Abstract
Multiple sclerosis (MS) is a chronic autoimmune disease of the CNS. Currently, six medications are approved for immunmodulatory and immunosuppressive treatment of the relapsing disease course and secondary-progressive MS. In the first part of this review, the pathogenesis of MS and its current treatment options are discussed.
During the last decade, our understanding of autoimmunity and the pathogenesis of MS has advanced substantially. This has led to the development of a number of compounds, several of which are currently undergoing clinical testing in phase II and III studies. While current treatment options are only available for parenteral administration, several oral compounds are now in clinical trials, including the immunosuppressive agents cladribine and laquinimod. A novel mode of action has been described for fingolimod, another orally available agent, which inhibits egress of activated lymphocytes from draining lymph nodes. Dimethylfumarate exhibits immunomodulatory as well as immunosuppressive activity when given orally. All of these compounds have successfully shown efficacy, at least in regards to the surrogate marker contrast-enhancing lesions on magnetic resonance imaging.
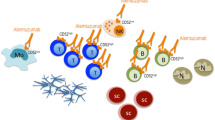
Another class of agents that is highlighted in this review are biological agents, namely monoclonal antibodies (mAb) and recombinant fusion proteins. The humanized mAb daclizumab inhibits T-lymphocyte activation via blockade of the interleukin-2 receptor. Alemtuzumab and rituximab deplete leukocytes and B cells, respectively; the fusion protein atacicept inhibits specific B-cell growth factors resulting in reductions in B-cells and plasma cells. These compounds are currently being tested in phase II and III studies in patients with relapsing MS.
The concept of neuro-protection and -regeneration has not advanced to a level where specific compounds have entered clinical testing. However, several agents approved for conditions other than MS are highlighted. Finally, with the advent of these highly potent novel therapies, rare, but potentially serious adverse effects have been noted, namely infections and malignancies. These are critically reviewed and put into perspective.


Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
French Research Group on Multiple Sclerosis. Multiple sclerosis in 54 twinships: concordance rate is independent of zygosity. Ann Neurol 1992; 32: 724–7
Noseworthy JH, Lucchinetti C, Rodriguez M, et al. Multiple sclerosis. N Engl J Med 2000; 343: 938–52
Frohman EM, Racke MK, Raine CS. Multiple sclerosis: the plaque and its pathogenesis. N Engl J Med 2006; 354: 942–55
Compston A, Coles A. Multiple sclerosis. Lancet 2002; 359: 1221–31
Weinshenker BG, Bass B, Rice GP, et al. The natural history of multiple sclerosis — a geographically based study: I, clinical course and disability. Brain 1989; 112(1): 133–46
Tullman MJ, Oshinsky RJ, Lublin FD, et al. Clinical characteristics of progressive relapsing multiple sclerosis. Mult Scler 2004; 10: 451–4
Sadovnick AD, Ebers GC, Dyment DA, et al. Evidence for genetic basis of multiple sclerosis. The Canadian Collaborative Study Group. Lancet 1996; 347: 1728–30
Dyment DA, Ebers GC, Sadovnick AD. Genetics of multiple sclerosis. Lancet Neurol 2004; 3: 104–10
Rosati G. The prevalence of multiple sclerosis in the world: an update. Neurol Sci 2001; 22: 117–39
Haines JL, Ter Minassian M, Bazyk A, et al. A complete genomic screen for multiple sclerosis underscores a role for the major histocompatability complex. The Multiple Sclerosis Genetics Group. Nat Genet 1996; 13: 469–71
Sawcer S, Jones HB, Feakes R, et al. A genome screen in multiple sclerosis reveals susceptibility loci on chromosome 6p21 and 17q 22. Nat Genet 1996; 13: 464–8
Ebers GC, Kukay K, Bulman DE, et al. A full genome search in multiple sclerosis. Nat Genet 1996; 13: 472–6
Kuokkanen S, Sundvall M, Terwilliger JD, et al. A putative vulnerability locus to multiple sclerosis maps to 5pl4-p l2 in a region syntenic to the murine locus Eae 2. Nat Genet 1996; 13: 477–80
Barcellos LF, Oksenberg JR, Begovich AB, et al. HLA-DR2 dose effect on susceptibility to multiple sclerosis and influence on disease course. Am J Hum Genet 2003; 72: 710–6
Villoslada P, Barcellos LF, Rio J, et al. The HLA locus and multiple sclerosis in Spain: role in disease susceptibility, clinical course and response to interferon-beta. J Neuroimmunol 2002; 130: 194–201
Hafler DA, Compston A, Sawcer S, et al. Risk alleles for multiple sclerosis identified by a genomewide study. N Engl J Med 2007; 357: 851–62
Gregory SG, Schmidt S, Seth P, et al. Interleukin 7 receptor alpha chain (IL7R) shows allelic and functional association with multiple sclerosis. Nat Genet 2007; 39: 1083–91
Giovannoni G, Cutter GR, Lunemann J, et al. Infectious causes of multiple sclerosis. Lancet Neurol 2006; 5: 887–94
Ascherio A, Munger KL. Environmental risk factors for multiple sclerosis: part I, the role of infection. Ann Neurol 2007; 61: 288–99
Lipton HL, Liang Z, Hertzler S, et al. A specific viral cause of multiple sclerosis: one virus, one disease. Ann Neurol 2007; 61: 514–23
Gardell JL, Dazin P, Islar J, et al. Apoptotic effects of Human Herpesvirus-6A on glia and neurons as potential triggers for central nervous system autoimmunity. J Clin Virol 2006; 37 Suppl. 1: S11–6
Sotelo J, Martinez-Palomo A, Ordonez G, et al. Varicella-zoster virus in cerebrospinal fluid at relapses of multiple sclerosis. Ann Neurol 2008; 63: 303–11
Merkler D, Horvath E, Brack W, et al. “Viral deja vu” elicits organ-specific immune disease independent of reactivity to self. J Clin Invest 2006; 116: 1254–63
McCoy L, Tsunoda I, Fujinami RS. Multiple sclerosis and virus induced immune responses: autoimmunity can be primed by molecular mimicry and augmented by bystander activation. Autoimmunity 2006; 39: 9–19
Flugel A, Berkowicz T, Ritter T, et al. Migratory activity and functional changes of green fluorescent effector cells before and during experimental autoimmune encephalomyelitis. Immunity 2001; 14: 547–60
Hickey WF. Basic principles of immunological surveillance of the normal central nervous system. Glia 2001; 36: 118–24
Gay FW, Drye TJ, Dick GW, et al. The application of multifactorial cluster analysis in the staging of plaques in early multiple sclerosis: identification and characterization of the primary demyelinating lesion. Brain 1997; 120: 1461–83
Ransohoff RM, Kivisakk P, Kidd G. Three or more routes for leukocyte migration into the central nervous system. Nat Rev Immunol 2003; 3: 569–81
Ulbrich H, Eriksson EE, Lindbom L. Leukocyte and endothelial cell adhesion molecules as targets for therapeutic interventions in inflammatory disease. Trends Pharmacol Sci 2003; 24: 640–7
Ubogu EE, Cossoy MB, Ransohoff RM. The expression and function of chemokines involved in CNS inflammation. Trends Pharmacol Sci 2006; 27: 48–55
Babbe H, Roers A, Waisman A, et al. Clonal expansions of CD8(+) T cells dominate the T cell infiltrate in active multiple sclerosis lesions as shown by micromanipulation and single cell polymerase chain reaction. J Exp Med 2000; 192: 393–404
Jacobsen M, Zhou D, Cepok S, et al. Clonal accumulation of activated CD8+ T cells in the central nervous system during the early phase of neuroborreliosis. J Infect Dis 2003; 187: 963–73
Sospedra M, Martin R. Immunology of multiple sclerosis. Annu Rev Immunol 2005; 23: 683–747
Cannella B, Raine CS. The adhesion molecule and cytokine profile of multiple sclerosis lesions. Ann Neurol 1995; 37: 424–35
Lock C, Hermans G, Pedotti R, et al. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat Med 2002; 8: 500–8
Tzartos JS, Friese MA, Craner MJ, et al. Interleukin-17 production in central nervous system-infiltrating T cells and glial cells is associated with active disease in multiple sclerosis. Am J Pathol 2008; 172: 146–55
Cannella B, Raine CS. Multiple sclerosis: cytokine receptors on oligodendrocytes predict innate regulation. Ann Neurol 2004; 55: 46–57
Bieber AJ, Kerr S, Rodriguez M. Efficient central nervous system remyelination requires T cells. Ann Neurol 2003; 53: 680–4
Hohlfeld R, Kerschensteiner M, Meinl E. Dual role of inflammation in CNS disease. Neurology 2007; 68: S58–63
Baranzini SE, Jeong MC, Butunoi C, et al. B cell repertoire diversity and clonal expansion in multiple sclerosis brain lesions. J Immunol 1999; 163: 5133–44
Qin Y, Duquette P, Zhang Y, et al. Clonal expansion and somatic hypermutation of V(H) genes of B cells from cerebrospinal fluid in multiple sclerosis. J Clin Invest 1998; 102: 1045–50
Colombo M, Dono M, Gazzola P, et al. Accumulation of clonally related B lymphocytes in the cerebrospinal fluid of multiple sclerosis patients. J Immunol 2000; 164: 2782–9
Owens GP, Ritchie AM, Burgoon MP, et al. Single-cell repertoire analysis demonstrates that clonal expansion is a prominent feature of the B cell response in multiple sclerosis cerebrospinal fluid. J Immunol 2003; 171: 2725–33
Prineas JW. Multiple sclerosis: presence of lymphatic capillaries and lymphoid tissue in the brain and spinal cord. Science 1979; 203: 1123–5
Magliozzi R, Howell O, Vora A, et al. Meningeal B-cell follicles in secondary progressive multiple sclerosis associate with early onset of disease and severe cortical pathology. Brain 2007; 130: 1089–104
Corcione A, Casazza S, Ferretti E, et al. Recapitulation of B cell differentiation in the central nervous system of patients with multiple sclerosis. Proc Natl Acad Sci U S A 2004; 101: 11064–9
Hemmer B, Archelos JJ, Hartung HP. New concepts in the immunopathogenesis of multiple sclerosis. Nat Rev Neurosci 2002; 3: 291–301
Cepok S, Rosche B, Grammel V, et al. Short-lived plasma blasts are the main B cell effector subset during the course of multiple sclerosis. Brain 2005; 128: 1667–76
Lucchinetti C, Brack W, Parisi J, et al. Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann Neurol 2000; 47: 707–17
Breij EC, Brink BP, Veerhuis R, et al. Homogeneity of active demyelinating lesions in established multiple sclerosis. Ann Neurol 2008; 63: 16–25
Lassmann H, Bruck W, Lucchinetti CF. The immunopathology of multiple sclerosis: an overview. Brain Pathol 2007; 17: 210–8
Charcot M. Histologie de la sclerose on plaque. Gaz Hop Paris 1868; 141: 554–5
Trapp BD, Peterson J, Ransohoff RM, et al. Axonal transection in the lesions of multiple sclerosis. N Engl J Med 1998; 338: 278–85
Kuhlmann T, Lingfeld G, Bitsch A, et al. Acute axonal damage in multiple sclerosis is most extensive in early disease stages and decreases over time. Brain 2002; 125: 2202–12
Bjartmar C, Trapp BD. Axonal and neuronal degeneration in multiple sclerosis: mechanisms and functional consequences. Curr Opin Neurol 2001; 14: 271–8
De Stefano N, Matthews PM, Fu L, et al. Axonal damage correlates with disability in patients with relapsing-remitting multiple sclerosis: results of a longitudinal magnetic resonance spectroscopy study. Brain 1998; 121(8): 1469–77
Dutta R, Trapp BD. Pathogenesis of axonal and neuronal damage in multiple sclerosis. Neurology 2007; 68: S22–31
Craner MJ, Newcombe J, Black JA, et al. Molecular changes in neurons in multiple sclerosis: altered axonal expression of Navl.2 and Navl.6 sodium channels and Na+/Ca2+ exchanger. Proc Natl Acad Sci U S A 2004; 101: 8168–73
Waxman SG. Ions, energy and axonal injury: towards a molecular neurology of multiple sclerosis. Trends Mol Med 2006; 12: 192–5
Barkhof F, Bruck W, De Groot CJ, et al. Remyelinated lesions in multiple sclerosis: magnetic resonance image appearance. Arch Neurol 2003; 60: 1073–81
Patrikios P, Stadelmann C, Kutzelnigg A, et al. Remyelination is extensive in a subset of multiple sclerosis patients. Brain 2006; 129: 3165–72
Scolding NJ, Franklin RJ. Remyelination in demyelinating disease. Baillieres Clin Neurol 1997; 6: 525–48
Chang A, Tourtellotte WW, Rudick R, et al. Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. N Engl J Med 2002; 346: 165–73
Franklin RJ. Why does remyelination fail in multiple sclerosis? Nat Rev Neurosci 2002; 3: 705–14
Comi G. Is it clinically relevant to repair focal multiple sclerosis lesions? J Neurol Sci 2008; 265: 17–20
Archelos JJ, Storch MK, Hartung HP. The role of B cells and autoantibodies in multiple sclerosis. Ann Neurol 2000; 47: 694–706
Archelos JJ, Trotter J, Previtali S, et al. Isolation and characterization of an oligodendrocyte precursor-derived B-cell epitope in multiple sclerosis. Ann Neurol 1998; 43: 15–24
McFarland HF, Barkhof F, Antel J, et al. The role of MRI as a surrogate outcome measure in multiple sclerosis. Mult Scler 2002; 8: 40–51
Berger T, Reindl M. Multiple sclerosis: disease biomarkers as indicated by pathophysiology. J Neurol Sci 2007; 259(1–2): 21–6
Gold R, Linington C, Lassmann H. Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalo-myelitis research. Brain 2006; 129: 1953–71
Stohlman SA, Hinton DR. Viral induced demyelination. Brain Pathol 2001; 11: 92–106
Yong VW, Giuliani F, Xue M, et al. Experimental models of neuroprotection relevant to multiple sclerosis. Neurology 2007; 68: S32–37
Lenercept Multiple Sclerosis Study Group. TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. Lenercept Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group. Neurology 1999; 53: 457–65
Bielekova B, Goodwin B, Richert N, et al. Encephalitogenic potential of the myelin basic protein peptide (amino acids 83–99) in multiple sclerosis: results of a phase II clinical trial with an altered peptide ligand. Nat Med 2000; 6: 1167–75
Huseby ES, Liggitt D, Brabb T, et al. A pathogenic role for myelin-specific CD8(+) T cells in a model for multiple sclerosis. J Exp Med 2001; 194: 669–76
Bettelli E, Baeten D, Jager A, et al. Myelin oligodendrocyte glycoprotein-specific T and B cells cooperate to induce a Devic-like disease in mice. J Clin Invest 2006; 116: 2393–402
Krishnamoorthy G, Lassmann H, Wekerle H, et al. Spontaneous opticospinal encephalomyelitis in a double-transgenic mouse model of autoimmune T cell/B cell cooperation. J Clin Invest 2006; 116: 2385–92
Brisebois M, Zehntner SP, Estrada J, et al. A pathogenic role for CD8+ T cells in a spontaneous model of demyelinating disease. J Immunol 2006; 177: 2403–11
Madsen LS, Andersson EC, Jansson L, et al. A humanized model for multiple sclerosis using HLA-DR2 and a human T-cell receptor. Nat Genet 1999; 23: 343–7
Friese MA, Jensen LT, Willcox N, et al. Humanized mouse models for organ-specific autoimmune diseases. Curr Opin Immunol 2006; 18: 704–9
Miller SD, Olson JK, Croxford JL. Multiple pathways to induction of virus-induced autoimmune demyelination: lessons from Theiler's virus infection. J Autoimmun 2001; 16: 219–27
Goodin DS, Frohman EM, Garmany Jr GP, et al. Disease modifying therapies in multiple sclerosis: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the MS Council for Clinical Practice Guidelines. Neurology 2002; 58: 169–78
Beck RW, Cleary PA, Trobe JD, et al. The effect of corticosteroids for acute optic neuritis on the subsequent development of multiple sclerosis. The Optic Neuritis Study Group. N Engl J Med 1993; 329: 1764–9
Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids: new mechanisms for old drags. N Engl J Med 2005; 353: 1711–23
Jacobs LD, Beck RW, Simon JH, et al. Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. CHAMPS Study Group. N Engl J Med 2000; 343: 898–904
Comi G, Filippi M, Barkhof F, et al. Effect of early interferon treatment on conversion to definite multiple sclerosis: a randomised study. Lancet 2001; 357: 1576–82
Kappos L, Polman CH, Freedman MS, et al. Treatment with interferon beta-1b delays conversion to clinically definite and McDonald MS in patients with clinically isolated syndromes. Neurology 2006; 67: 1242–9
IFNB Multiple Sclerosis Study Group. Interferon beta-1b is effective in relapsing-remitting multiple sclerosis: I, clinical results of a multicenter, randomized, double-blind, placebocontrolled trial. Neurology 1993; 43: 655–61
PRISMS. Randomised double-blind placebo-controlled study of interferon beta-la in relapsing/remitting multiple sclerosis. PRISMS (Prevention of Relapses and Disability by Interferon beta-la Subcutaneously in Multiple Sclerosis) Study Group. Lancet 1998; 352: 1498–504
Jacobs LD, Cookfair DL, Rudick RA, et al. Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. Multiple Sclerosis Collaborative Research Group (MSCRG). Ann Neurol 1996; 39: 285–94
European Study Group on interferon beta-1b in secondary progressive MS. Placebo-controlled multicentre randomised trial of interferon beta-1b in treatment of secondary progressive multiple sclerosis. Lancet 1998; 352: 1491–7
Stiive O, Bennett JL, Hemmer B, et al. Pharmacological treatment of early multiple sclerosis. Drugs 2008; 68: 73–83
Yong VW. Differential mechanisms of action of interferon-beta and glatiramer acetate in MS. Neurology 2002; 59: 802–8
Neuhaus O, Archelos JJ, Hartung HP. Immunomodulation in multiple sclerosis: from immunosuppression to neuroprotection. Trends Pharmacol Sci 2003; 24: 131–8
Clerico M, Contessa G, Durelli L. Interferon-beta1a for the treatment of multiple sclerosis. Expert Opin Biol Ther 2007; 7: 535–42
Markowitz CE. Interferon-beta: mechanism of action and dosing issues. Neurology 2007; 68: S8–11
Teitelbaum D, Meshorer A, Hirshfeld T, et al. Suppression of experimental allergic encephalomyelitis by a synthetic polypeptide. Eur J Immunol 1971; 1: 242–8
Weber MS, Prod'homme T, Youssef S, et al. Type II monocytes modulate T cell-mediated central nervous system autoimmune disease. Nat Med 2007; 13: 935–43
Tennakoon DK, Mehta RS, Ortega SB, et al. Therapeutic induction of regulatory, cytotoxic CD8+ T cells in multiple sclerosis. J Immunol 2006; 176: 7119–29
Weber MS, Hohlfeld R, Zamvil SS. Mechanism of action of glatiramer acetate in treatment of multiple sclerosis. Neurotherapeutics 2007; 4: 647–53
Johnson KP, Brooks BR, Cohen JA, et al. Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis: results of a phase III multicenter, double-blind placebo-controlled trial. Copolymer 1 Multiple Sclerosis Study Group. Neurology 1995; 45: 1268–76
Comi G, Filippi M, Wolinsky JS. European/Canadian multicenter, double-blind, randomized, placebo-controlled study of the effects of glatiramer acetate on magnetic resonance imaging: measured disease activity and burden in patients with relapsing multiple sclerosis. European/Canadian Glatiramer Acetate Study Group. Ann Neurol 2001; 49: 290–7
Teva Pharmaceutical Industries. A study to evaluate the effect of early glatiramer Aacetate treatment in delaying the conversion to CDMS of subjects presenting with a clinically isolated syndrome (CIS) (PreCISe) [ClinicalTrials.gov identifier NCT00666224]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 10]
Hartung HP, Gonsette R, Konig N, et al. Mitoxantrone in progressive multiple sclerosis: a placebo-controlled, double-blind, randomised, multicentre trial. Lancet 2002; 360: 2018–25
Krapf H, Morrissey SP, Zenker O, et al. Effect of mitoxantrone on MRI in progressive MS: results of the MIMS trial. Neurology 2005; 65: 690–5
Durr FE, Wallace RE, Citarella RV. Molecular and biochemical pharmacology of mitoxantrone. Cancer Treat Rev 1983; 10 Suppl. B: 3–11
Neuhaus O, Wiendl H, Kieseier BC, et al. Multiple sclerosis: mitoxantrone promotes differential effects on immunocompetent cells in vitro. J Neuroimmunol 2005; 168: 128–37
Kopadze T, Dehmel T, Hartung HP, et al. Inhibition by mitoxantrone of in vitro migration of immunocompetent cells: a possible mechanism for therapeutic efficacy in the treatment of multiple sclerosis. Arch Neurol 2006; 63: 1572–8
Neuhaus O, Kieseier BC, Hartung HP. Therapeutic role of mitoxantrone in multiple sclerosis. Pharmacol Ther 2006; 109: 198–209
Yednock TA, Cannon C, Fritz LC, et al. Prevention of experimental autoimmune encephalomyelitis by antibodies against alpha 4 beta 1 integrin. Nature 1992; 356: 63–6
Tubridy N, Behan PO, Capildeo R, et al. The effect of anti-alpha4 integrin antibody on brain lesion activity in MS. UK Antegren Study Group. Neurology 1999; 53: 466–72
O'Connor PW, Goodman A, Willmer-Hulme AJ, et al. Randomized multicenter trial of natalizumab in acute MS relapses: clinical and MRI effects. Neurology 2004; 62: 2038–43
Miller DH, Khan OA, Sheremata WA, et al. A controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 2003; 348: 15–23
O'Connor P, Miller D, Riester K, et al. Relapse rates and enhancing lesions in a phase II trial of natalizumab in multiple sclerosis. Mult Scler 2005; 11: 568–72
Polman CH, O'Connor PW, Havrdova E, et al. A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 2006; 354: 899–910
Rudick RA, Stuart WH, Calabresi PA, et al. Natalizumab plus interferon beta-la for relapsing multiple sclerosis. N Engl J Med 2006; 354: 911–23
Stuve O, Marra CM, Jerome KR, et al. Immune surveillance in multiple sclerosis patients treated with natalizumab. Ann Neurol 2006; 59: 743–7
Yonekawa K, Harlan JM. Targeting leukocyte integrins in human diseases. J Leukoc Biol 2005; 77: 129–40
Monson NL, Cravens PD, Frohman EM, et al. Effect of rituximab on the peripheral blood and cerebrospinal fluid B cells in patients with primary progressive multiple sclerosis. Arch Neurol 2005; 62: 258–64
Berger JR, Koralnik IJ. Progressive multifocal leukoencephalopathy and natalizumab: unforeseen consequences. N Engl J Med 2005; 353: 414–6
Bozic C, Belcher G, Kooijmans-Coutinho M, et al. Natalizumab utilization and safety in patients with relapsing multiple sclerosis: updated results from TOUCH and TYGRIS [abstract]. Neurology 2008; 70 Suppl. 1: A104
Hemmer B, Frohman E, Hartung HP, et al. Central nervous system infections: a potential complication of systemic immunotherapy. Curr Opin Neurol 2006; 19: 271–6
Yao K, Gagnon S, Akhyani N, et al. Reactivation of human herpesvirus-6 in natalizumab treated multiple sclerosis patients. PLoS ONE 2008; 3: e2028
Zohren F, Toutzaris D, Klarner V, et al. The monoclonal anti-VLA-4 antibody natalizumab mobilizes CD34+ hematopoietic progenitor cells in humans. Blood 2008; 111: 3893–5
Stuve O, Marra CM, Bar-Or A, et al. Altered CD4+/CD8+ T-cell ratios in cerebrospinal fluid of natalizumab-treated patients with multiple sclerosis. Arch Neurol 2006; 63: 1383–7
Kappos L, Bates D, Hartung HP, et al. Natalizumab treatment for multiple sclerosis: recommendations for patient selection and monitoring. Lancet Neurol 2007; 6: 431–41
Gold R, Jawad A, Miller DH, et al. Expert opinion: guidelines for the use of natalizumab in multiple sclerosis patients previously treated with immunomodulating therapies. J Neuroimmunol 2007; 187: 156–8
Boster A, Edan G, Frohman E, et al. Intense immunosuppression in patients with rapidly worsening multiple sclerosis: treatment guidelines for the clinician. Lancet Neurol 2008; 7: 173–83
Weiner HL. Immunosuppressive treatment in multiple sclerosis. J Neurol Sci 2004; 223: 1–11
Oger J. Immunosuppression: promises and failures. J Neurol Sci 2007; 259: 74–8
Casetta I, Iuliano G, Filippini G. Azathioprine for multiple sclerosis. Cochrane Database Syst Rev 2007; (4): CD003982
Confavreux C, Saddier P, Grimaud J, et al. Risk of cancer from azathioprine therapy in multiple sclerosis: a case-control study. Neurology 1996; 46: 1607–12
Miller DH, Leary SM. Primary-progressive multiple sclerosis. Lancet Neurol 2007; 6: 903–12
Cree BA, Lamb S, Morgan K, et al. An open label study of the effects of rituximab in neuromyelitis optica. Neurology 2005; 64: 1270–2
Cross AH, Stark JL, Lauber J, et al. Rituximab reduces B cells and T cells in cerebrospinal fluid of multiple sclerosis patients. J Neuroimmunol 2006; 180: 63–70
Bar-Or A, Calabresi PA, Arnold D, et al. Rituximab in relapsing-remitting multiple sclerosis: a 72-week, open-label, phase I trial. Ann Neurol 2008; 63: 395–400
Hauser SL, Waubant E, Arnold DL, et al. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N Engl J Med 2008; 358: 676–88
Coles AJ, Cox A, le Page E, et al. The window of therapeutic opportunity in multiple sclerosis: evidence from monoclonal antibody therapy. J Neurol 2006; 253: 98–108
Coles AJ, Compston DA, for the CAMMS223 Trial Investigators. Alemtuzumab vs. interferon beta-1a in early multiple sclerosis. N Engl J Med 2008; 359: 1786–801
Jones JL, Coles AJ. Campath-1H treatment of multiple sclerosis. Neurodegener Dis 2008; 5: 27–31
Paolillo A, Coles AJ, Molyneux PD, et al. Quantitative MRI in patients with secondary progressive MS treated with monoclonal antibody Campath 1H. Neurology 1999; 53: 751–7
Genzyme. Comparison of alemtuzumab and Rebif® efficacy in multiple sclerosis, study one (CARE-MS I) [ClinicalTrials.gov identifier NCT00530348]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 30]
Genzyme. Comparison of alemtuzumab and Rebif® efficacy in multiple sclerosis, study two (CARE-MS II) [ClinicalTrials.gov identifier NCT00548405]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 30]
Ciancio G, Burke GW, Suzart K, et al. Efficacy and safety of daclizumab induction for primary kidney transplant recipients in combination with tacrolimus, mycophenolate mofetil, and steroids as maintenance immunosuppression. Transplant Proc 2003; 35: 873–4
Bielekova B, Richert N, Howard T, et al. Humanized anti-CD25 (daclizumab) inhibits disease activity in multiple sclerosis patients failing to respond to interferon beta. Proc Natl Acad Sci US A 2004; 101: 8705–8
Rose JW, Watt HE, White AT, et al. Treatment of multiple sclerosis with an anti-interleukin-2 receptor monoclonal antibody. Ann Neurol 2004; 56: 864–7
Biogen Idec. Safety and efficacy study of daclizumab HYP to treat relapsing-remitting multiple sclerosis [ClinicalTrials.gov identifier NCT00390221]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 30]
Tak PP, Thurlings RM, Rossier C, et al. Atacicept in patients with rheumatoid arthritis: results of a multicenter, phase Ib, double-blind, placebo-controlled, dose-escalating, single- and repeated-dose study. Arthritis Rheum 2008; 58: 61–72
Dall'Era M, Chakravarty E, Wallace D, et al. Reduced B lymphocyte and immunoglobulin levels after atacicept treatment in patients with systemic lupus erythematosus: results of a multicenter, phase Ib, double-blind, placebo-controlled, doseescalating trial. Arthritis Rheum 2007; 56: 4142–50
Huntington ND, Tomioka R, Clavarino C, et al. A BAFF antagonist suppresses experimental autoimmune encephalomyelitis by targeting cell-mediated and humoral immune responses. Int Immunol 2006; 18: 1473–85
Munafo A, Priestley A, Nestorov I, et al. Safety, pharmacokinetics and pharmacodynamics of atacicept in healthy volunteers. Eur J Clin Pharmacol 2007; 63: 647–56
EMD Serono. Atacicept in multiple sclerosis, phase II [Clinical-Trials.gov identifier NCT00642902]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 30]
Pirko I, Ciric B, Gamez J, et al. A human antibody that promotes remyelination enters the CNS and decreases lesion load as detected by T2-weighted spinal cord MRI in a virus-induced murine model of MS. FASEB J 2004; 18: 1577–9
Warrington AE, Bieber AJ, Ciric B, et al. A recombinant human IgM promotes myelin repair after a single, very low dose. J Neurosci Res 2007; 85: 967–76
Wiendl H, Hohlfeld R. Therapeutic approaches in multiple sclerosis: lessons from failed and interrupted treatment trials. BioDrugs 2002; 16: 183–200
Brunmark C, Runstrom A, Ohlsson L, et al. The new orally active immunoregulator laquinimod (ABR-215062) effectively inhibits development and relapses of experimental autoimmune encephalomyelitis. J Neuroimmunol 2002; 130: 163–72
Polman C, Barkhof F, Sandberg-Wollheim M, et al. Treatment with laquinimod reduces development of active MRI lesions in relapsing MS. Neurology 2005; 64: 987–91
Comi G, Pulizzi A, Rovaris M, et al. Effect of laquinimod on MRI-monitored disease activity in patients with relapsingremitting multiple sclerosis: a multicentre, randomised, double-blind, placebo-controlled phase lib study. Lancet 2008; 371: 2085–92
Teva Pharmaceutical Industries. BRAVO study: laquinimod double blind placebo controlled study in RRMS patients with a rater blinded reference arm of interferon β-1a (Avonex®) [ClinicalTrials.gov identifier NCT00605215]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Teva Pharmaceutical Industries. Safety and efficacy of orally administered laquinimod for treatment of relapsing remitting multiple sclerosis (RRMS) [ClinicalTrials.gov identifier NCT00509145]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov [Accessed 2008 Oct 21]
Beutler E, Sipe JC, Romine JS, et al. The treatment of chronic progressive multiple sclerosis with cladribine. Proc Natl Acad Sci U S A 1996; 93: 1716–20
Sipe JC, Romine JS, Koziol JA, et al. Cladribine in treatment of chronic progressive multiple sclerosis. Lancet 1994; 344: 9–13
Rice GP, Filippi M, Comi G. Cladribine and progressive MS: clinical and MRI outcomes of a multicenter controlled trial. Cladribine MRI Study Group. Neurology 2000; 54: 1145–55
EMD Serono. Phase II cladribine add-on to inteferon-beta (IFN-b) therapy in MS subjects with active disease (ONWARD) [ClinicalTrials.gov identifier NCT00436826]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
EMD Serono. CLARITY — Safety and Efficacy of Oral Cladribine in Subjects With Relapsing-Remitting MS [ClinicalTrials.gov identifier NCT00213135]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
EMD Serono. Oral cladribine in early multiple sclerosis (MS) (ORACLE MS) [ClinicalTrials.gov identifier NCT00725985]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov [Accessed 2008 Oct 21]
Korn T, Magnus T, Toyka K, et al. Modulation of effector cell functions in experimental autoimmune encephalomyelitis by leflunomide: mechanisms independent of pyrimidine depletion. J Leukoc Biol 2004; 76: 950–60
Korn T, Toyka K, Hartung HP, et al. Suppression of experimental autoimmune neuritis by leflunomide. Brain 2001; 124: 1791–802
Smolen JS, Emery P, Kalden JR, et al. The efficacy of leflunomide monotherapy in rheumatoid arthritis: towards the goals of disease modifying antirheumatic drug therapy. J Rheumatol 2004; 71 Suppl.: 13–20
O'Connor PW, Li D, Freedman MS, et al. A phase II study of the safety and efficacy of teriflunomide in multiple sclerosis with relapses. Neurology 2006; 66: 894–900
Sanofi-Aventis. Phase III study with teriflunomide versus placebo in patients with first clinical symptom of multiple sclerosis [ClinicalTrials.gov identifier NCT00622700]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov [Accessed 2008 Oct 21]
Sanofi-Aventis. An efficacy study of teriflunomide in patients with relapsing multiple sclerosis [ClinicalTrials.gov identifier NCT00751881]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Sanofi-Aventis. Pilot study of teriflunomide as adjunctive therapy to glatiramer acetate in subjects with multiple sclerosis [ClinicalTrials.gov identifier NCT00475865]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Sanofi-Aventis. Pilot study of teriflunomide as adjunctive therapy to interferon-beta in subjects with multiple sclerosis [ClinicalTrials.gov identifier NCT00489489]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Weissert R, Wiendl H, Pfrommer H, et al. Action of treosulfan in myelin-oligodendrocyte-glycoprotein-induced experimental autoimmune encephalomyelitis and human lymphocytes. J Neuroimmunol 2003; 144: 28–37
Wiendl H, Kieseier BC, Weissert R, et al. Treatment of active secondary progressive multiple sclerosis with treosulfan. J Neurol 2007; 254: 884–9
Cavaletti G, Cavalletti E, Crippa L, et al. Pixantrone (BBR2778) reduces the severity of experimental allergic encephalomyelitis. J Neuroimmunol 2004; 151: 55–65
Youssef S, Stuve O, Patarroyo JC, et al. The HMG-CoA reductase inhibitor, atorvastatin, promotes a Th2 bias and reverses paralysis in central nervous system autoimmune disease. Nature 2002; 420: 78–84
Dunn SE, Youssef S, Goldstein MJ, et al. Isoprenoids determine Thl/Th2 fate in pathogenic T cells, providing a mechanism of modulation of autoimmunity by atorvastatin. J Exp Med 2006; 203: 401–12
Menge T, Hartung HP, Stuve O. Statins: a cure-all for the brain? Nat Rev Neurosci 2005; 6: 325–31
Aktas O, Waiczies S, Smorodchenko A, et al. Treatment of relapsing paralysis in experimental encephalomyelitis by targeting Th1 cells through atorvastatin. J Exp Med 2003; 197: 725–33
Stuve O, Youssef S, Weber MS, et al. Immunomodulatory synergy by combination of atorvastatin and glatiramer acetate in treatment of CNS autoimmunity. J Clin Invest 2006; 116: 1037–44
Vollmer T, Key L, Durkalski V, et al. Oral simvastatin treatment in relapsing-remitting multiple sclerosis. Lancet 2004; 363: 1607–8
Paul F, Waiczies S, Wuerfel J, et al. Oral high-dose atorvastatin treatment in relapsing-remitting multiple sclerosis. PLoS ONE 2008; 3: el928
Birnbaum G, Cree B, Altafullah I, et al. Combining beta interferon and atorvastatin may increase disease activity in multiple sclerosis. Neurology. Epub 2008 Jun 4
Yong VW, Wells J, Giuliani F, et al. The promise of minocycline in neurology. Lancet Neurol 2004; 3: 744–51
Hartung HP, Kieseier BC. The role of matrix metalloproteinases in autoimmune damage to the central and peripheral nervous system. J Neuroimmunol 2000; 107: 140–7
Gijbels K, Galardy RE, Steinman L. Reversal of experimental autoimmune encephalomyelitis with a hydroxamate inhibitor of matrix metalloproteases. J Clin Invest 1994; 94: 2177–82
Yong VW, Agrawal SM, Stirling DP. Targeting MMPs in acute and chronic neurological conditions. Neurotherapeutics 2007; 4: 580–9
Yong VW. Metalloproteinases: mediators of pathology and regeneration in the CNS. Nat Rev Neurosci 2005; 6: 931–44
Agrawal SM, Lau L, Yong VW. MMPs in the central nervous system: where the good guys go bad. Semin Cell Dev Biol 2008; 19: 42–51
Popovic N, Schubart A, Goetz BD, et al. Inhibition of autoimmune encephalomyelitis by a tetracycline. Ann Neurol 2002; 51: 215–23
Brundula V, Rewcastle NB, Metz LM, et al. Targeting leukocyte MMPs and transmigration: minocycline as a potential therapy for multiple sclerosis. Brain 2002; 125: 1297–308
Metz LM, Zhang Y, Yeung M, et al. Minocycline reduces gadolinium-enhancing magnetic resonance imaging lesions in multiple sclerosis [letter]. Ann Neurol 2004; 55: 756
Zabad RK, Metz LM, Todoruk TR, et al. The clinical response to minocycline in multiple sclerosis is accompanied by beneficial immune changes: a pilot study. Mult Scler 2007; 13: 517–26
Schilling S, Goelz S, Linker R, et al. Fumaric acid esters are effective in chronic experimental autoimmune encephalomyelitis and suppress macrophage infiltration. Clin Exp Immunol 2006; 145: 101–7
Treumer F, Zhu K, Glaser R, et al. Dimethylfumarate is a potent inducer of apoptosis in human T cells. J Invest Dermatol 2003; 121: 1383–8
Schimrigk S, Brune N, Hellwig K, et al. Oral fumaric acid esters for the treatment of active multiple sclerosis: an open-label, baseline-controlled pilot study. Eur J Neurol 2006; 13: 604–10
Kappos L, Miller DH, MacManus D, et al. Efficacy of a novel oral single-agent fumarate, BG00012, in patients with relapsing-remitting multiple sclerosis: results of a phase 2 study [abstract]. Mult Scler 2006; 12: 85
Biogen Idec. Efficacy and safety of oral BG00012 in relapsingremitting multiple sclerosis (DEFINE) [ClinicalTrials.gov identifier NCT00420212]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Biogen Idec. Efficacy and safety study of oral BG00012 with active reference in relapsing-remitting multiple sclerosis (CONFIRM) [ClinicalTrials.gov identifier NCT00451451]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Lovett-Racke AE, Hussain RZ, Northrop S, et al. Peroxisome proliferator-activated receptor alpha agonists as therapy for autoimmune disease. J Immunol 2004; 172: 5790–8
Klotz L, Schmidt M, Giese T, et al. Proinflammatory stimulation and pioglitazone treatment regulate peroxisome proliferator-activated receptor gamma levels in peripheral blood mononuclear cells from healthy controls and multiple sclerosis patients. J Immunol 2005; 175: 4948–55
Feinstein DL, Galea E, Gavrilyuk V, et al. Peroxisome proliferator-activated receptor-gamma agonists prevent experimental autoimmune encephalomyelitis. Ann Neurol 2002; 51: 694–702
Nessler S, Stadelmann C, Bittner A, et al. Suppression of autoimmune encephalomyelitis by a neurokinin-1 receptor antagonist: a putative role for substance P in CNS inflammation. J Neuroimmunol 2006; 179: 1–8
Reinke EK, Johnson MJ, Ling C, et al. Substance P receptor mediated maintenance of chronic inflammation in EAE. J Neuroimmunol 2006; 180: 117–25
Kim S, Liva SM, Dalai MA, et al. Estriol ameliorates autoimmune demyelinating disease: implications for multiple sclerosis. Neurology 1999; 52: 1230–8
Sicotte NL, Liva SM, Klutch R, et al. Treatment of multiple sclerosis with the pregnancy hormone estriol. Ann Neurol 2002; 52: 421–8
University of California, Los Angeles. A combination trial of copaxone plus estriol in RRMS (Estriol in MS) [ClinicalTrials.gov identifier NCT00451204]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Wells JE, Huribert RJ, Fehlings MG, et al. Neuroprotection by minocycline facilitates significant recovery from spinal cord injury in mice. Brain 2003; 126: 1628–37
Van Muiswinkel FL, Kuiperij HB. The Nrf2-ARE Signalling pathway: promising drug target to combat oxidative stress in neurodegenerative disorders. Curr Drug Targets CNS Neurol Disord 2005; 4: 267–81
Schutz B, Reimann J, Dumitrescu-Ozimek L, et al. The oral antidiabetic pioglitazone protects from neurodegeneration and amyotrophic lateral sclerosis-like symptoms in Superoxide dismutase-G93A transgenic mice. J Neurosci 2005; 25: 7805–12
Klawiter EC, Cross AH. B cells: no longer the nondominant arm of multiple sclerosis. Curr Neurol Neurosci Rep 2007; 7: 231–8
Antel J, Bar-Or A. Roles of immunoglobulins and B cells in multiple sclerosis: from pathogenesis to treatment. J Neuroimmunol 2006; 180: 3–8
Meinl E, Krumbholz M, Hohlfeld R. B lineage cells in the inflammatory central nervous system environment: migration, maintenance, local antibody production, and therapeutic modulation. Ann Neurol 2006; 59: 880–92
Browning JL. B cells move to centre stage: novel opportunities for autoimmune disease treatment. Nat Rev Drug Discov 2006; 5: 564–76
Dillon SR, Gross JA, Ansell SM, et al. An APRIL to remember: novel TNF ligands as therapeutic targets. Nat Rev Drug Discov 2006; 5: 235–46
Avery DT, Railed SL, Ellyard JI, et al. BAFF selectively enhances the survival of plasmablasts generated from human memory B cells. J Clin Invest 2003; 112: 286–97
Thangarajh M, Masterman T, Hillert J, et al. A proliferationinducing ligand (APRIL) is expressed by astrocytes and is increased in multiple sclerosis. Scand J Immunol 2007; 65: 92–8
Krumbholz M, Theil D, Derfuss T, et al. BAFF is produced by astrocytes and up-regulated in multiple sclerosis lesions and primary central nervous system lymphoma. J Exp Med 2005; 201: 195–200
Magliozzi R, Columba-Cabezas S, Serafini B, et al. Intracerebral expression of CXCL13 and BAFF is accompanied by formation of lymphoid follicle-like structures in the meninges of mice with relapsing experimental autoimmune encephalomyelitis. J Neuroimmunol 2004; 148: 11–23
Gross JA, Dillon SR, Mudri S, et al. TACI-Ig neutralizes molecules critical for B cell development and autoimmune disease: impaired B cell maturation in mice lacking BLyS. Immunity 2001; 15: 289–302
Matloubian M, Lo CG, Cinamon G, et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on SIP receptor 1. Nature 2004; 427: 355–60
Brinkmann V, Davis MD, Heise CE, et al. The immune modulator FTY720 targets sphingosine 1-phosphate receptors. J Biol Chem 2002; 277: 21453–7
Rosen H, Goetzl EJ. Sphingosine 1-phosphate and its receptors: an autocrine and paracrine network. Nat Rev Immunol 2005; 5: 560–70
Muller H, Hofer S, Kaneider N, et al. The immunomodulator FTY720 interferes with effector functions of human monocyte-derived dendritic cells. Eur J Immunol 2005; 35: 533–45
Webb M, Tham CS, Lin FF, et al. Sphingosine 1-phosphate receptor agonists attenuate relapsing-remitting experimental autoimmune encephalitis in SJL mice. J Neuroimmunol 2004; 153: 108–21
Kappos L, Antel J, Comi G, et al. Oral fingolimod (FTY720) for relapsing multiple sclerosis. N Engl J Med 2006; 355: 1124–40
Comi G, O'Connor P, Montalban X, et al. Oral FTY720 (fingolimod) in patients with relapsing multiple sclerosis: 3-year extension shows sustained low relapse rate and MRI activity [abstract no. S12.005]. American Academy of Neurology 60th Annual Meeting; 2008 Apr 15, Chicago (IL)
Novartis. Efficacy and safety of fingolimod in patients with relapsing-remitting multiple sclerosis (FREEDOMS) [ClinicalTrials.gov identifier NCT00289978]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov [Accessed 2008 Oct 21]
Novartis. Efficacy and safety of fingolimod (FTY720) in patients with relapsing-remitting multiple sclerosis (FREEDOMS II) [ClinicalTrials.gov identifier NCT00355134]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Goldacre MJ, Seagroatt V, Yeates D, et al. Skin cancer in people with multiple sclerosis: a record linkage study. J Epidemiol Com Health 2004; 58: 142–4
Nielsen NM, Rostgaard K, Rasmussen S, et al. Cancer risk among patients with multiple sclerosis: a population-based register study. Int J Cancer 2006; 118: 979–84
Jung S, Toyka K, Hartung HP. Suppression of experimental autoimmune encephalomyelitis in Lewis rats by antibodies against CD 2. Eur J Immunol 1995; 25: 1391–8
Tran GT, Carter N, He XY, et al. Reversal of experimental allergic encephalomyelitis with non-mitogenic, non-depleting anti-CD3 mAb therapy with a preferential effect on T(h)1 cells that is augmented by IL-4. Int Immunol 2001; 13: 1109–20
Ochi H, Abraham M, Ishikawa H, et al. Oral CD3-specific antibody suppresses autoimmune encephalomyelitis by inducing CD4+. Nat Med 2006; 12: 627–35
Sedgwick JD, Mason DW. The mechanism of inhibition of experimental allergic encephalomyelitis in the rat by monoclonal antibody against CD 4. J Neuroimmunol 1986; 13: 217–32
Weinshenker BG, Bass B, Karlik S, et al. An open trial of OKT3 in patients with multiple sclerosis. Neurology 1991; 41: 1047–52
van Oosten BW, Lai M, Hodgkinson S, et al. Treatment of multiple sclerosis with the monoclonal anti-CD4 antibody cMT412: results of a randomized, double-blind, placebo-controlled, MR-monitored phase II trial. Neurology 1997; 49: 351–7
Stuve O, Cepok S, Elias B, et al. Clinical stabilization and effective B-lymphocyte depletion in the cerebrospinal fluid and peripheral blood of a patient with fulminant relapsingremitting multiple sclerosis. Arch Neurol 2005; 62: 1620–3
Rastetter W, Molina A, White CA. Rituximab: expanding role in therapy for lymphomas and autoimmune diseases. Annu Rev Med 2004; 55: 477–503
McFarland HF. The B cell: old player, new position on the team. N Engl J Med 2008; 358: 664–5
Teva Pharmaceutical Industries Ltd. Early treatment with Copaxone® significantly delayed progression to clinically definite multiple sclerosis [online]. Available from URL: http://www.tevapharm.com/pr/2008/pr_750.asp Accessed 2008 Apr 16]
Multiple Sclerosis Society of Canada. World congress on MS takes place in Montreal [online]. Available from URL: http://www.mssociety.ca/en/research/medmmo_20081002.htm[Accessed 2008 Apr 16]
Eisenberg R, Albert D. B-cell targeted therapies in rheumatoid arthritis and systemic lupus erythematosus. Nat Clin Pract Rheumatol 2006; 2: 20–7
Harris HE. Progressive multifocal leucoencephalopathy in a patient with systemic lupus erythematosus treated with rituximab. Rheumatology (Oxford) 2008; 47: 224–5
Calabrese LH, Molloy ES, Huang D, et al. Progressive multifocal leukoencephalopathy in rheumatic diseases: evolving clinical and pathologic patterns of disease. Arthritis Rheum 2007; 56: 2116–28
Rituxan warning. FDA Consum 2007; 41: 3
Bielekova B, Catalfamo M, Reichert-Scrivner S, et al. Regulatory CD56(bright) natural killer cells mediate immunomodulatory effects of IL-2Ralpha-targeted therapy (daclizumab) in multiple sclerosis. Proc Natl Acad Sci U S A 2006; 103: 5941–6
Rose JW, Burns JB, Bjorklund J, et al. Daclizumab phase II trial in relapsing and remitting multiple sclerosis: MRI and clinical results. Neurology 2007; 69: 785–9
Watt G, Gauden V, McNeill K, et al. Effect of CDP323, a small molecule VLA-4 antagonist, on chronic experimental allergic encephalomyelitis in C57B1/6 mice [abstract]. Mult Scler 2005; 11 Suppl.: 199
Baker M, Shock A, Parton T, et al. Pharmacokinetic and pharmacodynamic properties of the VLA-4 inhibitor CDP323 [abstract]. Mult Scler 2006; 12 Suppl.: 392
UCB. Placebo controlled study in subjects with relapsing forms of MS to evaluate the safety, tolerability and effects of CDP323 [ClinicalTrials.gov identifier NCT00484536]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov[Accessed 2008 Oct 21]
Bar-Or A, Vollmer T, Antel J, et al. Induction of antigenspecific tolerance in multiple sclerosis after immunization with DNA encoding myelin basic protein in a randomized, placebocontrolled phase 1/2 trial. Arch Neurol 2007; 64: 1407–15
Garren H, Robinson WH, Krasulová E, et al. Phase 2 trial of a DNA vaccine encoding myelin basic protein for multiple sclerosis. Ann neurol 2008 May; 63(5): 611–20
Vanderlugt CL, Miller SD. Epitope spreading in immune-mediated diseases: implications for immunotherapy. Nat Rev Immunol 2002; 2: 85–95
Pascual AM, Martinez-Bisbal MC, Bosca I, et al. Axonal loss is progressive and partly dissociated from lesion load in early multiple sclerosis. Neurology 2007; 69: 63–7
Armstrong RJ, Barker RA. Neurodegeneration: a failure of neuroregeneration? Lancet 2001; 358: 1174–6
Comi G, Filippi M. Clinical trials in multiple sclerosis: methodological issues. Curr Opin Neurol 2005; 18: 245–52
Zivadinov R. Can imaging techniques measure neuroprotection and remyelination in multiple sclerosis? Neurology 2007; 68: S72–82
Sergott RC, Frohman E, Glanzman R, et al. The role of optical coherence tomography in multiple sclerosis: expert panel consensus. J Neurol Sci 2007; 263: 3–14
Teunissen CE, Dijkstra C, Polman C. Biological markers in CSF and blood for axonal degeneration in multiple sclerosis. Lancet Neurol 2005; 4: 32–41
Kapoor R. Neuroprotection in multiple sclerosis: therapeutic strategies and clinical trial design. Curr Opin Neurol 2006; 19: 255–9
Bechtold DA, Miller SJ, Dawson AC, et al. Axonal protection achieved in a model of multiple sclerosis using lamotrigine. J Neurol 2006; 253: 1542–51
Pitt D, Werner P, Raine CS. Glutamate excitotoxicity in a model of multiple sclerosis. Nat Med 2000; 6: 67–70
Gilgun-Sherki Y, Panet H, Melamed E, et al. Riluzole suppresses experimental autoimmune encephalomyelitis: implications for the treatment of multiple sclerosis. Brain Res 2003; 989: 196–204
Kalkers NF, Barkhof F, Bergers E, et al. The effect of the neuroprotective agent riluzole on MRI parameters in primary progressive multiple sclerosis: a pilot study. Mult Scler 2002; 8: 532–3
Kapoor R, Davies M, Blaker PA, et al. Blockers of sodium and calcium entry protect axons from nitric oxide-mediated degeneration. Ann Neurol 2003; 53: 174–80
Pryce G, Ahmed Z, Hankey DJ, et al. Cannabinoids inhibit neurodegeneration in models of multiple sclerosis. Brain 2003; 126: 2191–202
Centonze D, Bari M, Rossi S, et al. The endocannabinoid system is dysregulated in multiple sclerosis and in experimental autoimmune encephalomyelitis. Brain 2007; 130: 2543–53
Ullrich O, Merker K, Timm J, et al. Immune control by endocannabinoids: new mechanisms of neuroprotection? J Neuroimmunol 2007; 184: 127–35
Black JA, Liu S, Hains BC, et al. Long-term protection of central axons with phenytoin in monophasic and chronicrelapsing EAE. Brain 2006; 129: 3196–208
Smith KJ. Sodium channels and multiple sclerosis: roles in symptom production, damage and therapy. Brain Pathol 2007; 17: 230–42
Aktas O, Prozorovski T, Smorodchenko A, et al. Green tea epigallocatechin-3-gallate mediates T cellular NF-kappa B inhibition and exerts neuroprotection in autoimmune encephalomyelitis. J Immunol 2004; 173: 5794–800
Killestein J, Kalkers NF, Polman CH. Glutamate inhibition in MS: the neuroprotective properties of riluzole. J Neurol Sci 2005; 233: 113–5
Vaney C, Heinzel-Gutenbrunner M, Jobin P, et al. Efficacy, safety and tolerability of an orally administered cannabis extract in the treatment of spasticity in patients with multiple sclerosis: a randomized, double-blind, placebo-controlled, crossover study. Mult Scler 2004; 10: 417–24
Rudick RA, Cohen JA, Weinstock-Guttman B, et al. Management of multiple sclerosis. N Engl J Med 1997; 337: 1604–11
Racke MK, Dhib-Jalbut S, Cannella B, et al. Prevention and treatment of chronic relapsing experimental allergic encephalomyelitis by transforming growth factor-beta 1. J Immunol 1991; 146: 3012–7
Rodriguez M. Effectors of demyelination and remyelination in the CNS: implications for multiple sclerosis. Brain Pathol 2007; 17: 219–29
Calabresi PA, Fields NS, Maloni HW, et al. Phase 1 trial of transforming growth factor beta 2 in chronic progressive MS. Neurology 1998; 51: 289–92
Mi S, Miller RH, Lee X, et al. LINGO-1 negatively regulates myelination by oligodendrocytes. Nat Neurosci 2005; 8: 745–51
Mi S, Hu B, Hahm K, et al. LINGO-1 antagonist promotes spinal cord remyelination and axonal integrity in MOG-induced experimental autoimmune encephalomyelitis. Nat Med 2007; 13: 1228–33
Wang S, Sdrulla AD, diSibio G, et al. Notch receptor activation inhibits oligodendrocyte differentiation. Neuron 1998; 21: 63–75
Imbimbo BP. Therapeutic potential of gamma-secretase inhibitors and modulators. Curr Top Med Chem 2008; 8: 54–61
Jurynczyk M, Jurewicz A, Bielecki B, et al. Inhibition of Notch signaling enhances tissue repair in an animal model of multiple sclerosis. J Neuroimmunol 2005; 170: 3–10
Jurynczyk M, Jurewicz A, Bielecki B, et al. Overcoming failure to repair demyelination in EAE: gamma-secretase inhibition of Notch signaling. J Neurol Sci 2008; 265: 5–11
Rodriguez M, Lennon VA. Immunoglobulins promote remyelination in the central nervous system. Ann Neurol 1990; 27: 12–7
Miller DJ, Sanborn KS, Katzmann JA, et al. Monoclonal autoantibodies promote central nervous system repair in an animal model of multiple sclerosis. J Neurosci 1994; 14: 6230–8
Trebst C, Stangel M. Promotion of remyelination by immunoglobulins: implications for the treatment of multiple sclerosis. Curr Pharm Des 2006; 12: 241–9
Stuve O, Marra CM, Cravens PD, et al. Potential risk of progressive multifocal leukoencephalopathy with natalizumab therapy: possible interventions. Arch Neurol 2007; 64: 169–76
Ghalie RG, Mauch E, Edan G, et al. A study of therapy-related acute leukaemia after mitoxantrone therapy for multiple sclerosis. Mult Scler 2002; 8: 441–5
van Oosten BW, Truyen L, Barkhof F, et al. Choosing drug therapy for multiple sclerosis: an update. Drugs 1998; 56: 555–69
Kesselring J, Beer S. Symptomatic therapy and neurorehabilitation in multiple sclerosis. Lancet Neurol 2005; 4: 643–52
Stankoff B, Waubant E, Confavreux C, et al. Modafinil for fatigue in MS: a randomized placebo-controlled double-blind study. Neurology 2005; 64: 1139–43
Pucci E, Branas P, D'Amico R, et al. Amantadine for fatigue in multiple sclerosis. Cochrane Database Syst Rev 2007; (2): CD002818
Martino G, Pluchino S. The therapeutic potential of neural stem cells. Nat Rev Neurosci 2006; 7: 395–406
Blanco Y, Saiz A, Carreras E, et al. Autologous haematopoieticstem-cell transplantation for multiple sclerosis. Lancet Neurol 2005; 4: 54–63
Byun E, Caillier SJ, Montalban X, et al. Genome-wide pharmacogenomic analysis of the response to interferon beta therapy in multiple sclerosis. Arch Neurol 2008; 65: 337–44
Acknowledgements
Dr Menge was a postdoctoral fellow of the National MS Society and was supported by grants from the Forschungskommission of the Medical Faculty of the Heinrich-Heine-University, the German MS Society. Dr Stüve was supported by a Start-up Grant from the Dallas VA Research Corporation, a New Investigator Award from VISN 17, Department of Veteran’s Affairs, a Merit Review Award from the Department of Veteran’s Affairs, Research Grants from National Multiple Sclerosis Society (NMSS; RG3427A8/T, and RG2969B7/T), and a grant from the Viragh Foundation. Dr Hemmer was supported by the Deutsche Forschungsgemeinschaft (He2386/4-2, 7-1), the Gemeinnützige Hertie-Stiftung, and the MS Society Chapter of Düsseldorf.
Dr Hemmer has received honoraria from Roche, Amgen, Novartis and Biogen Idec, and research grants from Teva-Aventis, Bayer, Merck-Serono and Biogen Idec. Dr Zamvil has received honoraria from Teva Neuroscience, Biogen Idec and Genentech, and research grants from the National Institutes of Health, National Multiple Sclerosis Society and the Maisin Foundation and Dana Foundation. Dr Khan has received honoraria and research grants from Teva, Biogen Idec, Bayer, Serono and Genentech. Dr Hartung has received fees for consultancies and speaking at symposium from Bayer, Biogen Idec, BioMS, BayerSchering, Merck-Serono, Sanofi/Teva/Aventis, Novartis and Genzyme with approval of the CEO of the University of Düsseldorf hospital and the Dean of the Heinrich-Heine University, Düsseldorf. Dr Hartung’s department has received unrestricted grants for research projects from BayerSchering, Biogen Idec, Merck-Serono, and Teva with approval of the CEO of the University of Düsseldorf hospital and the Dean of the Heinrich-Heine University, Düsseldorf. Dr Stüve has received honoraria from or acted as a consultant to Teva Neuroscience, Merck-Serono, Novartis, Genetech and Biogen Idec. Drs Menge, Weber, Kieseier, von Büdingen, Warnke and Boster have no conflicts of interest that are directly relevant to the contents of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Menge, T., Weber, M.S., Hemmer, B. et al. Disease-Modifying Agents for Multiple Sclerosis. Drugs 68, 2445–2468 (2008). https://doi.org/10.2165/0003495-200868170-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/0003495-200868170-00004