., Moir R. D., Wasco W., and Tanzi R. E. (1997) Endoproteolytic cleavage and proteosomal degradation of presenilin 2 in transfected cells. J. Biol. Chem. 272, 11,006–11,010.
Capell A., Grunberg J., Pesold B., Diehlmann A., Citron M., Nixon R., et al. (1998) The proteolytic fragments of the Alzheimer’s disease-associated presenilin-1 form heterodimers and occur as a 100–150-kDa molecular mass complex. J. Biol. Chem. 273, 3205–3211.
Yu G., Chen F., Levesque G., Nishimura M., Zhang D. M., Levesque L., et al. (1998) The presenilin 1 protein is a component of a high molecular weight intracellular complex that contains β-catenin. J. Biol. Chem. 273, 16,470–16,475.
Thinakaran G., Harris C. L., Ratovitski T., Davenport F., Slunt H. H., Price D. L., et al. (1997) Evidence that levels of presenilins (PS1 and PS2) are coordinately regulated by competition for limiting cellular factors. J. Biol. Chem. 272, 28,415–28,422.
Ratovitski T., Slunt H. H., Thinakaran G., Price D. L., Sisodia S. S., and Borchelt D. R. (1997) Endoproteolytic processing and stabilization of wild-type and mutant presenilin. J. Biol. Chem. 272, 24,536–24,541.
Steiner H., Capell A., Pesold B., Citron M., Kloetzel P. M., Selkoe D. J., et al. (1998) Expression of Alzheimer’s disease-associated presenilin-1 is controlled by proteolytic degradation and complex formation. J. Biol. Chem. 273, 32,322–32,331.
Cook D. G., Sung J. C., Golde T. E., Felsenstein K. M., Wojczyk B. S., Tanzi R. E., et al. (1996) Expression and analysis of presenilin 1 in a human neuronal system: localization in cell bodies and dendrites. Proc. Natl. Acad. Sci. USA 93, 9223–9228.
Kovacs D. M., Fausett H. J., Page K. J., Kim T., Moir R. D., Merriam DE, et al. W. (1996) Alzheimer-associated presenilins 1 and 2: neuronal expression in brain and localization to intracellular membranes in mammalian cells. Nat. Med. 2, 224–229.
Annaert W., Levesque L., Craessaerts K., Dierinck I., Snellings G., Westaway D., et al. (1999) Presenilin 1 controls γ-secretase processing of amyloid precursor protein in pre-Golgi compartments of hippocampal neurons. J. Cell. Biol. 147, 277–294.
Lah J. J. and Levey AI. (2000) Endogenous presenilin-1 targets to endocytic rather than biosynthetic compartments. Mol. Cell. Neurosci. 16, 111–126.
Li J., Xu M., Zhou H., Ma J., and Potter H. (1997) Alzheimer presenilins in the nuclear membrane, interphase kinetochores, and centrosomes suggest a role in chromosome segregation. Cell 90, 917–927.
Schwarzman A. L., Singh N., Tsiper M., Gregori L., Dranovsky A., Vitek M. P., et al. (1999) Endogenous presenilin 1 redistributes to the surface of lamellipodia upon adhesion of Jurkat cells to a collagen matrix. Proc. Natl. Acad. Sci. USA 96, 7932–7937.
Nowotny P., Gorski S. M., Han S. W., Philips K., Ray W. J., Nowotny V., et al. (2000) Posttranslational modification and plasma membrane localization of the Drosophila melanogaster presenilin. Mol. Cell. Neurosci. 15, 88–98.
Ray W. J., Yao M., Mumm J., Schroeter E. H., Saftig P., Wolfe M., et al. (1999) Cell surface presenilin-1 participates in the γ-secretase-like proteolysis of notch. J. Biol. Chem. 274, 36,801–36,807.
Levitan D. and Greenwald I. (1995) Facilitation of lin-12-mediated signalling by sel-12, a Caenorhabditis elegans S182 Alzheimer’s disease gene. Nature 377, 351–354.
Shen J., Bronson R. T., Chen D. F., Xia W., Selkoe D. J., and Tonegawa S. (1997) Skeletal and CNS defects in presenilin-1-deficient mice. Cell 89, 629–639.
Wong P. C., Zheng H., Chen H., Becher M. W., Sirinathsingh D. J. S., Trumbauer M. E., et al. (1997) Presenilin is required for Notch1 and Dll1 expression in the paraxial mesoderm. Nature 387, 288–292.
Swiatek P. J., Lindsell C. E., Franco del Amo F., Weinmaster G., and Gridley T. (1994) Notch1 is essential for postimplantation development in mice. Genes Dev. 8, 707–719.
Conlon R. A., Reaume A. G., and Rossant J. (1995) Notch1 is required for the coordinate segmentation of somites. Development 121, 1533–1545.
Donoviel D. B., Hadjantonakis A., Ikeda M., Zheng H., St. George-Hyslop P., and Bernstein A. (1999) Mice lacking both presenilin genes exhibit early embryonic patterning defects. Genes Dev. 13, 2801–2810.
Herreman A., Hartmann D., Annaert W., Saftig P., Craessaerts K., Serneels L., et al. (1999) Presenilin 2 deficiency causes a mild pulmonary phenotype and no changes in amyloid precursor protein processing but enhances the embryonic lethal phenotype of presenilin 1 deficiency. Proc. Natl. Acad. Sci. USA 96, 11,872–11,877.
Vito P., Lacana E., and D’Adamio L. (1996) Interfering with apoptosis: Ca2+-binding protein A1G-2 and Alzheimer’s disease gene ALG-3. Science 271, 521–525.
Vito P., Ghayur T., and D’Adamio L. (1997) Generation of anti-apoptotic presenilin-2 polypeptides by alternative transcription, proteolysis, and caspase-3 cleavage. J. Biol. Chem. 272, 28,315–28,320.
Deng G., Pike C. J., and Cotman C. W. (1996) Alzheimer-associated presenilin-2 confers increased sensitivity to apoptosis in PC12 cells. FEBS Lett. 397, 50–54.
Guo Q., Furukawa K., Sopher B. L., Pham D. G., Xie J., Robinson N., et al. (1996) Alzheimer’s PS-1 mutation perturbs calcium homoeostasis and sensitizes PC12 cells to death induced by amyloid β-peptide. Neuroreport 8, 379–383.
Wolozin B, Iwasaki K., Vito P., Ganjei J. K., Lacana E., Sunderland T., et al. (1996) Participation of presenilin 2 in apoptosis: enhanced basal activity conferred by an Alzheimer mutation. Science 274, 1710–1713.
Janicki S. and Monteiro M. J. (1997) Increased apoptosis arising from increased expression of the Alzheimer’s disease-associated presenilin-2 mutation (N1411). J. Cell. Biol. 139, 485–495.
De Strooper B., Saftig P., Craessaerts K., Vanderstichele H., Guhde G., Annaert W., et al. (1998) Deficiency of presenilin-1 inhibits the normal cleavage of amyloid precursor protein. Nature 391, 387–390.
Naruse S., Thinakaran G., Luo J., Kusiak J. W., Tomita T., Iwatsubo T., et al. (1998) Effects of PS1 deficiency on membrane protein trafficking in neurons. Neuron 21, 1213–1221.
Zhou J., Lyanage U., Medina M., Ho C., Simmons A. D., Lovett M., and Kosik K. S. (1997) Presenilin 1 interaction in the brain with a novel member of the Armadillo family. Neuroreport 8, 1489–1494.
Buxbaum J. D., Choi E.-K., Luo Y., Lilliehook C., Crowley A. C., Merriam D. E., and Wasco W. (1998) Calsenilin: a calcium-binding protein that interacts with the presenilins and regulates the levels of a presenilin fragment. Nat. Med. 4, 1177–1181.
Smine A., Xu X., Nishiyama K., Katada T., Gambett P., Yadav S. P., et al. (1998) Regulation of brain G-protein G0 by Alzheimer’s disease gen presenilin-1. J. Biol. Chem. 273, 16,281–16,288.
Takashima A., Murayama M., Murayama O., Kohno T., Honda T., Yasutake K., et al. (1998) Presenilin 1 associates with glycogen synthase kinase-3β and its substrate tau. Proc. Natl. Acad. Sci. USA 95, 9637–9641.
Zhang W., Han S. W., McKeel D. W., Goate A., and Wu J. Y. (1998) Interaction of presenilins with the filamin family of actin-binding proteins. J. Neurosci. 18, 914–922.
Georgakopoulos A., Marambaud P., Efthimiopoulos S., Shioi J., Cui W., Li H. C., et al. (1999) Presenilin-1 forms complexes with the cadherin/catenin cell-cell adhesion system and is recruited to intercellular and synaptic contacts. Mol. Cell 4, 893–902.
Imafuku I., Masaki T., Waragai M., Takeuchi S., Kawabata M., Hirai S.-I., et al. (1999) Presenilin 1 suppresses the function of c-Jun homodimers via interaction with QM/Jif-1. J. Cell. Biol. 147, 121–133.
Stabler S. M., Ostrowski L. L., Janicki S. M., and Monteiro M. J. (1999) A myristoylated calcium-binding protein that preferentially interacts with the Alzheimer’s disease presenilin 2 protein. J. Cell. Biol. 145, 1277–1292.
Borchelt D. R., Thinakaran G., Eckman C. B., Lee M. K., Davenport F., Ratovitsky T., et al. (1996) Familial Alzheimer’s disease-linked presenilin 1 variants elevate Aβ1-42/1-40 ratio in vitro and in vivo. Neuron 17, 1005–1013.
Duff K., Eckman C., Zehr C., Yu X., Prada C.-M., Perez-tur J., et al. (1996) Increased amyloid-β42(43) in brains of mice expressing mutant presenilin 1. Nature 383, 710–713.
Lemere C. A., Lopera F., Kosik K. S., Lendon C. L., Ossa J., Saido T., et al. (1996) The E280A presenilin 1 Alzheimer mutation produces increased Aβ42 deposition and severe cerebellar pathology. Nat. Med. 2, 1146–1150.
Scheuner D., Eckman C., Jensen M., Song X., Citron M., Suzuki N., et al. (1996) Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer’s disease in increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat. Med. 2, 864–870.
Borchelt D. R., Ratovitski T., van Lare J., Lee M. K., Gonzales V., Jenkins N. A., et al. (1997) Accelerated amyloid deposition in the brains of transgenic mice coexpressing mutant presenilin 1 and amyloid precursor proteins. Neuron 19, 939–945.
Citron M., Westaway D., Xia W., Carlson G., Diehl T., Levesque G., et al. (1997) Mutant presenilins of Alzheimer’s disease increase production of 42-residue amyloid β-protein in both transfected cells and transgenic mice. Nat. Med. 3, 67–72.
Tomita T., Murayama K., Saido T. C., Kume H., Shinozaki K., Tokuhiro S., et al. (1997) The presenilin 2 mutation (N141I) linked to familial Alzheimer disease (Volga German families) increases the secretion of amyloid β protein ending at the 42nd (or 43rd) residue. Proc. Natl. Acad. Sci. USA 94, 2025–2030.
Xia W., Zhang J., Kholodenko D., Citron M., Podlisny M. B., Teplow D. B., et al. (1997) Enhanced production and oligomerization of the 42-residue amyloid β-protein by Chinese hamster ovary cells stably expressing mutant presenilins. J. Biol. Chem. 272, 7977–7982.
Sudoh S., Kawamura Y., Sato S., Wang R., Saido T. C., Oyama F., et al. (1998) Presenilin 1 mutations linked to familial Alzheimer’s disease increase the intracellular levels of amyloid β-protein 1–42 and its N-terminally truncated variant(s) which are generated at distinct sites. J. Neurochem. 71, 1535–1543.
Nakano Y., Kondoh G., Kudo T., Imaizumi K., Kato M., Miyazaki J.-I., et al. (1999) Accumulation of murine amyloidβ42 in a gene-dosage-dependent manner in PS1 ‘knock-in’ mice. Eur. J. Neurosci. 11, 2577–2581.
Davis J. A., Naruse S., Chen H., Eckman C., Younkin S., Price D. L., et al. (1998) An Alzheimer’s disease-linked PS1 variant rescues the developmental abnormalities of PS1-deficient embryos. Neuron 20, 603–609.
Qian S., Jiang P., Guan X.-M., Singh G., Trumbauer M. E., Yu H., et al. (1998) Mutant human presenilin 1 protects presenilin 1 null mouse against embryonic lethality and elevates Aβ1-42/43 expression. Neuron 20, 611–617.
Herreman A., Serneels L., Annaert W., Collen D., Schoonjans L., and De Strooper B. (2000) Total inactivation of γ-secretase activity in presenilin-deficient embryonic stem cells. Nat. Cell Biol. 2, 461–462.
Zhang Z., Nadeau P., Song W., Donoviel D., Yuan M., Bernstein A., and Yankner B. A. (2000) Presenilins are required for γ-secretase cleavage of β-APP and transmembrane cleavage of Notch. Nat. Cell Biol. 2, 463–465.
Wolfe M. S., Xia W., Ostaszewski B. L., Diehl T. S., Kimberly W. T., and Selkoe D. J. (1999) Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and γ-secretase activity. Nature 398, 513–517.
Kimberly W. T., Xia W., Rahmati T., Wolfe M. S., and Selkoe D. J. (2000) The transmembrane aspartates in presenilin 1 and 2 are obligatory for γ-secretase activity and amyloid β-protein generation. J. Biol. Chem. 275, 3173–3178.
Li Y.-M., Lai M.-T., Xu M., Huang Q., DiMuzio-Mower J., Sardana M. K., et al. (2000) Presenilin 1 is linked with γ-secretase activity in the detergent solubilized state. Proc. Natl. Acad. Sci. USA 97, 6138–6143.
Xia W., Ray W. J., Ostaszewski B. L., Rahmati T., Kimberley W. T., Wolfe M. S., et al. (2000) Presenilin complexes with the C-terminal fragments of amyloid precursor protein at the sites of amyloid β-protein generation. Proc. Natl. Acad. Sci. USA 97, 9299–9304.
Esler W. P., Kimberly W. T., Ostaszewski B. L., Diehl T. S., Moore C. L., Tsai J. Y., et al. (2000) Transition-state analogue inhibitors of γ-secretase bind directly to presenilin-1. Nat. Cell Biol. 2, 428–434.
Li Y. M., Xu M., Lai M. T., Huang Q., Castro J. L., DiMuzio-Mower J., et al. (2000) Photoactivated γ-secretase inhibitors directed to the active site covalently label presenilin 1. Nature 405, 689–694.
Kopan R., Schroeter E. H., Weintraub H., and Nye J. S. (1996) Signal transduction by activated mNotch: importance of proteolytic processing and its regulation by the extracellular domain. Proc. Natl. Acad. Sci. USA 93, 1683–1688.
Schroeter E. H., Kisslinger J. A., and Kopan R. (1998) Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain. Nature 393, 382–386.
Struhl G. and Adachi A. (1998) Nuclear access and action for notch in vivo. Cell 93, 649–660.
De Strooper B., Annaert W., Cupers P., Saftig P., Craessaerts K., Mumm J. S., et al. (1999) A presenilin-1-dependent γ-secretase-like protease mediates release of Notch intracellular domain. Nature 398, 518–522.
Song W., Nadeau P., Yuan M., Yang X., Shen J., and Yankner B. A. (1999) Proteolytic release and nuclear translocation of Notch-1 are induced by presenilin-1 and impaired by pathogenic presenilin-1 mutations. Proc. Natl. Acad. Sci. USA 96, 6959–6963.
Struhl G. and Greenwald I. (1999) Presenilin is required for activity and nuclear access of Notch in Drosophila. Nature 398, 522–525.
Kulic L., Walter J., Multhaup G., Teplau D. B., Baumeister R., Romig H., Capell A., et al. (2000) Separation of presenilin function in amyloid β-peptide generation and endoproteolysis of Notch. Proc. Natl. Acad. Sci. USA 97, 5913–5918.
Mori K. (2000) Tripartite management of unfolded proteins in the endoplasmic reticulum. Cell 101, 451–454.
Niwa M., Sidrauski C., Kaufman R. J., and Walter P. (1999) A role for presenilin-1 in nuclear accumulation of Ire1 fragments and induction of the mammalian unfolded protein response. Cell 99, 691–702.
Katayama T., Imaizumi K., Sato N., Miyoshi K., Kudo T., Hitomi J., et al. (1999) Presenilin-1 mutations downregulate the signalling pathway of the unfolded-protein response. Nat. Cell Biol. 1, 479–485.
Murphy M. P., Uljon S. N., Fraser P. E., Fauq A., Lookingbill H. A., Findlay K. A., et al. (2000) Presenilin 1 regulates pharmacologically distinct γ-secretase activities. Implications for the role of presenilin in γ-secretase cleavage. J. Biol. Chem. 275, 26,277–26,284.
Yu G., Chen F., Nishimura M., Steiner H., Tandon A., Kawarai T., et al. (2000) Mutation of conserved aspartates affects maturation of both aspartate mutant and endogenous presenilin 1 and presenilin 2 complexes. J. Biol. Chem. 275, 27,348–27,353.
Yu G., Nishimura M., Arawaka S., Levitan D., Zhang L., Tandon A., et al. (2000) Nicastrin modulates presenilin-mediated notch/glp-1 signal transduction and βAPP processing. Nature 407, 48–54.
Schenk D., Barbour R., Dunn W., Gordon G., Grajeda H., Guido T., et al. (1999) Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 400, 173–177.
Bard F., Cannon C., Barbour R., Burke R. L., Games D., Grajeda H., et al. (2000) Peripherally administered antibodies against amyloid β-peptide enter the central nervous system and reduce pathology in a mouse model of alzheimer disease. Nat. Med. 6, 916–919.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huse, J.T., Doms, R.W. Closing in on the amyloid cascade. Mol Neurobiol 22, 81–98 (2000). https://doi.org/10.1385/MN:22:1-3:081
Issue Date:
DOI: https://doi.org/10.1385/MN:22:1-3:081